Known for creatively innovative products with high-performance ingredients, unique packaging and fascinating textures, K-beauty inspires new trends that greatly revolutionize the cosmetics industry. In the pursuit of achieving skin perfection, K-beauty brands have placed innovation at the heart of their global strategies. Within this innovation, bio-harmonious actives are highly sought to provide visible and long-term results by acting in phase with the cutaneous metabolism, and more particularly by supporting the epidermis integrity.
In relation, a next-generation peptide was recently demonstrated in vitro and in vivo to induce the production of endogenic α-crystallin, a molecular chaperone that plays a crucial role in skin proteostasis. The present article shows how targeting this protein can improve skin cell resistance to external aggressions, leading to desired effects for beauty care.
The Glass Skin Trend
In Asia, the consumption of beauty products is highly substantial, and consumers want smooth and glowing skin without imperfections. They adopt different beauty routines by buying several products at once to attain the holy grail: glass skin, which refers to translucent, smooth and dewy skin with a strikingly glowing complexion.
The glass skin trend echoes that of the recent strobing technique, which refers to the extensive use of highlighters to illuminate and sculpt the face using light reflection. Containing illuminating pigments, these makeup highlighter products attract light and create extra luminosity for iridescent, glowing skin.
Highlighting products are applied to the cushioned parts of the face: the eyebrow arch, center of the nose bridge, lip’s bow and cheekbones, to make skin appear radiant. The goal is to achieve a fresh and healthy glow, as this finish evokes youthfulness and ultra-moisturized skin.
This two-in-one global trend has challenged the cosmetic industry to innovate, as highlighter products have recorded increasing success; according to Mintel, they accounted for more than 94% of new products launched in the makeup category between 2015 and 2017.
Facing the consumer demand for naturally derived products with high performance, the newly developed peptide acts as a bio-highlighter and makeup alternative for a natural, glass skin appearance. By exerting a bio-harmonical mechanism of protection, it balances the epidermis renewal process using epigenetic and molecular regulation. It also preserves skin proteostasis through the induction of the α-crystallin molecular chaperone.
Crystallins
Crystallins (see Figure 1) are a class of water-soluble structural proteins found most abundantly in the eye lens tissue and cornea, though they also are present in other nonlenticular tissues.1 They have been characterized as small heat shock proteins (HSPs), not only from their striking sequence similarity with other small HSPs, but also because they accumulate under stress conditions; aggregate into supramolecular structures with the same sedimentation coefficient as heat shock granules; and are found associated with the nucleus under heat shock conditions.1
As chaperone proteins, after being activated by phosphorylation, crystallins bind and prevent misfolded target proteins from aggregating and precipitating, keeping them soluble and in a folding-competent state.2, 3 For example, they ensure the transparency of the eye structure by preventing protein aggregation and protecting related enzymes against heat-induced inactivation.4 They also prevent the accumulation of cellular waste.
In mammals, three groups of eye lens proteins can be distinguished: α, β and γ. They differ from each other in size; charge; immunological and physicochemical properties; amino acid composition of their subunits; and degree of aggregation. They are associated in the cytoplasm of lens fiber cells, thus forming a gel that shows a gradient in protein concentrations ranging from 200 mg/mL to 300 mg/mL at the edges, and up to 3 times more at the center of the eye lens.
Since crystallins are not renewed, they are necessarily very stable. They have a long lifespan and are highly resilient against light rays, heat and free radicals.5 These assets justified the current interest in this group of proteins for potential application. Furthermore, a study conducted by P.L. Carmichael6 showed that crystallins protect other proteins such as bovine serum albumin (BSA), hemoglobin, ovalbumin and lysozyme from degradation. Thus, crystallin proteins are not only stable, but are also capable of protecting and stabilizing a large array of proteins.
α-Crystallin and Skin
As noted, while α-crystallin, also called HspB5, is abundant in the transparent corneal epithelium, it is also present in other epithelia, such as the epidermis. Here, it occurs in the form of aggregates of about 800 kDa, and is built from only two types of subunits: α-A and α-B, each around 20 kDa. The α-A subunit is made up of 173 amino acids and has an acidic nature, whereas the α-B subunit contains 175 amino acids and is basic in nature. These aggregates are in the form of spherical particles ranging from 15-20 nm in diameter, and contain varying percentages of α-A and α-B.
These small proteins are related to the family of heat shock proteins (HSPs) that, once induced, have been shown to increase the cell’s resistance to stress, protecting epithelial cells by restoring their mitochondrial functions, and increasing their resistance to oxidative stress (see Figure 2). In addition, treatment with α-crystallin has been shown to reduce the production of oxygenated radical species due to inflammation.7
α-Crystallin has numerous cellular functions—e.g., cytoskeletal, cell growth and adhesion, signaling mechanisms, protein transport, apoptosis, proteolysis and transcription—all of which result from its interaction with a large spectrum of protein partners.8 It is found both in cells and their immediate environment, making them readily accessible upon exposure to UV, during inflammation and in response to other oxidative-type attacks.9 The antioxidant effect of α-crystallin was found to depend on reduced gluthatione under the control of glucose-6 phosphate dehydrogenase.10 α-Crystallin expression correlates with decreased levels of reactive oxygen species (ROS) and nitric oxide (NO•). Consequently, in cells exposed to oxidative challenges, α-crystallin expression reduces lipid peroxidation, protein oxidation and F-actin architecture disruption. It also upholds the mitochondrial membrane potential level; a phenomenon which provides the stressed cells with abundant ATP production that favors the activity of chaperones.11
Hydration is another key attribute of α-crystallin. NMR spin-echo studies measuring relaxation times from the protons of water adsorbed on proteins demonstrate the high capacity of α-crystallin to bind water molecules, compared with the capacities of BSA and collagen. These superhydration properties occur through tight binding to water and are important for both the physiological role of α-crystallin as a molecular chaperone, and to prevent the aggregation and denaturation of other proteins in skin tissues under low-humidity, dry conditions.4
Taken together, in skin, α-crystallin ensures cell proteostasis and prevents the aggregation and denaturation of proteins, which can impair cell functioning as well as skin’s refractive index. By analogy, considering its role in the eye lens, α-crystallin impedes the opacification of cutaneous structures.
Hydration is another important attribute of α-crystallin, which occurs through its tight bonding to water. This binding is also key to its role as a molecular chaperone.
Peptide Research
A peptide sequence composed of four amino acids—lysine, threonine, phenylalanine and an additional lysine—was identifieda active for various targets including epidermal maturation markers and inflammation markets in the routine screening tests. This tetrameric sequence was then attached to a palmitic acid to produce pal-KTFK. The resulting bio-highlighter peptide developed was tested as follow for its ability to induce α-crystallin proteins; ultimately to make skin look clearer, smoother and more glowing. Furthermore, adequate and specific vectorization allowed for the optimization of the peptide’s bioavailability in the epidermis, as well as its efficient deliverability at the skin surface with a gradual release to promote long-term activity.
Proteomics Protocol
Proteomics studies by liquid chromatography coupled tandem mass spectrometry (LC-MS/MS) were conducted to evaluate the induction of α-crystallin by pal-KTFK. This powerful technique makes it possible to evaluate several hundred proteins present in the same sample simultaneously, and to compare the results with control cases.
Normal human keratinocyte (NHK) cultures are brought into contact with the active ingredient in a suitable culture medium. Then, a cell homogenate is prepared with a suitable buffer, the proteins are recovered and cut out specifically using a protease, which creates longer or shorter peptide fragments. LC-MS/MS analysis separates the peptides by liquid chromatographyb according to their hydrophobicity, ionizing them and then separating them in a detectorc that classifies them according to their mass and charge (m/z).
Finally, softwared analyzes the mass and charge of each element to deduce the presence and quantity of the preexisting proteins above the detection threshold. This data can then be processed through bioinformatics/biostatistical analysis to identify the biological processes and metabolic pathways modulated by an active ingredient.
LC-MS/MS Test and Results
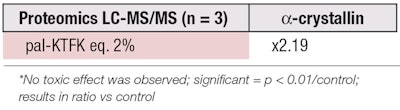
Measurements of α-crystallin protein induction were carried out according to the described protocol. The test peptide was brought into contact for seven days with confluent NHK (n = 3). The culture medium was changed every three days to provide cells with a constant supply of essential elements for both their survival and production. After seven days, cells were prepared to extract the proteins and analyze them by LC-MS/MS.
In order to carry out LC-MS/MS analyses on comparable protein quantities, the protein concentration was previously estimated on ground products by BCA assays. A study of the variances and a Student’s t-test for nonpaired series were performed to judge the significance of the results. Interestingly, the expression of α-crystallin in treated NHK appeared much higher (2.19×) than that of the control (see Table 1).
Skin Equivalent Tests and Results
This result led to additional tests in skin equivalents produced according to the Sederma protocol. On the eve of the emergence phase, skin composed of a single layer of confluent keratinocytes at this stage received the pal-KTFK ingredient. Twenty-four hours later, skin equivalents were placed at the air-liquid interface for 18 days so that the epidermis and stratum corneum could form.
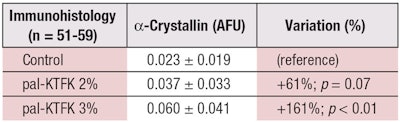
Eighteen days later, skin equivalents were frozen in liquid nitrogen and 7 μm cryo-sections were taken; α-crystallin was labelled by immunohistology. For each microscope image, fluorescence was quantified and reported to the analyzed surface (n = 51-59); again, a Student’s t-test was performed to validate significance.
Table 2 and Figure 3 confirm the first in vitro results. α-Crystallin was clearly stimulated in skin equivalents in the presence of pal-KTFK. Versus the control, at 2% pal-KTFK, α-crystallin content increased by 61% (p = 0.07) and at 3%, it increased by 161% (p < 0.01). Apparently, this is the first time α-crystallin has been identified in epidermis cells.
Clinical Study I: Protocol and Results
Following in vitro success, many clinical studies were carried out to explore the visible results in skin provided by the induction of α-crystallin by pal-KTFK. One study was performed in a panel of 29 women (mean age = 41 years; 30-50 year range), presenting with a dull complexion and irregular, rough skin texture. Volunteers observed a washout period for one month using a provided moisturizer and excluding the use of any cosmetics such as scrubs or masks, as well as any significant sun exposure.
In the pursuit of achieving skin perfection, K-beauty brands have placed innovation at the heart of their global strategies. Within this innovation, bio-harmonious actives are highly sought.
The single blind study was conducted on the face and forearms. Volunteers applied a cream containing 3% pal-KTFK and a placebo cream in a contralateral manner twice daily for six weeks. Standardized photographs were made at T0 and T6 weeks with a photographic bench using a high-definition digital camera, specific lighting and a system of restraint for volunteers. Face position, settings of photographs and lighting were standardized and controlled for reproduction throughout the study. The visible reduction of imperfections is illustrated by pictures before and after the six-week application of pal-KTFK in a cream, as exemplified in Figure 4. The collected photographs were examined by a panel of five expert judges trained for years on this type of evaluation. For each volunteer, the experts visualized the photographs before and after treatment with the test peptide and gave their opinion on the following statement: The complexion is more even and smoother, with less imperfections. The results of these assessments are shown in Figure 5.
The application of pal-KTFK led to a visible and significant improvement in the skin complexion, according to favorable opinions of 54% and unfavorable opinions of only 3%. On the contrary, use of the placebo registered 36% positive and 8% negative opinions. Treatment with the placebo was therefore clearly and significantly different (p < 0.05).
Clinical Study II: Protocol and Results
A second study was carried out in a panel of 26 women (mean age = 36 years; 22-44 year range) presenting with a dull complexion—according to descriptors of color, luminosity, clarity and transparency, i.e., the CLCT methode—and having a skin phototype of I to III. This study was conducted on subjects’ faces. Volunteers applied a cream containing 3% of the pal-KTFK peptide twice daily for six weeks.
The CLCT method is based on an expert’s visual analysis of seven representative descriptors of the complexion.12 For all measurements, the subject sits between two “daylight” lamps and wears a black cape and cap to avoid any external interference. Judges then evaluated the subject for the color variable, C, based on the skin’s intensity of four colors given on a chart: pink-red, olive, beige and soft pink (see Figure 6). The three remaining descriptors—i.e., luminosity, L; clarity, C; and transparency, T—were graded based on an analog scale ranging from 0-100.
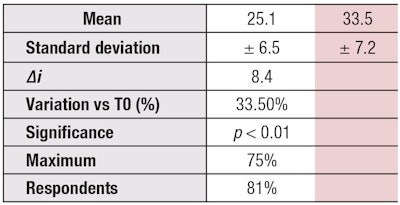
An index, i, is then calculated from the seven descriptors; changes to this index over the course of the six-week study are calculated as follows: αi = i(T6) – i(T0). If the result is greater than 5, it could be concluded that the test product effectively and significantly improved complexion radiance. The application of a cream formulated with 3% pal-KTFK altered the CLCT index by an impressive increase of 8.4 units (+33.5%; p < 0.01 vs T0; see Table 3), which indicates the active effectively and visibly improved the measured aspects of complexion radiance. The detailed variations in descriptors are shown in Figure 7.
Conclusion
In conclusion, and in light of what has been presented here, α-crystallin is a target of interest for effectively protecting the skin against the effects of various external harmful factors. It answers the search of cosmetic brands for innovative activities—especially in order to elevate the K-beauty glass skin trend.
Author’s note: Many in vitro and in vivo tests have been performed with the pal-KTFK ingredient. All results are not presented here, and are available upon request.
References
- Klemenz, R., Frohli, E., Steiger, R., Schafer, R. & Aoyama, A. (1991). Alpha-crystallin is a small heat shock protein. Prot Natl Acad Sci USA, 88, 3652-3656.
- Ecroyd, H., et al. (2007). Mimicking phosphorylation of αB-crystallin affects its chaperone activity. Biochemical Journal, 401, 129-141.
- Delbecq, S.P., Jehle, S. & Klevit, R. (2012). Binding determinants of the small heat shock protein, aB-crystallin: Recognition of the 'IxI' motif. EMBO J., 31, 4587-4594.
- Babizhayev, M.A., Nikolayev, G.M., Nikolayeva, J.G. & Yegorov, Y.E. (2011). A survey and analysis of the role of molecular chaperone proteins and imidazole-containing dipeptide-based compounds as molecular escorts into the skin during stress, injury, water structuring and other types of cutaneous pathophysiology. International Journal of Cosmetic Science, 33, 1-16.
- Wistow, G.J. & Piatigorsky, J. (1988). Lens crystallins—The evolution and expression of proteins for a highly specialized tissue. Annual Review of Biochemistry, 57, 479-504.
- Carmichael, P.L., Zimmermann, B. & Hipkiss, A. (1989). Differential susceptibility of crystallin and other proteins to free-radical-induced peptide bond cleavage. Biochemical Society Transactions, 17, 494-495.
- Zhu, Z., Li, R., Stricker, R. & Reiser, G. (2015). Extracellular α-crystallin protects astrocytes from cell death through activation of MAPK, PI3K/Akt signaling pathway and blockade of ROS release from mitochondria. Brain Res., 1620, 17-28.
- Arrigo, A.P. & Gibert, B. (2013). Protein interactomes of three stress inducible small heat shock proteins: HspB1, HspB5 and HspB8. Int J Hyperthermia, 29, 409-22.
- Mulders, J.W.M., Stokkermans, J., Leunissen, J. A., Benedetti, E.L., Bloemendal, H. & de Jong, W.W. (1985). Interaction of α-crystallin with lens plasma membranes: Affinity for MP26. European Journal of Biochemistry, 152, 721-728.
- Sreekumar, P.G., et al. (2012). Mechanism of RPE cell death in α-crystallin deficient mice: A novel and critical role for MRP1-mediated GSH efflux. PLoS One, 7, 1-13.
- Arrigo, A.P., et al. (2007). Hsp27 (HspB1) and alphaB-crystallin (HspB5) as therapeutic targets. FEBS Lett., 581, 3665-3674.
- Musnier, C., Piquemal, P., Beau, P. & Pittet, J.C. (2004). Visual evaluation in vivo of “complexion radiance” using the C.L.B.T. sensory methodology. Skin Res. Technol., 10, 50-56.