Editor's note: This review of the differences between adult and infant skin provides chemists who are new to the baby care arena with an understanding of special formulating considerations for this market segment.
The skin’s primary function is to provide a protective barrier that maintains internal hydration and electrolytes while preventing entry of exogenous substances such as bacteria, allergens, particulates, pollution, etc. The outer- most layers of the skin, the stratum corneum (SC), are often described as a bricks (cells)-and-mortar (lipid) structure. Scientist and clinicians believe that this structure creates a tortuous path through the “mortar” that inhibits the flow of water from the skin and contaminants into the skin. Cleansing infants is critical for both infant and family hygiene. Microbial colonization occurs shortly after birth, and a baby’s proclivity to be in contact with many substances is well-known.
Cleansing removes unwanted surface material and can prevent infection, ingestion, and transmission of microbes and matter on the surface of the baby skin. In addition to the hygiene benefits, bathing also provides an important opportunity for tactile contact between the baby and caregiver, and promotes bonding.1-3 As is true with any cleansing and particularly important in baby cleansing, the benefit of hygiene needs to be balanced with the preservation of the skin barrier.4,5 In order to maximize skin health, the appropriate balance between effective yet mild skin cleansing must be attained. Washing the skin with water alone provides insufficient cleansing and removal of unwanted material, such as fecal enzymes and urine components; therefore it is recommended to wash babies with a mild baby cleanser instead of only water.3
Baby and Adult Skin
There are significant differences between adult and baby skin, and these structural and functional differences create different needs from skin care products. Responding to the shift to an air environment at birth, the skin barrier rapidly adapts in the first days of life.6,7 The healthy baby skin barrier continues to develop during the first few years of life.8 The SC provides strong resistance to water transport across the skin, and in babies, this important layer is thinner than it is in adults. Additionally, Nikolovski et al. observed that the concentration of natural moisturizing factors (NMF), which act as humectants in the SC, is lower in babies than in adults.8
These structural and compositional differences result in functional differences between baby and adult skin. Giusti et al. observed that baby skin hydration, as measured by conductance, is higher than that of adult skin.9 Also, transepidermal water loss (TEWL) through baby skin is higher than through adult skin.8,10 Differences have been observed in the skin’s water-handling properties between adults and babies because of this developing barrier; babies have been shown to have a higher rate of water adsorption and desorption.8 This also suggests that the resistance to water flux through baby skin is lower than through adult skin. Additionally, baby skin has a greater tendency to develop irritant/allergic contact dermatitis, and it has been suggested that over-aggressive cleansing could lead to the development of atopic dermatitis.11
In addition to the differences between baby and adult skin, babies have a greater vulnerability to ocular exposure. Babies have neither a fully developed blink reflex nor a behavioral blink response in anticipation of accidental exposure, such as during bathing.
Historical Perspective on Baby Cleansing
New innovations continue to redefine what appropriate cleansing is for babies. Figure 1 shows the evolution of personal care cleansing over time; most of the progress made by new generations of cleansers reflects improvements in skin compatibility. Until the late 1800s, most personal care cleansing was performed with lye soap, sodium tallowate, which was made by hand or in small batches. These early soaps were often of inconsistent quality; however, with the advent of mass production during the industrial revolution, soaps of improved quality and purity could be obtained.
A number of larger personal care companies emerged at this time. In 1876, Ivorya began selling soap and shortly thereafter, in 1878, the company began marketing pure soap for babies. As the only cleansing option available, the soap was offered for all cleansing needs: “With Ivory Soap, she can give baby his daily bath; she can keep his bottle clean; she can wash his underwear, his socks … .”12 Unilever also promoted the use of Swan soap all household needs: “Swan is four swell soaps in one ... baby … dishes … duds … you.”13 Then into the 1940s, although soap was still the only cleanser, baby skin was acknowledged as having unique needs. Ads of the time reflected this: “Palmolive just can’t irritate your gentle skin [baby], or mine [mother] either … everything that Palmolive does for your baby, it will do for your complexion, too!”14
Raw material scarcity caused by supply disruptions during World War II drove the development of alternative chemistries and brought about rapid technological advances throughout society. Perhaps the most important development for the personal care industry was the emergence of synthetic detergents, petroleum-derived ingredients that could replace scarce plant oil-derived surfactants.
While soap bars have an alkaline pH, the new syndet bars were comprised of synthetic detergents and allowed for a lower, more skin-neutral pH and thereby healthier, more skin-compatible cleansing.15 Synthetic surfactants also enabled the popularization of liquid detergents that offered pH-neutral skin cleansing.
Using synthetic nonionic surfactants additionally allowed for cleansing without causing irritation to the eyes, a particularly important benefit for baby care. In 1953, Johnson & Johnson introduced the first liquid baby shampoo. This mild baby cleanser again redefined what constitutes appropriate baby cleansing;16 in fact, 2009 marks the 50th anniversary of the trademarked “No More Tears” claim associated with the product.17
Surfactant/Tissue Interactions
Cleansers can interact with skin in various ways, so mild and gentle cleansing for babies requires multiple considerations. A number of studies and reviews document how surfactants can interact with skin.18–21 There is ambiguity about which physical characteristics of a surfactant system are the most important for mildness to skin and skin compatibility. This is likely because of the multiple ways surfactants can affect skin. The dilemma is not just academic; a fundamental understanding can be critical when designing mild cleansing systems.
Initial advances focused on cleanser pH, which is an important difference between soap, glycerin soaps and syndet bars. Nonetheless, liquid surfactant systems can readily be formulated to a skin compatible pH value (below 7.0), and most are. Ideally, a cleanser should remove unwanted materials and interact minimally with the skin.
Understanding surfactant solution behavior is critical for designing effective skin-compatible cleansers. Generally, surfactants comprise a hydrophobic tail group and a hydrophilic head group. In solution, the vast majority of surfactant molecules exist as self-assembled species called micelles, which are accompanied by a smaller population of free monomeric species.22 The hydrophobic group of the surfactant partitions to the micelle interior while the hydrophilic moiety is presented to the water.
Due to their amphiphilic nature, surfactants can potentially solublize and remove the components of the SC lipid mortar, namely ceramides, cholesterol and free fatty acids. Cleansing can also remove the more polar NMF. In addition to the removal of skin components, surfactants that are left behind after rinsing can disrupt the complex structure of the skin. The specific ratio of the different ceramides, cholesterol and free fatty acids has a significant effect on the packing and structure of the mortar and therefore, the effectiveness of the barrier.
It has been shown that surfactants can effectively insert themselves into model bilayer systems.23 The interaction and disruption of this important skin structure could lead to reduced barrier function. Surfactants can also denature and solublize proteins. These modes of surfactant interaction with skin can weaken the barrier, increase TEWL and lower hydration, which in turn can lead to skin redness, dryness, irritation and itch.
Measuring Mildness
There are several perspectives about how to best describe surfactant mildness to skin. In the classic approach, the starting point has been a sodium lauryl sulfate (SLS) surfactant system, and the question has been what can be added to improve the mildness of SLS. From this perspective, the surfactant monomer theory developed, which holds that only surfactant monomers, being relatively small, are able to penetrate the SC.20,24 Guided by this theory, the formulator’s goal has been to reduce the concentration of the irritating monomeric surfactant species, and the micelles have been considered less important.
It is well-accepted that surfactant-induced skin irritation is a function of surfactant concentration (i.e., irritation increases with increasing surfactant dose). However, the monomer model would predict that the irritation response should reach a maximum at the critical micelle concentration (CMC) of the surfactant system and then remain constant with increasing surfactant concentration, since surfactants in micellar form should not contribute to irritation. More recently, Blankschtein and co-workers25–27 suggested that surfactant micelles are capable of penetrating the SC, and therefore both monomeric and micellar surfactant species should be considered.
Additionally, the presence of surfactant micelles in solution is necessary for solublization of lipid material, and the presence of micelles has been observed to increase the rate of vesicle disruption and leakage, suggesting an important role of surfactant micelles.28 Micelle size and surface charge29 are also important physical characteristics, both of which affect micelle stability. Surfactant systems are highly dynamic with rapid exchange between monomeric and micellar states; surfactants move in and out of micelles on the order of milliseconds, and micelle lifetimes (micelles associate and dissociate) are on the order of seconds.22
Many in vitro tests are used to quantify cleanser mildness, with each having strengths and weaknesses. As mentioned, baby cleansers are the mildest surfactant systems, so it is important to select a methodology that can discriminate at this mild end of the spectrum.
TEP, also known as fluorescein dye leakage, is widely used to assess the aggressiveness of surfactant systems and their compatibility with tissues, which have been described.30,31 Briefly, dye leakage across a monolayer of epithelial cells is quantified at various cleanser dilutions. Assessing cleanser effects on the trans-epithelial permeability of a tight epithelium,32 such as occurs in the ocular surface, enables effective differentiation of mild cleansers. Additionally, the response of differentiated epidermal skin equivalents have proven useful for discriminating products at the mild end of the spectrum.33 Other tests, such as Zein protein solubilization, are more effective at distinguishing between harsher systems and less useful for baby cleansers.
Baby vs. Adult Cleansers
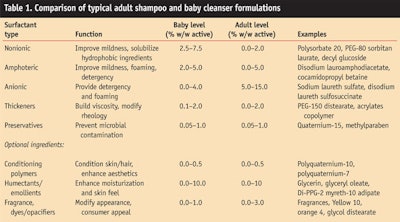
During the design of baby cleansers it is critical to consider the differences between adult and baby skin, as outlined above. Appropriate skin-compatible cleansing for babies is significantly less aggressive and milder than adult cleansing. Table 1 shows the contents of typical baby and adult cleansers. Adult cleansers usually contain relatively high levels of anionic surfactants such as sodium lauryl/laureth sulfate. They also frequently contain amphoteric surfactants, i.e., cocamidopropyl betaine. In contrast, baby cleansers are comprised primarily of amphoteric and nonionic surfactants (PEG-80 sorbitan laurate), and, when they are present, anionic surfactants in relatively lower levels; also, the total surfactant concentration is lower in baby cleansers.
In solution, mixed micelles form that contain blends of the surfactants present. The properties of the cleansers, including mildness, foaming and cleansing, are determined by the properties of these mixed micelles—micelle size, surface charge and dynamics. In Figure 2,
representations of mixed micelles from a typical adult shampoo, body wash and baby cleanser are schematically represented. Differences in micelle compositions lead to physical property and performance differences in the resulting cleansers. For instance, in baby cleansers, the addition of nonionic surfactant to the blend creates larger micelles with a lower micelle charge, resulting in reduced aggressiveness of the surfactant system.
The micelle size distribution of a typical adult shampoo and baby cleanser are shown in Figure 3, as determined by dynamic light scattering at typical in-use concentrations. Micelles of adult shampoos, primarily comprised of sodium lauryl sulfate and sodium laureth sulfate, are smaller than those of a typical baby cleanser. This smaller micelle size in adult shampoos results in differences in cleanser performance and mildness.
The aggressiveness of representative adult body wash, shampoo and baby cleanser are shown in Figure 4. The amount of dye leakage increases with increasing cleanser concentration as the tight junctions between the epithelial cells are disrupted, allowing dye across the cellular monolayer. The adult shampoo system disrupts the tight junctions between cells at the lowest product concentrations indicating a highly aggressive surfactant system, whereas the body wash induces disruption at higher concentrations.
In contrast, the tight junction can tolerate exposure to baby cleanser over a broader range of concentrations; relatively high concentrations of baby cleanser are required to elicit a response similar to the adult cleansers. Correspondingly, at equivalent concentrations less leakage is observed with the typical baby cleanser than adult body wash and shampoo. This indicates that the typical baby cleanser is significantly less disruptive to tissues and milder than the typical adult body wash or shampoo.
Designing more skin-compatible cleansers tends to involve a trade-off of foam for improved mildness, as is evident in these different cleanser types. Figure 5 shows the aggressiveness of a number of commercial cleansers as a function of the volume of foam generated. Adult shampoos are observed to be the harshest and highest-foaming systems; slightly milder and less-foaming are adult body washes; and much milder and lowest-foaming are baby cleansers.
Conclusions and Recent Developments
Baby skin continues to mature during the first years of life and thus requires milder, gentler cleansing than adult skin. While cleansing with water alone is insufficient, typical adult cleansers are also inappropriate for baby skin, since their surfactant systems are too aggressive. In addition to containing less total surfactant, baby cleanser surfactant blends create larger micelles and result in less aggressive albeit lower-foaming systems.
The formulator’s challenge is to design aesthetically desirable baby cleansers without compromising the critical requirements of mildness and skin compatibility. Not limited to baby cleansing, this trade-off between mildness and performance is present across the cleansing category; many consumers (e.g., individuals with sensitive skin, eczema, or atopic dermatitis) may also benefit from cleansers with increased skin compatibility.
There are two current developments in mild baby cleansing that are worth noting. First, cleansers are increasingly being formulated with surfactants that are partially derived from renewable sources, as opposed to petroleum. Commonly, plant oils are chemically modified with a polar group to create these natural surfactants. While not inherently milder, natural surfactants suffer the same trade-off between mildness and performance; however, they do broaden the palette available to formulators.
Second, recent technical advances in the understanding of surfactant-tissue interactions have led to the use of hydrophobically modified polymers (HMP) to create cleansers with improved mildness. It has been shown that polymer-surfactant association with HMP is a practical method for effectively reducing the free micelle concentration and thereby reducing the aggressiveness of the cleansers.34 The addition of HMPs to cleansing systems allows the formulator to attain new levels of skin compatibility for enhanced consumer benefits.
References
1. J Bryanton, D Walsh, M Barrett, D Gaudet, Tub bathing versus traditional sponge bathing for the newborn. J Obstet Gynecol Neonatal Nurs 2004; 33: 704–12.
2. GL Darmstadt, JG Dinulos, Neonatal skin care. Pediatr Clin North Am 2000; 47: 757–82.
3. U Blume-Peytavi, MJ Cork, J Faergemann, J Szczapa, FV Sebastian and C Gelmetti, Bathing and cleansing in newborns from day one to first year of life: Recommendations from a European round table meeting, J Eur Acad Dermatol (in press)
4. C Gelmetti, Skin cleansing in children, J Euro Acad Derm Vener 15 (suppl. 1) 12–15 (2001)
5. T Penny-MacGillivray, A newborn’s first bath: When, J Obst Gynec Neonatal Nursing 25 481–487 (1996)
6. KG Hernes, L Morkid, A Fremming, S Odegarden, OG Martinsen and H Storm, Skin conductance changes during the first year of life in full-term infants, Pediatr Res 52 837–43 (2002)
7. MO Visscher, R Chatterjee, JP Ebel, AA LaRuffa and SB Hoath, Biomedical assessment and instrumental evaluation of healthy infant skin, Pediatr Dermatol 19 473–81
(2002)
8. J Nikolovski, GN Stamatas, N Kollias and BC Wiegand, Barrier function and water-holding and transport properties of infant stratum corneum are different from adult and continue to develop through the first year of life, J Invest Dermatol 128(7) 1728–36
(2008)
9. F Giusti, A Martella, L Bertoni and S Seidenari, Skin barrier, hydration and pH of the skin of infants under two years of age, Pediatr Dermmatol18 93–6 (2001)
10. VA Harper and N Rutter, Barrier properties of the newborn infant’s skin, J Pepiatr 102 419–25 (1983)
11. RE Callard and JI Harper, The skin barrier, atopic dermatitis and allergy: A role for Langerhans cells?, TRENDS in Immunology 28 (7) 294–298 (2007)
12. Scribner’s Magazine advertisement, author unknown,136 (1900)
13. Good Housekeeping Magazine advertisement, author unknown (1941)
14. Life Magazine advertisement, author unknown (1942)
15. KP Ananthapadmanabhan, DJ Moore, K Subramanyan, M Misra and F Meyer, Cleansing without compromise: The impact of cleansers on the skin barrier and the technology of mild cleansing, Dermatol Ther 17(1) 16–25
(2004)
16. US Pat 2,999,069, Detergent Composition, JN Masci, NA Poirier, assigned to Johnson & Johnson (Sept 5, 1961)
17. US 791,169 a registered trademark of Johnson & Johnson Corp. (Jun 15, 1965)
18. A Mehling, M Kleber and H Hensen, Compartive studies on the ocular and dermal irritation potential of surfactants, Food and Chemical Toxicology 45 747–758 (2007)
19. LD Rhein, CR Robbins, K Frernee and RJ Contore, Surfactant structure effects on swelling of isolated human stratum corneum, J Soc Cosmet Chem 37 125 (1986)
20. MM Rieger, Surfactant interactions with skin, Cosm Toil 110(4) 31–50
(1995)
21. LD Rhein, Review of propterties of surfactants that determine their interaction with stratum corneum, J Soc Cosmet Chem 48 253–274 (1997)
22. R Zana, Dynamics of Surfactant Self-assemblies, Boca Raton, FL: CRC Press
(2005)
23. A Lopez-Castellano, C Cortell-Ivars, G Lopez-Carballo and M Herraez-Dominguez, The influence of Span 20 on the stratum corneum lipids in Langmuir monolayers: Comparison with Azone Intl J of Pharma 203 245–253 (2000)
24. AJ O’Lenick, Jr, Surfactants: Strategic Personal Care Ingredients, Allured Business Media: Carol Stream, IL (2005)
25. PN Moore, S Puvvada and D Blankschtein, Challenging the surfactant monomer skin penetration model: Penetration of sodium dodecyl sulfate micelles into the epidermis, J Cosmetic Sci 54(1), 29–46 (2003)
26. PN Moore, A Shiloach, S Puvvada and D Blankschtein, Penetration of mixed micelles into the epidermis: Effect of mixing sodium dodecyl sulfate with dodecyl hexa(ethylene oxide), J Cosmetic Sci 54(2) 143–159 (2003)
27. S Ghosh and D Blankschtein, Why is sodium cocoyl isethionate (SCI) mild to the skin barrier?—An in vitro investigation based on the relative sizes of the SCI micelles and the skin aqueous pores, J Cosmetic Sci 58(3) 229–244 (2007)
28. M Apel-Paz, GF Doncel and TK Vanderlick, Membrane perturbation by surfactant candidates for STD prevention, Langmuir 19 591–597 (2003)
29. A Lips, KP. Ananthapadmanabhan, M Vethamuthu, XY Hua, L Yang, C Vincent: Role of surfactant charge in protein denaturation and surfactant-induced skin irritation, Chapter 9 in Surfactants in Personal Care Products and Decorative Cosmetics L D Rhein, M Schlossman, A O’Lenick, P Somasundaran, eds, Boca Raton, FL: CRC Press (2006) pp 177–187
30. F Van Goethem et al, Prevalidation of a new in vitro reconstituted human cornea model to assess eye irritation potential of chemicals, Toxicology In Vitro 20 1–17 (2006)
31. K Martin and C Stott, Trans-epithelial permeability assay for predicting ocular irritation of surfactant formulations, International Congress on Cell and Tissue Culture, Washington, D.C., USA (1992)
32. R Tchao, Trans-epithelial permeability of fluorescein in vitro as an assay to determine eye irrititants, in: AM Goldberg (ed), Alternative Methods in Toxicology, Mary Ann Liebert Inc: New York (1988) pp 271–283
33. LP Bernhofer, S Barkovic, Y Appa and KM Martin, IL-1a and IL-1ra Secretion from epidermal equivalents and the prediction of the irritation potential of mild soap and surfactant-based consumer products, Toxicology In Vitro (13) 231–239 (1999)
34. MJ Fevola, JJ LiBrizzi, RM Walters, Reducing Irritation Potential of Surfactant-Based Cleansers with Hydrophobically-Modified Polymers, Polymer Preprints 49(2) 671–672 (2008)