Many people utilize non-UV tanning methods to meet fashion demands without exposing themselves to UV radiation. This article reviews the health benefits and possible drawbacks of dihydroxyacetone, the main active ingredient in self-tanning products.
The desire to achieve an attractive and fashionable tan has long been common among adolescents and young adults, especially those with lighter skin tones. Today, a segment of individuals—primarily those living in western countries—darken their skin by exposure to UV radiation either from the sun or indoor tanning equipment; i.e., tanning beds, booths and sun lamps.1
According to extensive scientific studies, however, prolonged exposure to UV radiation is inherently carcinogenic and damages the DNA. Therefore, it is not surprising that direct UV tanning can significantly increase the risk of melanoma and possibly non-melanoma skin cancers, along with premature skin aging.2, 3 And according to the U.S. Cancer Statistics Working Group, melanoma is the deadliest type of skin cancer, even though it is less prevalent.4, 5
Meeting the Tan Demand
Although the detrimental effects of sunlight and other sources of UV radiation on the skin are well-documented, a sun-tanned appearance remains a symbol of beauty and fashion for many individuals. As such, the extreme measures people were taking to look tan encouraged cosmetic companies to produce an alternative solution: the self-tanner, which gives the skin a tanned appearance without UV radiation.
In the early 1960s, the first self-tanning product—named Quick Tan—entered the beauty market. Unfortunately, its overall effect on skin was rather orange and not natural-looking. Over the last two decades, though, sunless-tanning products have experienced considerable improvements in their formulations to achieve more natural-looking tanned skin.
Various self-tanners such as lotions, creams and sprays are now available, and these are not only safer than UV radiation, but also more convenient to use.6 Such products produce a temporary tan via a chemical reaction in the stratum corneum, and last for only the life cycle of the skin cells, due to shedding and exfoliation.7
DHA in Action
Dihydroxyacetone (DHA) acts as the main substance in self-tanning lotions, and is a safe browning agent approved by the U.S. Food and Drug Administration (FDA).8 The usual concentration of DHA in these products ranges from 5–15% and depends on coloration levels from light to dark; however, lower concentrations (3–5%) are preferred.7
DHA (C3H6O3) is a white, non-toxic crystalline powder derived from plant sources such as sugar beets and sugarcane, or even produced by bacteria such as Acetobacter suboxydans.8 DHA is stable between pH 4 and 6, and reacts with nitrogen and oxygen. After application to the skin, this active agent reacts chemically with the free amino acids in the stratum corneum. As a result of this non-UV Maillard reaction, melanoidin pigments gradually form and provide the appearance of a tan.9 This chemical staining is apparent within approximately one hour; however, it can take more time to complete.
The extreme measures people were taking to look tan encouraged cosmetic companies to produce an alternative solution: the self-tanner.
Since shedding of the stratum corneum cells is a continuous natural phenomenon, this color dissipates in 5–7 days after application. Therefore, activities that accelerate the rate of cell shedding—such as tape-stripping, prolonged water submersion or heavy sweating—can affect color persistence. Some individuals may apply the self-tanner several times to achieve the desired tan appearance, and repeat the application every 2–4 days for a more prolonged effect.8 The effect may last longer on some thin and sensitive body sites, such as the face, that are rubbed less often. In relation, it is recommended to shave, wax and shower before the initial application to produce a more evenly-tanned effect.10
Formula Improvements
The original color of skin to be self-tanned plays a pivotal role in DHA's ability to achieve the desired color. For example, self-tanning products work best in individuals with Fitzpatrick skin types II and III, while on pale or ruddy skin, they may appear somewhat yellow. In response, various additives to the basic DHA formulations can overcome undesired results.
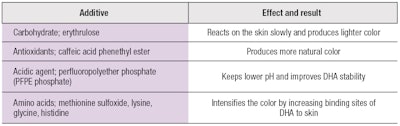
For example, antioxidants can significantly enhance color quality. Also, caffeic acid phenethyl ester (CAPE), a component of propolis from honeybee hives,11, 12 has traditionally been used to treat ailments due to its antibacterial, anti-inflammatory, analgesic and anti-carcinogenic benefits.13 Combinations of CAPE with DHA produce a more natural-looking tan by decreasing yellow and increasing red tones in the self-tanner.
Another substance used is erythrulose. Despite its similarity to DHA, erythrulose reacts on the skin surface more slowly and produces a lighter and shorter-lasting tan. However, the FDA has not yet approved erythrulose as a self-tanning agent.14
Amino acids can be used with DHA to produce a better brown color. This is achieved by increasing DHA's binding sites to skin. In addition, acidic agents can be used to help keep the skin pH low; near ~5.15, 16 Table 1 shows some additives combined with DHA in self-tanning products.
Possible Adverse Effects
Although self-tanners are considered safe and effective for tanning without UV exposure, they pose some risks. Keratinocyte treatment with DHA has shown it penetrates cells underlying the stratum corneum in a time- and dose-dependent manner. This reaction could potentially cause DNA damage and apoptosis. Tests to detect DHA in the bloodstream immediately after topical application might prove this penetration.17
Another possible deleterious side effect of DHA self-tanners is inadequate UV protection, since they make the skin more susceptible to UV radiation for the first 24 hr after product application. This is due to the Amadori products formed during the Maillard reaction, which can increase the number of free radicals in the skin after sun exposure.
In fact, one study investigated the effect of DHA-containing self-tanners on cell survival and proliferation in a human keratinocyte cell line. These authors proposed a high percentage of free radicals would be generated after using DHA. To overcome the problem, self-tanners could be formulated with added sunscreens; however, the duration of the UV protection would be more short-lived. Adding antioxidants also could minimize oxidative stress and reduce the number of generated free radicals.13
Conclusion
Taken together, the advantages of self-tanners outweigh the risks of UV tanning. And through greater awareness of the dangers of UV tanning, along with improvements in self-tanning product formulas, the industry is seeing a remarkable increase in the use of sunless tanners as a reasonable option for a tanned appearance. Advances in understanding the physical chemistry and chemical bonding to stratum corneum protein, as well as in vitro models to quantify the biology, may also lead to formulations demonstrating greater efficiency.18
References
- M Watson, DM Holman, KA Fox, GP Guy Jr, AB Seidenberg, BP Sampson, C Sinclair, D Lazovich, Preventing skin cancer through reduction of indoor tanning,AJPM 44 682–689 (2013)
- FE Ghissassi, R Baan, K Straif, Y Grosse, B Secretan, V Bouvard, L Benbrahim-Tallaa, N Guha, C Freeman, L Galichet, V Cogliano, A review of human carcinogens-Part D: Radiation,The Lancet,Oncology 10 751–752 (2009)
- S Gandini, F Sera, MS Cattaruzza, P Pasquini, O Picconi, P Boyle, CF Melchi, Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure,Eur J Cancer 41 45–60 (2005)
- U.S. Cancer Statistics Working Group, United States Cancer Statistics: 1999–2012 Incidence and Mortality Web-based Report
- MK Tripp, M Watson, SJ Balk, SM Swetter, JE Gershenwald, State of the science on prevention and screening to reduce melanoma incidence and mortality: The time is now,CA Cancer J Clin 66 460–480 (2016)
- CL Paul, J Bryant, H Turon, I Brozek, N Noble, A Zucca, A narrative review of the potential for self-tanning products to substitute for solaria use among people seeking a tanned appearance,Photodermatol Photoimmunol Photomed 30 160–166 (2014)
- CG Burkhart, CN Burkhart, Dihydroxyacetone and methods to improve its performance as artificial tanner,Open Dermatol J 3 42–43 (2009)
- SB Levy, Dihydroxyacetone-containing self-tanning lotions, JAm Acad Dermatol 27 989–993 (1992)
- GP Ellis, The Maillard reaction,Adv Carbohydr Chem Biochem 14 63–135 (1959)
- HI Maibach, AM Kligman, Dihydroxyacetone: A suntan stimulating agent, Arch Dermatol 82 505–507 (1960)
- N Muizzuddin, KD Marenus, DH Maes, Tonality of suntan vs sunless tanning with dihydroxyacetone,Skin Res Technol 6 199–204 (2000)
- GF Sud’ina, OK Mirzoeva, MA Pushkareva, GA Korshunova, NV Sumbatyan, SD Varfolomeev, Caffeic acid phenethyl ester as a lipoxygenase inhibitor with antioxidant properties,FEBS Lett 329 21–24 (1993)
- K Jung, M Seifert, T Herrling, The fatal effect of self-tanning agents during UV irradiation,SÖFW J 134 3 (2008)
- G Pantini, R Ingoglia, F Brunetta, A Brunetta, Sunless tanning products containing dihydroxyacetone in combination with a perfluoropolyether phosphate,Int J Cosm Sci 29 201–209 (2007)
- AB Petersen, HC Wulf, R Gniadecki, B Gajkowskab, Dihydroxyacetone, the active browning ingredient in sunless tanning lotions, induces DNA damage, cell-cycle block and apoptosis in cultured HaCaT keratinocytes,Mutat Res 560 173–186 (2004)
- BC Nguyen, IE Kochevar, Factors influencing sunless tanning with dihydroxyacetone,Br J Dermatol 149 332–340 (2003)
- L Goldman, DJ Blaney, Dihydroxyacetone,Recent Clin Invest Stud 85 86–90 (1962)
- X Hui, HI Maibach, Biology of chemical binding to stratum corneum protein (manuscript in preparation)