*Adapted from S Nafisi, M Schaffer Korting and HI Maibach, Perspectives on percutaneous penetration: Silica nanoparticles, Nanotoxicology (in press)
Skin is a unique barrier composed of highly organized and heterogeneous layers, including appendages such as hair follicles and sweat and sebaceous glands. It consists of three layers; from outside to inside: the epidermis, dermis and hypodermis. The outermost layer of the epidermis is the stratum corneum (SC), to which the main barrier function of skin is attributed. Skin keeps chemicals and pathogens from entering the body, protects against sunlight and retains the body’s water-rich internal organs from drying out by preventing water loss, especially in dry environments (see Figure 1).1
Recently, the use of nanoparticles has become widespread to increase the penetration of compounds into skin. Nanoparticles are commonly defined as 1 nm to 100 nm in size, and have at least one dimension on this nano-scale.2 Those that are 40 nm or smaller in diameter have been successful in penetrating skin.3 Silica nanoparticles (SNPs) in particular have attracted significant interest as cosmetic ingredients and for drug delivery, due to unique properties such as their hydrophilic surface; the versatility of silane chemistry for surface functionalization; the ease and relatively low cost of their large-scale synthesis; and their excellent biocompatibility.4 SNPs may offer revolutionary treatment for several skin diseases by controlling the sustained release of cosmetic ingredients or drugs to the skin, as well as enhancing the skin penetration of encapsulated ingredients. They are also candidates for skin cancer therapy, transcutaneous vaccination, and gene delivery. Further, they can act as carriers for drugs having low solubility, and they may improve drug safety, stability and performance.5, 6 Following is a review of their chemistry, along with potential applications, toxicology considerations and future perspectives on their use.
Silica Chemistry
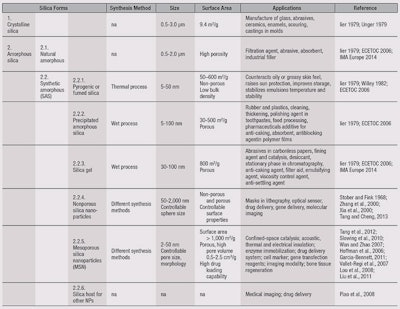
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula SiO2. It is the most common element found in nature. Silica can be divided into the main two classes: crystalline and amorphous. Crystalline micron-sized silica is a basic component of soil, sand, granite and many other minerals; amorphous silica is synthetic except for biogenic diatomaceous earth, which is composed of particles or structural units less than 1 μm in diameter. Synthetic amorphous (non-crystalline) silica particles are found in different forms7 (see Table 1), three of which have potential in topical skin care treatments and cosmetics. These include pyrogenic or fumed silica, non-porous silica nanoparticles and mesoporous silica nanoparticles.
Pyrogenic or fumed silica: Pyrogenic or fumed silica has an extremely low bulk density and a high surface area. It is a readily emulsifiable powder and has excellent dispersion properties and compatibility with other ingredients. It is used as a rheological additive in personal care products such as face powder, rouge, eye shadow, mascara and cosmetic pencils. It also enables high pigment levels in cosmetics by preventing re-agglomeration of the pigments. Another advantage of fumed silica is its low sensitivity to temperature, electrolytes and pH.
In formulations, it counteracts oily or greasy skin feel, and its particles create stable Pickering emulsions. In sun care emulsions, it can improve water resistance, and hydrophobic grades can produce a more homogeneous distribution of physical and chemical UV filters, raising the SPF. In lipsticks and nail polish, fumed silica improves the homogeneous distribution of pigments and prevents them from settling. It is also nontoxic and a non-irritant.8
Based on biomedical applications, silica NPs can be classified as mesoporous or nonporous. Furthermore, the size and shape of silica NPs can be controlled by tuning the composition and concentration of surfactants during synthesis.5, 6
Nonporous silica nanoparticles: Nonporous silica nanoparticles or monodisperse silica spheres were firstly prepared by the Stöber production process. Their biomedical applications for therapy and diagnosis are categorized based on the actives they deliver; e.g., small-molecule drugs, proteins or photo sensitizers; genes; or different contrast agents for molecular imaging. Nonporous SNPs can deliver their cargo through encapsulation or conjugation.5
Mesoporous silica nanoparticles: Mesoporous silica nanoparticles (MSNs) having a uniform pore size and long-range, ordered pore structure, were first reported in the early 1990s using different alkyl chain lengths of cationic surfactants such as dodecyltrimethylammonium or hexadecyltrimethylammonium ions as structure-directing agents (SDAs).
The abundant availability of various surfactants and a deep understanding of sol-gel chemistry have enabled the development of ordered MSNs with different structures, such as Mobil Crystalline Materials (MCM) and Santa Barbara Amorphous (SBA) types. SBA has thicker pore walls and wider pore sizes, i.e., 5 nm to 30 nm, than MCM. Until now, most research on drug delivery and cancer therapy applications of ordered MSNs are based on MCM-41 and SBA-15. These have been synthesized as ordered or hollow/rattle-type mesoporous silica structures, and they are widely used for the delivery of active payloads based on physical or chemical adsorption.6
Potential Applications and Ongoing Research
As noted, SNPs can be used as penetration enhancers, which help to promote drug diffusion through the SC to the viable epidermis and dermis. In one study, SNP-coated submicron o/w emulsions were shown to increase the stability and skin penetration of lipophilic agents, retinol and the fluorescent dye acridine orange 10-nonyl bromide. Lecithin and oleylamine addition, respectively, were used for the induction of negative and positive charges to the emulsion. Both formulations improved: retinol resistance to UV-induced degradation, control of release, and the penetration of both agents into excised porcine skin, as compared with a free agent control.9, 10
In a separate study of human volunteers, the addition of monodisperse silica spheres (~486 nm diameter) to an emulsion significantly increased quercetin penetration into the SC.11 In relation, complexes of quercetin with plain or octyl-functionalized MCM-41 increased emulsion stability without undermining the antioxidant efficacy of quercetin, thus suggesting an innovative use for such mesoporous composite materials in skin care.12 Moreover, in another study, the immobilization of rutin in the pores of an aminopropyl silica (NH2-MCM-41) stabilized it against UV degradation and enhanced its accumulation in porcine skin ex vivo while maintaining its antioxidant properties.13
In relation to photostability and safety, a different study trapped octyl methoxycinnamate within the pores of the mesoporous silicate MCM-41, pore openings were plugged, and the loaded nanoparticles were incorporated into a lipid-based cosmetic formulation. This provided broader photoprotection and a remarkable improvement in sunscreen photostability.14 In other work, the inclusion of a water-soluble analog of vitamin E, Trolox, in the MCM-41 matrix slowed its in vitro release and increased the photostability for the complexed agent, particularly in o/w emulsions. Importantly, the radical-scavenging activity of it also was maintained after immobilization.15 Finally, caffeine loading in SNPs resulted in the formulation of both core-shell and multilayered caffeine-silica structures, reducing and delaying the permeation of caffeine into pig skin, in comparison with a reference gel, independently from the amount of the tested formulation.16
The toxicity of SNPs depends strongly on their physicochemical properties, such as particle size, shape, porosity, chemical purity and solubility.5, 6 Surface area also plays a crucial role in toxicity due to interfacing with biological milieu.
Conclusions and Future Perspectives
SNPs are promising for the delivery of cosmetic ingredients and topical therapies, as they exhibit many advantages such as having highly controllable size, surface chemistry and shape. They are already finding application in the delivery of cosmetic ingredients, drugs, proteins and genes, and for molecular imaging. However, before SNPs can be used routinely, some major challenges must be overcome. These include the need to improve their ingredient/drug loading, spatial and temporal control of drug release, targeting to diseased sites, scalable manufacturing and long-term stability. Also, their biocompatibility and potential toxicity remain sine qua nons, requiring accurate penetration assessments for absorption, distribution, metabolism and excretion. Further details of the current knowledge and future opportunities for these assessments can be found elsewhere.17 Finally, organo-silica hybrid NPs, having the unique properties of SNPs but functionalities introduced by organic functional groups, could provide more sophisticated silica-based nanomedicines, with highly controllable drug loading and responsive drug release.
References
- http://ec.europa.eu/environment/chemicals/nanotech/faq/definition_en.htm (Accessed Mar 16, 2015)
- MR Prausnitz et al, Skin barrier and transdermal drug delivery, in Dermatology, J Bolognia, JL Jorizzo and JV Schaffer, eds, Saunders/Elsevier, Atlanta (1) ch 124 (2012) pp 2065-2073
- CA Poland, SAK Read, J Varet, G Carse, FM Christensen and SM Hankin, Dermal absorption of nanomaterials. The Danish Environmental Protection Agency, Environmental Project No. 1504, Denmark (2013)
- II Slowing, JL Vivero-Escoto, CW Wu and VSY Lin, Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers, Adv Drug Deliv Rev 60 1278-1288 (2008)
- L Tang and J Cheng, Nonporous silica nanoparticles for nanomedicine application, Nano Today 8 290-312 (2013)
- F Tang, L Li and D Chen, Mesoporous silica nanoparticles: Synthesis, biocompatibility and drug delivery, Adv Mater 24 1504-1534 (2012)
- RK Iier, The Chemistry of Silica, John Wiley and Sons, New York (1979)
- JP Hewitt, Formulating with nanotechnology in skin care opportunities and challenges, in Nanotechnology in Dermatology, A Nasir, A Friedman and S Wang, eds, Springer, New York (2012) pp 1-8
- N Ghouchi-Eskandar, S Simovic and CA Prestidge, Chemical stability and phase distribution of all-trans-retinol in nanoparticle-coated emulsions, Int J Pharm 376 186-194 (2009a)
- N Ghouchi-Eskandar, S Simovic and CA Prestidge, Nanoparticle coated emulsions as novel dermal delivery vehicles, Curr Drug Deliv 6 367-373 (2009b)
- S Scalia, E Franceschinis, D Bertelli and V Iannuccelli, Comparative evaluation of the effect of permeation enhancers, lipid nanoparticles and colloidal silica on in vivo human skin penetration of quercetin, Skin Pharmacol Physiol 26 57-67 (2013)
- G Berlier, L Gastaldi, E Ugazio, I Miletto, P Iliade and S Sapino, Stabilization of quercetin flavonoid in MCM-41 mesoporous silica: Positive effect of surface functionalization, J Colloid Interface Sci 393 109-118 (2013a)
- G Berlier et al, MCM-41 as a useful vector for rutin topical formulations: Synthesis, characterization and testing, Int J Pharm 457 177-186 (2013b)
- V Ambrogi, L Latterini, F Marmottini, C Pagano and M Ricci, Mesoporous silicate MCM-41 as a particulate carrier for octyl methoxycinnamate: Sunscreen release and photostability, J Pharm Sci 102 1468–1475 (2013)
- L Gastaldi, E Ugazio, S Sapino, P Iliade, I Miletto and G Berlier, Mesoporous silica as a carrier for topical application: The Trolox case study, Phys Chem Chem Phys 14 11318-11326 (2012)
- M Pilloni et al, Drug silica nanocomposite: Preparation, characterization and skin permeation studies, Pharm Dev Technol 18 626-633 (2013)
- S Nafisi, M Schaffer Korting and HI Maibach, Perspectives on percutaneous penetration: Silica nanoparticles, Nanotoxicology (in press)