Since ancient times, changing one’s hair color has been a fascination and status symbol.1 To many individuals today, it is a practical necessity with life expectancies inching upward—“50 is the new 40.” Influenced by the youth-oriented workplace and Madison Avenue, consumers are encouraged to eliminate the telltale signs of aging and to keep a youthful look as long as possible.
The promise of extending one’s youthful appearance is driving the personal care industry today. Popular antiaging formulas typically require several weeks or months to change the appearance of skin, but hair color and style, also crucial components of a person’s image, can be altered in the matter of a few hours. Most women and an increasing number of men find themselves hiding their gray, adding highlights or low-lights, and lightening or darkening a few shades, in attempt to make a visual statement about themselves through their hair.
Modern chemistry provides the hair color industry with a wide spectrum of possible colors. Hair coloring treatments range from simple and convenient temporary—i.e., diffusion/substantivity-controlled colorants—to the full commitment of a permanent or oxidative coloring process. Permanent hair color, however, is far from “permanent.” Factors such as washing, UV exposure and heat styling are generally known causes of fading.
Although some fading is actually desirable to blur the line of demarcation or regrowth between natural hair color and applied color, there is an ever-growing demand from consumers for hair care products that retain color, especially considering the costs of salon visits and the time it requires for permanent hair coloring.
Formulators of hair coloring products are challenged to maintain the original vibrancy and intensity of the first day’s coloring despite the harsh regimens hair is exposed to on a daily basis. Of the many causes of fading in artificial hair color, the primary one unfortunately is hair washing.2 Each time that color-treated hair is exposed to water and shampoo, more and more colorant is lost.
To address this challenge, the present paper addresses a new olefin-graft polymer technology engineered to improve the permanency of oxidative hair colorant in human hair exposed to repeated washings.
Olefin-graft Polymer Technology
Acrylic-based polymers have been manufactured with a variety of functional groups and side chains to offer diverse features and benefits to the formulator. Polyolefins, from paraffin waxes to polyethylene derivatives, have similarly provided a variety of benefits and the grafting of these two diverse compounds for the personal care market offers some interesting formulating properties. Polymers of this type have been used in the industrial market for nearly three decades to improve the properties of a variety of surfaces. However, by proper selection of olefin backbone and acrylic side chain, formulators can attach functional groups to provide properties that are not easily achieved by individual components or blends.
Polyolefins are crystalline in nature yet also contain noncrystalline side chains or tails that can offer features such as barrier and wear properties, moisture control, lubricity and feel. The crystalline section is grouped together as the crystalline polymer portion (CPP). The acrylic side chains of this particular composition are soft and nontacky, with either quaternary ammonium (quat) or amino functionalities. For ease of nomenclature, this will be referred to as the amorphous polymer portion (APP). The APP contributes adhesion, substantivity and stability to the olefin, along with enhanced film-formation.
The APP is bonded and entangled in a section of the noncrystalline olefin tails of the CPP. The lower density of an olefin normally would force its migration to the surface of a film during the drying process. However, by incorporating the grafted polymer composition, the APP portion of the polymer makes the CPP or olefin portion less mobile and it thus is more evenly distributed throughout the film (see Figure 1 and Figure 2).
One possible structure for polymers of this type involves the polyolefin backbone with grafted and entangling acrylic copolymers containing terminal quaternary ammonium groups (see Figure 3). By properly controlling the hydrophilic/hydrophobic balance, the formulator can obtain a polymer composition with the flexibility of the acrylate and substantivity of the quat while maintaining the barrier properties of the olefin.
Mechanism of Action
Block polymers, silicones and other assorted compounds have been used by formulators to provide protective barriers in an effort to retard the leaching of artificial colorants from hair. These barriers can function either by occlusion or by forming a relatively substantive ionic barrier to secure the color within the hair shaft. Quat functional olefin-grafted polymers appear to work as occlusive barriers, whose cationic charges serve to provide substantivity to the hair surface and use the crystalline olefin portion to occlude the cuticle openings, limiting color loss.
Color Retention Test Protocol
To test for color retention, bleached blonde human hair, pre-tressed and secured with epoxy in a single plastic clip, was first obtaineda. Each complete tress was six inches in length and weighed 5 g; 5 in/3 g of hair was accessible for coloring.
Olefin-graft in shampoo—color washout method: Dry, bleached hair tresses were combed and laid out on a plastic surface at a room temperature (RT) of 22°C and 55% RH. A retail, “medium auburn” permanent hair colorant was mixed by vigorous shaking for 1 min in the applicator bottle provided with the kit and quickly dispensed into a large plastic weigh boat. The dye mixture was applied evenly to each side of the tress with a 2-in applicator brush. After allowing the dye to process for 25 min at RT, each tress was rinsed with 42°C-tap water until the water ran clean. Tresses were gently blotted and allowed to lie undisturbed on paper towels for 1 hr. Each tress was then blow-dried and allowed to sit overnight at RT.
Initial L, a, b color measurements were recorded using a spectrophoto-meterb. Ten scans were recorded along the axis of both sides of each tress. Each group of tresses was washed and dried through 5, 10 and 15 wash/dry cycles using select laboratory-prepared shampoos. Tresses were wetted and 1 g of test shampoo per tress was applied. Tresses were massaged between the palms for 25 sec and rinsed with tap water at 42–45°C for 25 sec. Tresses were gently blotted and air dried with a blow dryer at ~51°C until dry to the touch. L, a, b color measurements were recorded after 5, 10 and 15 wash/dry cycles. ΔE values were calculated for each tress as follows (see Figure 4):
Eq. 1
ΔE = SQRT [(Lt - Lo)² + (at - ao)² + (bt - bo)²]
The control shampoo formula contained the following ingredients: 38.4% water, 25% ammonium lauryl sulfate, 25% ammonium laureth sulfate, 10% cocoamidopropyl betaine,
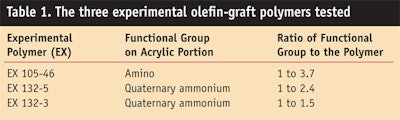
1% cocamide MEA, 0.2% fragrance and 0.4% DMDM hydantoin. The control shampoo was adjusted to a pH level of 6 with ammonium hydroxide. The control shampoo was clear. Test shampoo formulas were also prepared, identical to the control formulation with the exception that each contained 5% by weight of a pre-selected olefin-graft polymer emulsion (20–30% nonvolatile content). The test shampoos were also adjusted to a pH level of 6 with citric acid. All test shampoos were opaque. The three experimental olefin-graft polymers tested in this study are listed in Table 1.
Olefin-graft in dye preparation—color washout method: A professional, “intense medium auburn blonde” permanent hair color was prepared by combining one part 20-volume cream developer and one part cream dye-precursor base. Each olefin-graft polymer emulsion was post-added to the dye mixture. Distilled water was added as the control. Dye mixture was applied evenly to both sides of each dry, bleached hair tress with a 2-in applicator brush. The tresses were allowed to process for 25 min at RT. Each tress was rinsed with 42–45°C tap water until the water ran clean. Tresses were gently blotted and allowed to lie, undisturbed, on paper towels for 1 hr. Each tress was then blow-dried and allowed to sit overnight at RT. Initial L, a, b color measurements were recorded using a spectrophotometerb. Ten scans were recorded along the axis of both sides of each tress.
Each group of tresses was washed and dried through three wash/dry cycles using only the control shampoo (see Figure 4).
Tresses were wetted and 1 g control shampoo per tress was applied. Tresses were massaged between the palms for
25 sec and rinsed with tap water at 42–45°C for 25 sec. Tresses were gently blotted and air dried with a blow dryer at ~51°C, until dry to the touch. This wash/dry procedure was repeated three times. L, a, b color measurements were recorded after the third wash/dry cycle. ΔE values were calculated for each tress (see Eq. 1).
Results and Discussion
Olefin-graft in shampoo—color washout results: The three experimental polymers containing either a terminal amino or quaternary ammonium group on the acrylate portion of the polymer (see Table 1) were compared. The ΔE values calculated after 5, 10 and 15 wash/dry cycles showed that the shampoo containing experimental polymer EX 132-3 was consistently better at preserving hair color intensity than the control shampoo. Shampoos containing experimental polymers EX 132-5 and EX 105-46 initially appeared to contribute color preservation after 5 and 10 wash/dry cycles but failed to show significant color retention benefit after 15 wash/dry cycles (see Figure 5). The effect is perceivable to the human eye (see Figure 6).
Olefin-graft in dye preparation—color washout results: In order to better understand the potential of modified olefin-graft polymers for color protection, a second method of incorporation was evaluated. To a professional cream-based permanent hair colorant, each of the modified olefin-graft polymers was post-added; distilled water was also added as control. The various preparations were applied to bleached hair tresses and processed as indicated in experimental section. Each tress was subsequently run through three wash/dry cycles using only the control shampoo. ΔE values were calculated. Figure 7 is a comparison of ΔE values after three wash/dry cycles.
All three polymers had a positive effect on color retention when applied from the dye preparation. It can be noted that the highest level of color protection was observed when 2% by weight of experimental polymer
EX 132-3 emulsion was added to the dye preparation. In all the cases, initial color before washing was unaffected by the addition of polymer emulsions.
Multifunctional Attributes of Modified Olefin-graft Polymer
Effects of modified olefin-graft polymers on shampoo wet/dry attributes on hair: Figure 8 shows a spider graph of wet/dry evaluation of the control shampoo base and the base shampoo containing 5% of each of the modified olefin-graft polymers. The experimental polymer EX 132-3 in the shampoo base showed superior scores, 5 being the highest, in such parameters as wet combing, wet feel, frizz control, dry combing and static reduction.
Effects of modified olefin-graft polymers on foam height: Figure 9 contains foam height scores determined via a Ross Miles foam test utilizing a 1000-mL graduated cylinder. The graph clearly indicates a slight depression in foam height when 5% of each of the modified olefin-graft polymers is added to a shampoo base. The comparison also included a shampoo in which 5% of a commercially available amodimethicone emulsion was added, as-is. The silicone emulsion depressed the foam height to a greater extent than any of the modified olefin-graft polymers. It is noteworthy to mention the foam generated by shampoos containing the modified olefin-graft polymers had a more luxurious and rich texture than the control shampoo.
Effects of modified olefin-graft polymers on shampoo viscosity: Table 3 compares the viscosity response of adding 5% modified olefin-graft polymers to the shampoo base. It is quite clear that in a sulfate-based shampoo, the addition of the polymers increases the viscosity. The ionic nature of the polymer emulsions may be contributing to this observed viscosity response.
Conclusion
The overall positive results generated by these studies indicate that the novel quat modified olefin-graft polymers that were evaluated protect permanent, artificial hair colorant from color washout. This protection
was noted when the polymer was applied through a sulfate-based shampoo, or alternatively applied via the dye preparation itself. The aesthetics of sulfate-based shampoo formulations containing experimental quat olefin-graft polymer EX 132-3 were more desirable to consumers in parameters such as wet/dry combing, detangling, residual feel, smoothness and reduced static/flyaway. In addition, foam height and viscosity were not negatively affected. Samples of the sulfate shampoo base used in this study containing experimental olefin-graft polymer EX 132-3 showed slight instability after being stored for two months at 40°C.
New technologies such as this olefin-graft polymer can assist formulators in preparing hair-coloring and after-coloring preparations to meet the needs of the current hair care market. This technology could be a viable alternative to using silicone polymers when formulating color-retention products. In addition, olefin-graft polymer technology unlocks an array of possible new film-forming polymer combinations. By utilizing a wide assortment of modified acrylic portions and combining with a variety of different molecular weight polyolefins, new film-formers can be synthesized to meet the needs of the personal care industry.
References
1. FE Wall, Hair Coloring, in Cosmetics, Science and Technology, 2nd ed., MS Balsam and E Sagarin, eds, Wiley-Interscience: New York vol.2 (1972) pp 279, 289–320
2. A Schlosser, Silicones used in permanent and semi-permanent hair dyes to reduce the fading and color change process of dyed hair occurred by washout or UV radiation, J Cosmet Sci 55 (suppl), S123–131 (2004)