The microbiology quality control laboratory performs a critical function for cosmetic product manufacturing in safeguarding the quality, integrity and safety of finished products. Under the U.S. Food, Drug and Cosmetic Act (FD&C Act), the U.S. Food and Drug Administration (FDA) is charged with ensuring cosmetic products are safe. In order to fulfill this responsibility, the FDA may enter an establishment to conduct an inspection.
The portion of the FD&C Act relevant to microbiology refers to an adulterated cosmetic as one that contains any poisonous or deleterious substance, has become filthy or contaminated, or has been prepared, packed or held under insanitary conditions.1 Thus, the role of the microbiology laboratory in assuring unadulterated finished products is intrinsic to a compliant manufacturing process.
FDA inspections have serious regulatory implications and cosmetic manufacturers should have a documented standard procedure in place to manage an FDA inspection. The procedure should describe both the FDA’s and the company’s rights, and outline the company’s practices to be followed during an inspection. It is important that the appropriate personnel are trained for the inspection process and that the written procedure is made available for reference during an inspection.2
The importance of preparation for an inspection cannot be underestimated. Understanding the FDA’s expectations and preparing your facility accordingly will demonstrate good management practices. In the event that a contracted facility is utilized for testing, the same principles and expectations apply; i.e., a written agreement should be established and both the manufacturer and contractor should work together to maintain quality oversight.
In a cosmetic manufacturing facility, the FDA inspector is entitled to inspect all pertinent manufacturing and filling equipment, finished and unfinished materials, storage containers, labeling materials and personnel qualification and training records associated with the manufacturing of the product. Documents may also be requested by the inspector and must be readily accessible. These may include: cleaning and sanitization protocol and records, water validation testing and records, equipment validation and records, environmental monitoring data and training procedures and records. Since the laboratory is often a key focus by the FDA inspector, conducting a mock inspection or performing routine internal audits is advisable.2
Personnel
Laboratory personnel should have the education, training and experience necessary to perform their job responsibilities. Their core educational background should be in microbiology or a closely related biological field. Responsibilities assigned to each laboratory person should commensurate with their skill level and experience.
Job descriptions: A document should be in place describing the responsibilities for each role within the microbiology laboratory. This description should provide the information needed for each individual to successfully perform in the laboratory—and be made readily available for review.
Each job description should include the title for that position and the title for that position’s immediate supervisor. Ideally, the key functional and relational responsibilities in the order of significance for the position should be included. There should also be an objective statement, summary of the general nature and level of the job, as well as a description of the broad function and scope of the position. A list of the duties or tasks for which the position is responsible should also be clearly described.
Organizational charts: Documentation should be in place describing the reporting relationships within the microbiology laboratory, as well as within the quality system. This organizational chart should illustrate the direct line of communication microbiology personnel have with executive level personnel of the company.
The FDA inspector is entitled to inspect all pertinent equipment, finished and unfinished materials, storage containers and more.
Training Records
The regular training of each person within a laboratory is critical to its successful operation. Importantly, all training should also be documented and it is the responsibility of each company to provide an organized system for maintaining training records. Documented training should exist for any individual in the laboratory and be centered toward their specific responsibilities and tasks; as noted, the demands of microbiological testing require that the core educational background of the staff, supervisors and managers is in microbiology or a closely related biological science.
Furthermore, no laboratory person should independently conduct a microbial test until they are qualified to run it. A coherent system of standard operating procedures (SOPs) and methods is required for the successful operation of a microbiology laboratory. The SOPs should describe, in detail, the methodology the microbiologist will follow to obtain accurate and reproducible results.
Performance assessments or proficiency testing should accompany the training for each SOP and/or method as well. This testing should be designed to demonstrate that laboratory microbiologists understand and comprehend each SOP. Periodic performance testing should also provide evidence of competency in key activities of the microbiology laboratory such as hygiene, plating, aseptic technique, documentation and others, as indicated in each job function. Proficiency testing should occur at a scheduled frequency, which can be used as a tool to continually improve the performance of the microbiologists and serve as a means for improving methods.
Finally, for each laboratory employee, a training record or log should be established. The documentation should include such items as training and dates whereby proficiency has been demonstrated for each particular test method, technique and policy or procedure used by that individual during a work day.
Microbiologists with supervisory or managerial responsibilities should have the appropriate education or training in supervisory skills, laboratory safety, scheduling, budgeting, investigational skills, technical report writing, relevant SOPs and other critical aspects of their role. As would be expected, laboratory supervisors and managers should demonstrate a level of competence in microbiology at least as high as those they supervise. Additionally, laboratory supervisors and managers should be encouraged to improve their level of competency by obtaining certifications from accredited bodies and engaging in relevant continuing education.
Microbiology Laboratory Facilities
Adequate laboratory facilities should be provided to minimize the possibility of errors in the test results for a given sample due to accidental microbial contamination, equipment failure or sampling mistakes. For example, separate areas should be kept for conducting microbial analysis of samples and support functions such as media preparation, sample preparation, sample login and storage of personal belongings. Furthermore, all surfaces in the laboratory should be nonporous, cleanable and sanitizable.
The design of the laboratory should include: adequate lighting and ventilation to minimize air currents; adequate electrical service appropriate for the equipment used; adequate sink areas with hot and cold water; and restricted access in order to minimize foot traffic by non-laboratory personnel. Finally, the laboratory should have sufficient counter and shelf space available to prevent overcrowding, safety hazards or cross-contamination between samples and/or microorganisms.
A master equipment list should be kept for calibration and maintenance purposes, along with individual logs for service and calibration dates.
Microbiology Laboratory Equipment
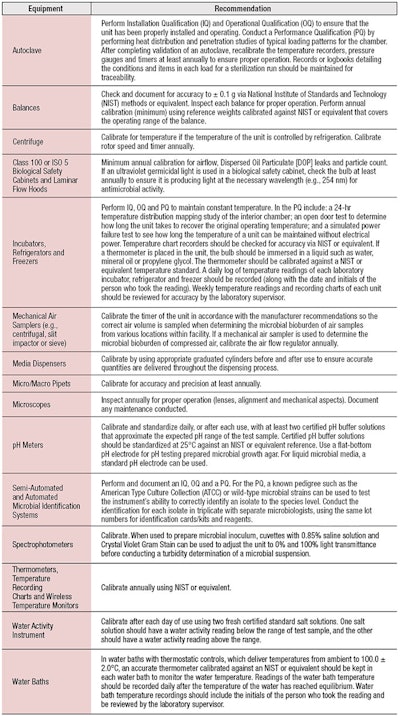
Regarding laboratory equipment, each piece should have a standard operating procedure as well as supporting documentation, e.g., equipment manuals and calibration data, readily available for review. All laboratory equipment should be maintained according to the manufacturer’s directions and routinely calibrated as necessary, as indicated by a calibration sticker bearing the calibration date, calibrator’s name and the next scheduled date for calibration.
Furthermore, a master equipment list should be kept for calibration and maintenance and each piece of equipment should have a maintenance log containing service reports for dates of service, repairs and whether re-calibration was necessary. If a piece of laboratory equipment is malfunctioning or out of calibration, it should be removed for service and labeled accordingly. Malfunctioning or uncalibrated equipment should not be used until it has been repaired and calibrated; Table 1 describes the minimum recommendations for calibrating specific pieces of equipment.
In addition to this list, other laboratory equipment such as disposable plastic (e.g., polystyrene) or reusable labware (e.g., test tubes, pipets, Petri dishes, etc.) should be chemically inert. Borosilicate glass is recommended for reusable glassware due to its ability to withstand high autoclave temperatures. Screw caps for dilution bottles and tubes also should be equipped with inert liners.
Water treatment systems are often used in microbiology laboratories to produce the desired grade of water, e.g., deionized or distilled, to prepare microbial growth media and other applications. A standard operating procedure should be provided to describe how to use and maintain the laboratory water system. Periodically, water produced by this unit should be tested to ensure it is meeting the desired microbial and chemical quality attributes, and a logbook should be kept to document when maintenance on the equipment is performed. If a UV light is part of the water system, it should be maintained and operated according to the manufacturer’s directions.
Conclusions
In order to adequately prepare for an FDA microbiology lab inspection, the described documentation for personnel training, procedures, lab facilities and equipment are essential. Part II in this discussion, in the July/August edition, will cover microbial growth media, conditions, etc. Finally, conducting periodic mock inspections and internal audits of your microbiology lab are advisable.
References
- http://uscode.house.gov/view.xhtml?req=(title:21%20section:361%20edition:prelim) (Accessed Apr 3, 2018)
- JM Nikitakis, ed, Personal Care Products Council Quality Assurance Guidelines, FDA Inspections, The Personal Care Products Council, Washington, DC USA (2014)
- JF Krowka and BA Jonas, eds, Personal Care Products Council Microbiology Guidelines, Microbial Validation and Documentation, The Personal Care Products Council, Washington, DC USA (2016)
- JF Krowka and BA Jonas, eds, Personal Care Products Council Microbiology Guidelines, Microbiology Laboratory Audit, The Personal Care Products Council, Washington, DC USA (2016)
- United States Pharmacopeia, 341-National Formulary-364 (2017), Microbiological Best Laboratory Practices <1117>, United States Pharmacopeia Convention, Inc., Rockville, MD USA (2017)