As is generally known, skin is a unique barrier comprising, from outside to inside, the epidermis, dermis and hypodermis; and that it includes appendages such as hair follicles, sweat and sebaceous glands. From a penetration perspective, the epidermis and dermis appear to be the most important components.
Skin Function
The main barrier function of the skin is attributed to the stratum corneum (SC), through which transdermal delivery (TD) of chemicals takes place.1, 2 Transdermal delivery of drugs, which is routinely used for delivery of medications including hormones and analgesics, is an effective and widely used delivery route for many conditions. It involves the passage of therapeutic drugs through the skin and into the general circulation for systematic effects.3
Topical drug application produces a higher local concentration of the drug, where it preferentially distributes, persists and achieves sufficient levels of concentration to exert local therapeutic activity.4 Percutaneous penetration is also effective for the distribution of drugs to the surface of the skin, so it can affect the efficacy of topical formulation.5
At least 15 factors6 affect percutaneous penetration:
- Physiochemical properties of the penetrant
- Vehicle effects
- Dose, duration, surface area and frequency of exposure
- Sub-anatomical path
- Regional variation in flux
- Population variability in absorption
- Skin surface conditions
- Skin health and integrity
- Substantivity and binding to the SC, epidermis and dermis
- Distribution of chemical to epidermis, dermis and sebaceous glands
- Loss from skin surface and exfoliation
- Wash effect
- Metabolic and photochemical transformation in absorption into and
through skin - Excretion kinetics
- Method of determining absorption and toxicity
The act of rubbing or massaging a topical drug on the surface of skin is often perceived to provide additional benefits for percutaneous penetration. The present article surveys research regarding rubbing on percutaneous penetration and the mechanism that may trigger increased penetration of cosmetic ingredients and drugs.
Ongoing Research
Despite the common perception that rubbing may increase penetration, no convincing research has been done to prove this. Zheng et al. demonstrated that washing and rubbing an area of urea-treated skin can remove upwards of 60% of the dose, even as long as 6 hr after application.7 In reality, washing either human or animal skin after exposure to certain chemicals can leave a significant portion of the dose on or in the washed skin8 and increase penetration.9
Hasler-Nguyen and Fotopoulos10 demonstrated that application of 1.16% diclofenac-diethylamine gel, followed by 45 sec of rubbing, increased diclofenac flux through the skin up to five-fold after 8 hr. However, after 24 hr, the amount of diclofenac penetrated through the skin was identical in samples using the standard application and the application with rubbing. This indicates that rubbing may accelerate the speed of the drug’s transcutaneous delivery without altering skin integrity.
When compared with the standard application, rubbing the skin for 45 sec decreased its electrical resistance two-fold. The application of the 1.16% diclofenac-diethylamine gel with rubbing tended to result in higher accumulations in the stripped skin versus the superficial skin layers, when applied without rubbing (p = 0.2). These results suggest that rubbing may alter the superficial skin layer, resulting in a faster transient initial diffusion of topically applied diclofenac through the SC into the deeper layers of the dermis, to the target tissue.10
In an experiment with excised hairless rat skin, Sugibayashi et al.11 utilized a broadly used corticoid, triamcinolone acetonide (TA)—MW = 434.5; mp = 290°C; logP = 2.53; solubility in water at 25°C = 22.05 ± 0.05 μg/mL; solubility parameter = 9.45 (cal/cm3 )1/2—as a model lipophilic drug, and white petrolatum as an ointment base. Skin permeation of TA from 1.0% TA original ointment, with 30 sec of rubbing, was nine times higher than that after drug-free ointment-rubbing and application of 1.0% TA original ointment.
They concluded that ointment rubbing further increased the skin partition and permeation of TA because the rubbing causes a transient decrease in skin impedance and enhances delivery of the ointment base to the skin.10
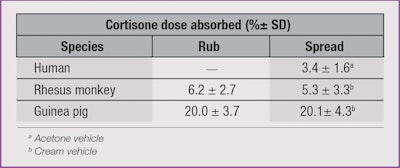
In another study, a rhesus monkey12 and a guinea pig13 were used to quantify percutaneous absorption. Cortisone, a low penetrant in both humans and rhesus monkeys, was used to determine the effect that rubbing a compound had on its absorption into the skin.
There was no significant difference (p > 0.05) in absorption of the cortisone between applications by rubbing versus non-rubbing in either of the animal models. But, as Table 1 shows, there was a three- to four-fold increase in the amount of absorption in the guinea pig compared to the rhesus monkey.14 This data suggested that the application by rubbing compared to simply spreading an ointment had no obvious effect on the dermal absorption. Treatment of patients with damaged skin might therefore be more effective if the less traumatic application of spreading were employed instead of rubbing.15
It is also possible that the lateral spreading phenomenon influences rubbing effects. Lateral spread—largely via skin surface furrows—adds approximately 30% greater surface exposure than the original application area.16 Also, clothing rubbing against the skin surface may rub away chemical residues or facilitate the exfoliation of skin cells, causing a subsequent loss of residues bound to them.7
Conclusion
The effect of rubbing on percutaneous penetration has long been questioned, yet it has been minimally investigated; and it has not been documented that rubbing universally increases or decreases percutaneous penetration of a chemical. The few experiments that have been performed to date have shown different results when it comes to the effects of rubbing on percutaneous absorption, largely based on the type of chemical used, species, skin thickness, site of application, etc.
Mechanisms in which absorption may increase are not well understood. More data regarding the more than 15 factors that affect percutaneous penetration is needed to understand rubbing’s effect. The question is hardly trivial: Should the clinician and beauty/health care industry advise rubbing to increase flux, and if so, for how long?
References:
- JA Bouwstra, PL Honeywell-Nguyena, GS Gooris and M Ponec, Structure of the skin barrier and its modulation by vesicular formulations, Prog Lipid Res 42 1–36 (2003)
- YC Kim, JH Park and MR Prausnitz, Microneedles for drug and vaccine delivery, Adv Drug Deliv Rev 64 1547–1568 (2012)
- A Loyd V Jr, Transdermals: The skin as part of a drug delivery system, Int J Pharm Compd 15 308–315 (2011)
- K Brune, Persistence of NSAIDs at effect sites and rapid disappearance from side-effect compartments contributes to tolerability, Curr Med Res Opin 23 2985–2995 (2007)
- FN Marzulli, Barriers to skin penetration, J Invest Dermatol 39 387–393 (1962)
- MA Ngo, M O’Malley and HI Maibach, Perspectives on percutaneous penetration of nanomaterials in Nanotechnology in Dermatology, A Nasir, A Friedman and S Wang, eds, Springer, New York ch 7 (2013) pp 63-86
- Y Zheng, A Vieille-Petit, S Chodoutaud and HI Maibach, Dislodgeable stratum corneum exfoliation: Role in percutaneous penetration? Cutan Ocul Toxicol 30 198–204 (2011)
- RP Zendzian, Pesticide residue on/in the washed skin and its potential contribution to dermal toxicity, J Appl Toxicol 23 121–136 (2003)
- RP Moody and HI Maibach, Skin decontamination: Importance of the wash-in effect, Food Chem Toxicol 44 1783–1788 (2006)
- N Hasler-Nguyen and G Fotopoulos, Effect of rubbing on the in vitro skin permeation of diclofenac-diethylamine 1.16% gel, BMC Res Notes 5 321–325 (2012)
- H Shii, H Todo and K Sugibayashi, Effect of sebum and ointment rubbing on the skin permeation of triamcinolone acetonide from white petrolatum ointment, Biol Pharm Bull 33 876–880 (2010)
- RC Wester and HI Maibach, Percutaneous absorption in the rhesus monkey compared to man, Toxicol Appl Pharmacol 32 394–398 (1975)
- KE Andersen, HI Maibach and MD Ango, The guinea pig: An animal model for human skin absorption of hydrocortisone, testosterone and benzoic acid? Br J Dermatol 102 447–453 (1980)
- HI Maibach and RC Wester, Percutaneous absorption: In vivo methods in humans and animals, J Am Coll Toxicol 8 803–813 (1989)
- J McMaster, HI Maibach, RC Wester and DAW Bucks, Does rubbing enhance in vivo dermal absorption? in Percutaneous Absorption, R Bronaugh and HI Maibach, eds, Marcel Dekker, New York (1985) pp 359–361
- A Vieille-Petit, F Pirot and HI Maibach, Metrics and clinical relevance of percutaneous penetration and lateral spreading, Skin Pharmacol Physical 28 57–64 (2015)