Recently, attention has been drawn to lifestyle improvement drugs and wellness products that aim to provide individuals with better health and more comfortable daily lives. The reason behind this is an aging population that not only desires to live longer, but also to enjoy a higher quality of life. Hair-growth products are a niche subset of this lifestyle-improvement category and products currently available in this area are classified variously as drugs, cosmetics or quasi-drugs.
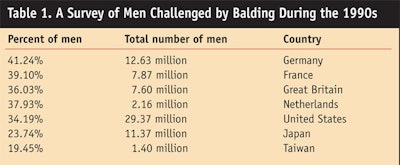
Surveys conducted during the 1990s revealed a significant number of men were challenged by some form of alopecia or balding (see Table 1);1 additionally, a growing number of women were experiencing thinning hair. It was around this time that several treatments intended for other ailments were commercialized to treat hair loss. These are described in further detail later in this review.
Targeting Hair Loss
Hair repeatedly grows and falls out in accordance with the hair cycle (see Hair Growth Process); in a normal scalp, 85% to 90% of all follicles will be in anagen phase and 10% in telogen phase at any given time.
The total number of scalp hairs maintained is estimated to be about 100,000. However, several factors can disrupt the phases of the normal hair cycle. If these shorten the anagen phase, they can cause the hair to become more fine, thus turning what was terminal hair into vellus hair. This is the case in male pattern baldness where large hair follicles become smaller and the change from terminal hair to vellus hair is accelerated.
When the telogen phase becomes longer, the ratio of hair in the telogen stage increases. As a result, a considerable amount of hair falls out without immediate replacement. As this situation continues, the scalp hair becomes thin and noticeable balding results. Female pattern baldness, or so-called diffuse baldness, also shows this pattern.
Balding may also show up as a phenomenon that accompanies aging, whereby terminal hair changes to vellus hair. Possible reasons for this disruption to the hair cycle, which hair growth products commonly target, include:
- Decreased function of hair matrix cells that produce hair, resulting in hair follicle atrophy and very fine hair;
- Decline in blood flow that carries nutrients to follicles, resulting in decreased metabolic function of hair matrix cells;
- Increased sensitivity of androgenic hormones resulting in male pattern baldness that develops due to blockage of hair growth;
- Activation of sebaceous gland functioning that deteriorates the scalp and hair follicle environment;
- Dietary imbalance resulting in the lack of nutrients to the hair follicle that adversely affect sebaceous gland functioning; and
- Other factors such as heredity or extreme mental stress.
To counter these factors, hair growth products have been developed that incorporate active agents to increase cell viability, accelerate blood flow, act as antiandrogenic agents and inhibit sebum secretion.
When a product developer is searching for effective materials or elucidating their mechanisms of action during the development stage, it is important that they select an appropriate method.
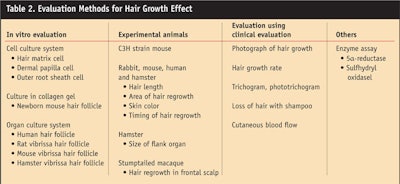
Table 2 outlines various common evaluation methods. It is important to note that when evaluating on animals, product developers should determine in which phase of the hair growth cycle the test subjects are. Hair growth efficacy should ultimately be evaluated objectively by clinical evaluation in humans.
Hair Growth Ingredients
An aging population and expanding market for hair-growth products have resulted in the aggressive development of active ingredients. Table 3 provides an overview, based on function, of traditional ingredients used in hair-growth products.
Of these, glyceryl monopentadecanoate and 6-(benzylamino)purin, which were approved in Japan as quasi-drugs after the revision of the Japanese Pharmaceutical Affairs Law in 1982, merit further discussion, as does extract of Excrementum bombycis—although not a quasi-drug component, it has been ascertained as effective in hair growth by the author. Minoxidil and finasteride are hair-growth agents marketed in the United States and Europe and also will be discussed.
Glyceryl Monopentadecanoate
It is suggested that hair follicles associated with male pattern baldness have reduced metabolic activity, that terminal hair transforms to vellus hair, and that clusters of hair become single strands, leading to baldness.6, 7 Ohba et al. found glyceryl monopentadecanoate to be highly effective in promoting hair growth after observing its ability to convert telogen to anagen in rabbits.8
This substance seems to follow a unique metabolic pathway as an odd-numbered fatty acid, promoting ATP production in hair follicle mitochondria and thereby supplying energy.9 Reportedly, when it is added to cultures of rat whisker papilla cells, ATP production and DNA synthesis increase. When it is added to organ cultures of human scalp hair follicles, lengthening is promoted, suggesting an activating function due to energy supply.10 This substance was also shown to be useful in clinical tests targeting male pattern baldness, with efficacy recorded at 76% in a double blind study on 155 subjects.11 Glyceryl monopentadecanoate is commercialized in the marketed product Yakuyo Mohatsuryoku (Lion).
6-(Benzylamino)purin
Also known as 6-benzyladenine, cytokinin B and benzylkinetin, this material is an adenine derivative with cytokinin effects in plants. Hatae et al. found in a hair growth study on C3H mice that this substance promotes the conversion of the hair cycle from telogen to anagen, stimulates DNA synthesis, and promotes the lengthening of the hair shaft in organ cultures of hamster whisker follicles.12 A double-blind study conducted with 86 human subjects showed that 6-(benzylamino)purin reduces the number of hairs that fall out during shampooing and showed an improvement rate of 47%, thus deemed “slightly effective or more” in clinical studies of hair on the human head. 6-(Benzylamino)purin is commercialized in the marketed products Grovis GX (Takeda Pharmaceuticals) and Yakuyo Furontesu (SS Pharmaceuticals).
Extract of Excrementum bombycis
According to hair growth tests conducted by the author on more than 100 crude drugs, a 50% ethanol extract of the Chinese crude drug Excrementum bombycis was found to be as effective as minoxidil in converting telogen into anagen in hair growth studies in C3H mice (Figure 1).13
In tests with human subjects, the material was effective in promoting hair growth rate, as well as in improving male pattern baldness and diffusing baldness (see Figure 2). The extract promoted the proliferation of outer root sheath cells that pass through hair papilla cells, preventing hair loss and improving hair quality.
Excrementum bombycis extract is incorporated in the commercial product Analys Will GTEX (POLA Corp.).
Minoxidil
First marketed in the United States in 1979 by Upjohn Corp. as an anti-hypertension drug, minoxidil encourages vasodilation based on its K+ channel opening action. During clinical studies with the material, excessive hair growth also was observed. The company therefore pursued approval in 1987 to market the material as a prescription topical drug to treat hair loss, launching Rogaineacontaining 2% minoxidil in 1988. In 1996, minoxidil was approved by the FDA as an OTC drug, whereupon the market rapidly expanded to nearly US$150 million in annual sales.14
In 1998, the company began marketing its OTC Rogaine Extra Strength for Men that boosted the material’s hair-growth effect by increasing its minoxidil content to 5%, following FDA approval. Hair-growth products containing minoxidil are also marketed in Western Europe and since 1999, the drug has been sold in Japan as an OTC drug, commercialized by Taisho Pharmaceuticals.
The mechanism by which minoxidil stimulates hair growth is not clear. One mechanism has been ruled out-it has been shown not to produce changes in hair follicle cells or hair papilla cells.15 The only known activity of minoxidil is that it dilates skin vessels causing blood flow to increase;16, 17 this appears to stimulate hair growth, which has been further confirmed by clinical human studies.18, 19
Finasteride
Propeciab, which contains the active ingredient finasteride, was the world’s first oral hair growth drug and forged a new market.
Merck Corp. marketed the material as a prescription drug after obtaining US Food and Drug Administration (FDA) approval in 1998. Finasteride was originally intended for use in treating prostatomegaly but as with minoxidil, it was observed to have the side effect of promoting hair growth. Subsequently it was developed as a hair growth drug.
Finasteride acts by blocking the production of dihydrotestosterone through inhibition of 5α-reductase. A clinical study of approximately 1,500 male subjects revealed the material to have a 48% efficacy rate.20
Sample Formulas
As described previously, hair loss can be caused by a variety of factors. Thus, hair growth formulations are often complex and can act on several of these factors. See Formulas 1, Formula 2 and Formula 3 for examples of hair growth formulas.
Regulatory Aspects of Hair-growth Products
Regulations that govern hair-growth products differ substantially from country to country. In Japan, regulations are stipulated in the Pharmaceutical Affairs Law and requirements for approval are governed by the Ministry of Health and Welfare (MHW). Products are classified as cosmetics, quasi-drugs and drugs according to their ingredients and effects; most are classified as quasi-drugs.
In the case of cosmetics, the name of the product, the composition of ingredients, the method of manufacture, and the usage method are submitted in documents to the MHW. Manufacturing approval is granted once safety has been reviewed. With quasi-drugs, it is necessary to submit an application for review of the safety and efficacy of the product and a request for approval for manufacture because the product contains an active ingredient. When applying for approval, the product name, composition of ingredients, manufacturing method, usage method and efficacy, as well as product specifications and measured values are submitted in documents to the MHW.
In the United States, hair growth products fall within a drug category and documents including the product name, the active ingredient and concentrations, and labeled contents are submitted for approval to the FDA.
In Europe, hair growth products fall within the cosmetics category and many countries require notification of the product. Safety studies and the control of such information are the responsibility of corporations that provide product assurance; administrative authorities are not involved.
The Future of Hair-growth Products
The main aim of hair-growth products is not to cure conditions such as pathological alopecia areata. Rather, they are used to prevent hair loss and promote hair growth for male pattern baldness, diffuse baldness in women and senile alopecia. On the scalp where hair loss is expressed, hair follicles are small and the activity of hair-producing matrix cells is reduced. Consequently, the activation of matrix cells is thought to prevent hair loss. This matrix cell activation has been the focus of hair growth ingredients in the past by two primary mechanisms: expanding capillaries in the tissue surrounding the hair follicles, therefore increasing blood flow, and directly activating the dermal papilla cells and matrix cells.
As a result, more effective product formulas and new ingredients with greater hair-growth effectiveness have been developed. A large number of currently available active ingredients in hair growth products are blood flow accelerants, as shown in Table 3. Representative of these include swertia herb extract, carpronium chloride, vitamin E and their derivatives.
More recently, hypertensive drugs such as minoxidil and diazoxide have likewise been developed as hair-growth agents that increase blood flow. By transporting nutrients and other substances into the matrix cells, increased blood flow activates the dermal papilla and matrix cells. Blood flow accelerants have therefore become essential components in hair-growth products to act on hair follicle vessels, increasing blood flow.
If drugs were developed that directly stimulate activity in the dermal papilla and matrix cells, then more effective hair-growth products would undoubtedly be produced. Future developments will likely continue as it has become possible to culture dermal papilla cells, hair matrix cells and hair follicle tissue.
New treatments for hair loss are on the horizon. Reports have emerged showing phenyl/thiophenylpropionamides could act as androgen receptor modulators.21 In addition, dutasteride has been shown to improve male pattern hair loss, as a randomized study in identical twins revealed.22
Although the causes and mechanisms of hair loss are still not completely known, research on hair continues and the relationship between cytokines—such as cell growth factors, angiogenic factors and apoptosis-related factors—and the hair cycle or hair follicle-forming cells is underway. The knowledge gained, together with the development of technology to manufacture preparations such as percutaneous absorption and sustained release preparations, will lead to the development of hair-growth products with even greater efficacy.
References
1.Aderanc Inc., Aderanc NEWS 3–2 (1998)
2.Y Hirai, K Takebe, M Takahashina, S Kobayashi and M Takeichi, Epimorphin: A mesenchymal protein essential for epithelial morphogenesis, Cell 69 471 (1992)
3.G Cotsaleris, T-T Sun and R Lavker, Label-retaining cells reside in the bulgi area of pilosebaceous unit: Implication for follicular stem cells, hair cycle and skin sarcinogenesis, Cell 61 1329 (1990)
4.J Reynolds and C Jahoda, Inductive properties of hair follicle cells, Ann NY Acad Sci 642 226 (1991)
5.S Arase, T Shikiji, N Uchida, S Katoh, T Fujie and Y Urano, Experimental approaches for the reconstitution of hair in vitro, Skin Pharmacol7 12 (1994)
6.K Adachi, S Takayasu, I Takashima, M Kano and S Kondo, Human hair follicles: Metabolism and control mechanisms, J Soc Cosmet Chem 21 901 (1970)
7.G Fazekas and T Sandor, Metabolism of dehydroepiandrosterone by human scalp hair follicles, J Clin Endocrinol 36 582 (1973)
8.K Ohba and H Tamai, Research and development of an activator on the energy metabolism in hair follicle and the efficacy of PDG on hair growth, Fragrance Journal 17(5) 85 (1989)
9.K Adachi, D Yokoyama, H Tamai, M Sadai and K Ohba, Effect of the glyceride of pentadecanoic acid on energy metabolism in hair follicles, Int J Cosmet Sci 15 125 (1993)
10.K Adachi, Evaluation method of hair growth in relation to the activation of hair matrix cells, Fragrance Journal 21(9) 50 (1993)
11.K Takeda, S Arase, S Watanabe, T Nagashima, Y Watanabe and A Sakuma, Clinical evaluations of LHOP on male pattern baldness, Nishinihon J Dermatol 55 727 (1993)
12.S Hatae, S Hamada, S Toshitani and Y Mishima, Benzyladenine(CTP) as an active ingredient for hair growth promoter, Fragrance Journal 21(9) 20 (1993)
13.M Suzuki and T Maeda, Effect of Excrementum bombycis extract on hair growth, Fragrance Journal 25(5) 52 (1997)
14.FDC Report, Rose Sheet (Jan 16, 1998)
15.K Katsuoka, H Schell, B Wessel and O Hornstein, Effects of epidermal growth factor, fibroblast growth factor, minoxidil and hydrocortisone on growth kinetics in human hair bulb papilla cells and root sheath fibroblasts cultured in vitro, Arch Dermatol Res 279 247 (1987)
16.C Wester, H Maibach, R Guy and E Novak, Minoxidil stimulates cutaneous blood flow in human balding scalps: Phamacodynamics measured by laser doppler velocimetry and photopulse plethysmography, J Invest Dermatol 82 515 (1984)
17.M Hirkaler and L Rosenberger, Simultaneous two-probe laser doppler velocimetric assessment of topically applied drugs in rats, J Pharmacol Method 21 123 (1989)
18.L DeVillez, Androgenetic alopecia treated with topical minoxidil, J Am Acad Dermatol 16 669 (1987)
19.K Takeda et al, Clinical evaluations of minoxidil on male pattern baldness, Nishinihon J Dermatol54 1009 (1992)
20.K Kaufman, Clinical studies on the effects of oral finasteride, a type II 5α-reductase inhibitor, on scalp hair in men with male pattern baldness, Hair Research for the Next Millenium, Elsevier, Amsterdam 363 (1996)
21.Patent appl PCG/US2007/021984, Int. class A61K 31/4436, Phenyl/thiophenylpropionamides as androgen receptor modulators, Merck & Co., Inc. (Apr 24, 2008)
22.D Sough, Dutasteride improves male pattern hair loss in a randomized study in identical twins, J Cosmet Dermatol 6(1) 9-13 (2007)