The conditioning performance of a formulation reflects the distribution of cationic conditioning polymers and agents such as silicone oils along the hair fiber. Thus, to deposit conditioners as well as fragrances, actives and other materials onto hair surfaces from cleansing and conditioning formulas, various deposition technologies have been developed.1 In cleansing formulations, the most broadly used deposition technology is based on the complex coacervate formed from a combination of cationic polymers and anionic surfactants. Alternatively, pearlized wax networks provide deposition in cleansing formulations.
In order to target the deposition of materials, innovative methods are required to assess the delivery of conditioning agents to the fiber surface and damaged ends. One such spectroscopic technique, generally used with flat film substrates, is called attenuated total reflectance-infrared spectroscopy (ATR-IR). ATR-IR has effectively been used to monitor the migration of low molecular weight silicone polymers through a silicone matrix.2 In the present study, ATR-IR is used to map silicone polymer depositions along the length of hair fibers from conditioning shampoo formulations. In addition, a combination of confocal laser scanning (CLS), fluorescent microscopy and atomic force microscopy (AFM) are used to define the corresponding distributions of cationic polymers along hair fiber surfaces.
Materials and Methods
For the present study, three conditioning polymers were used: guar hydroxypropyltrimonium chloride (GHPTC)a, acrylamidopropyltrimonium chloride/acrylamide copolymer (APTAC-Acm)b, and a developmental polymer system combining the two—i.e., GHPTC (and) APTAC-Acmc. All three polymers had high molecular weights, ≥ 1 MM kdaltons. GHPTC and the developmental polymer system also exhibited a medium charge density, 0.5–1.1 meq/g, whereas APTAC-Acm had a high charge density, 1.5–3.0 meq/g.
GHPTC is well-known for delivering conditioning benefits to keratin substrates from cleansing formulations.3, 4 In addition, cationic acrylamide polymers are known for their conditioning performance. In the present work, APTAC-Acm was redesigned for further improved deposition performance. As a result, the developmental polymer system and APTAC-Acm were found to provide uniform conditioning along the hair fiber as well as targeted conditioning to the damaged ends of the hair fibers, as will be shown.
GHPTC, APTAC-Acm and the combined developmental system were formulated into three conditioning shampoos, each with a different polymer (see Formula 1). Virgin brown hair tresses were prewashed with 5% w/w sodium laureth sulfate with 2 ethylene oxide (SLES-2EO) used as a spacer. The SLES-2EO surfactant solution was used in the prewashing step to mimic residual adsorbed surfactant left on the hair from repeated shampoo treatments. The three silicone shampoos were applied to three separate sets of tresses to compare the performance of the polymers.
Deposition by ATR-IR
The total amount of silicone deposited on the tresses was quantified by extracting the silicone from the hair tress using a solvent and the subsequent measurement of the SiCH3 band near 1,261 cm-1 in the infrared spectrum of the evaporated solvent extract. In addition to total silicone deposition, the location of the silicone deposit was mapped using ATR-IR (see Figure 1). Attenuated total reflectance (ATR) often is used with a Fourier transform infrared (FTIR) spectrometer, referred to as FTIR/ATR. This technique is a surface analysis method that can vary in the depth of analyte studied, from ~0.3–5.0 µm, depending on factors including ATR crystal angle, the refractive index of the ATR crystal and the refractive index of the sample.
The technique used here measured the relative amount of silicone in the surface layer to a depth of 3 µm. This method relies on ratioing the height of a silicone band absorption to a band from the hair itself. The ratio removes any effects due to varying contact between the hair and the ATR crystal. This technique may be used to measure approximately 10–20 hair fibers in a 1 mm circular area. The limit of detection for the ATR-IR technique is 300 ppm silicone.
The FTIR/ATR technique has several advantages. This technique is relatively inexpensive, simple and requires no sample preparation. In addition, it requires only a small sampling area—0.8 mm2 to a depth of 3 µm at multiple points along the hair tress without extraction or destruction of the hair tress—and it provides results that correlate with absolute silicone levels.
Some techniques such as surface tension are sensitive to the presence of silicone and not to the amount present.5 Other techniques like X-ray photoelectron spectroscopy (ESCA) or time-of-flight secondary ion spectroscopy (TOF-SIMS) have better limits of detection but are often expensive, or they measure only a small spot size.5 Atomic force microscopy (AFM), while not a measure of silicone levels, gives the topography of the hair surface, which will correlate with the surface properties the silicone delivers. Thus, AFM and ATR results should be complementary.
For the present measurement, a bundle of fibers from each tress was positioned on the ATR accessoryd of the FTIR spectrometere equipped with a deuterated triglycine sulfate (DTGS) detector. Infrared spectra were collected at 12 different locations on the hair tress, starting from the top and working toward the bottom.
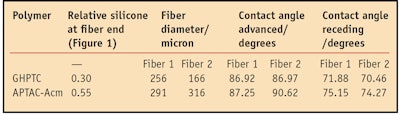
As shown in Figure 1, the developmental system enhanced silicone deposition by 50–140% relative to the individual polymers GHPTC and APTAC-Acm. It can also be seen that the developmental and APTAC-Acm polymers delivered more uniform silicone deposition across the length of the hair tress, including the more damaged tress ends. Increasing the silicone at the hair end should increase the hydrophobicity of the hair end. Therefore, the hydrophobicity of the ends of hair treated with the shampoo containing GHPTC was compared with hair treated with the shampoo containing APTAC-Acm using a single fiber tensiometerf to measure the wettability of the hair ends as they are immersed in water. The larger advancing and receding contact angles in Table 1 for hair treated with the shampoo containing APTAC-Acm versus GHPTC further support the enhanced hydrophobicity of the hair ends with the shampoo containing APTAC-Acm.
Deposition by CLS and AFM
The polymer coacervate deposited on the hair from the cationic polymer shampoo performs a dual role of conditioning and depositing ingredients (silicone) onto the hair fiber surface.6, 7 In silicone conditioning shampoos, water-insoluble silicone is present in the shampoo as an emulsified phase, and upon dilution of the shampoo with water, silicone and cationic polymer coacervate are deposited onto the hair. Microscopic and chemical methods, including scanning electron microscopy (SEM),3 ESCA8 and AFM4, 9 have been used to study the deposition of conditioning polymers on hair. Confocal laser scanning microscopy (CLSM) offers the advantage of viewing the hair fiber surface under ambient conditions,10 with the enhanced capabilities of optical sectioning and a fluorescent channel for viewing fluorescent polymer deposits and fluorescent probes applied to hair.7, 10–13 This fluorescence capability has been used on hair to create detailed graphical images of conditioning polymer deposits on human hair.13–15 To map the location of the deposits formed by the conditioning polymer coacervate and silicone oil, a combination of confocal laser scanning (CLS) fluorescent microscopy and AFM was used to view the hair fiber surface under ambient conditions of temperature and pressure. In other studies using CLS fluorescent microscopy, the fluorescent probe has been covalently attached to the cationic polymer.11, 12 In the current and previous studies,7, 15 the anionic fluorescent probe was bound to the cationic polymer deposits on the hair fibers through only electrostatic and hydrophobic interactions. This CLS fluorescence imaging was conducted by shampooing the hair samples and treating them with the fluorescent probe in a sodium acetate buffer at pH 7g, h. The anionic fluorescent probe used in this study was shown to bind only to hair that had been treated with shampoo containing a cationic polymer. Previous work showed that there was no binding of the dye on hair treated with a control shampoo containing no cationic polymer.
The anionic charge of the fluorescent probe thus leads to attraction of the probe to the cationic polymer coacervates on the hair. As shown in Figure 2, the fluorescent dyej intensity is strongest at the cuticle edges on virgin brown hair, especially for hair treated with shampoos containing GHPTC and the developmental system. The dye used possesses galactosyl residue specificity and strong affinity to cationically charged deposits, making it especially useful for locating galactose-containing residues like GHPTC. Delivering the cationic polymer coacervate to the cuticle edge allows it to smooth the cuticle surface. The micrographs in Figure 2 demonstrate that GHPTC, APTAC-Acm and the developmental system delivered the cationic polymer coacervate to the cuticle edge.
To further characterize the distribution and topography of the coacervate on the hair, AFM of the top and end of the hair was carried out on virgin brown hair using a tapping mode, a method of imaging where the AFM probe tip taps the fiber surface, compared with the contact mode where the probe tip drags across the surface with constant contact. This is conducted in ambient air rather than in liquid or in another environmentk.
The atomic force micrographs in Figure 3, Figure 4 and Figure 5 confirm the full coverage provided by the three conditioning polymers to the hair cuticle at both the top and end of the virgin brown hair tresses. These micrographs showed that the developmental system and APTAC-Acm consistently provided more deposition to the end of the hair, better coating the hair ends. This result is consistent with the silicone deposition results in Figure 1 and the tensiometer results in Table 1, further substantiating the link between coacervate deposition and silicone deposition on hair.
Conclusions
Cationic polymers such as GHPTC, APTAC-Acm and the combination of the two enable uniform deposition of conditioning oils along the hair fiber, as well as targeted conditioning at the damaged ends. These polymers were also shown to effectively deposit silicone onto the hair fiber surface, with different deposition profiles and efficiencies of silicone deposition from shampoo formulations. By improving deposition, these polymers may reduce the amount of conditioning oils and active ingredients required in formulas to achieve the specified performance.
ATR-IR proved useful as an efficient screening method for mapping the distribution of silicone along the length of the hair fiber. In addition, the silicone deposition profile and type and location of the coacervate deposit on the hair cuticle could be related to the conditioned state of the hair. AFM and CLS fluorescent microscopy also proved to be complementary techniques to ATR-IR for mapping the coacervate location and structure on the hair cuticle. Techniques such as ATR-IR, AFM and CLS fluorescent microscopy give the formulator tools to differentiate the performance of shampoos or conditioners, as well as substantiate claims of enhanced conditioning performance.
Acknowledgements: The author wishes to thank Delaware Biotechnology Institute’s Kirk Czymmek, PhD, for his expertise in the performance of the confocal microscopy experiments, and Liz Adams, PhD, for assistance in the AFM experiments.
References
- RY Lochhead and LR Huisinga, Advances in polymers for hair conditioning shampoos, ch 17 in Hair Care: From Physiology to Formulation, Angela C. Kozlowski, ed, Allured Business Media, Carol Stream, IL 123–136 (2008)
- H Homma et al, Diffusion of low molecular weight siloxane, from bulk to surface, IEEE Transactions on Dielectrics & Electrical Insulation 6 3 370–375 (1999)
- SB Ruetsch, YK Kamath and HD Weigmann, The role of cationic conditioning compounds in reinforcement of the cuticula, J Cos Sci 54 63–83 (2003)
- P Hossel, R Dieing, R Norenberg, A Fau and R Sander, Conditioning polymers in today’s shampoo formulations—efficacy, mechanism and test methods, Intl J Cos Sci 22 1–10 (2000)
- A De Smedt, I Van Reeth, S Marchioretto, DA Glover and J Naud, Measurement of silicone deposited on hair by various analytical methods, poster presented at the 19th IFSCC Congress, Sydney (1996)
- YK Kamath, CJ Dansizer and HD Weigmann, Surface wettability of human hair. III. Role of surfactants in the surface deposition of cationic polymers, J Applied Polymer Sci 30 3 937–953 (1985)
- PE Erazo-Majewicz, SC Su, Cationic conditioning—polymer deposits on hair, J Cosm Sci 55 125–127 (2004)
- ED Goddard and WC Harris, An ESCA study of the substantivity of conditioning polymers on hair substrates, J Soc Cos Chem 38 233–246 (1987)
- VF Monteiro, AS Pinheiro, ER Leite, JAM Agnelli, MA Pereira-Da-Silva and E Longo, UV radiation: Aggressive agent to the hair-AFM, a new methodology of evaluation, J Cos Sci 54 271–281 (2003)
- C Hadjur, G Daty, G Madry and P Corcuff, Cosmetic assessment of the human hair by confocal microscopy, Scanning, 24 2 59–64, (2002)
- STA Regismond, YM Heng, ED Goddard, FM Winnick, Fluorescence Microscopy Study of the Sorption of Cationic Polymers on Hair, ch 19 in Polysaccharide Applications, Langmuir 276–286 (1999)
- V Gruber, FM Winnick, A Lapierre, ND Khaloo, N Joshi and PN Konish, Examining cationic polysaccharide deposition onto keratin surfaces through biopolymer fluorescent labeling, J Cos Sci 52(3/4) 119–129 (2001)
- HD Weigmann, YK Kamath, SB Ruetsch, P Busch, H Tesmann, Characterization of Surface Deposits on Human Hair Fibers, J Soc Cos Chem 41 379–390 (1990)
- ML Tate, YK Kamath, SB Ruetsch and HD Weigmann, Quantification and prevention of hair damage, J Soc Cos Chem 44 347–371 (1993)
- S Jindal, A 3-Dimensional View of Cationic Deposition on Hair, presentation at the HBA Global 2002 Product Development Conference (2002)