Nail polish must function as a coating that is both decorative and wear-resistant, as discussed in a previous article.1 Nail polish encounters harsher wear than any other makeup product, causing it to dull, chip and thin. People use their fingers for almost everything they do, including texting, washing hair, cleaning, etc., so nails are exposed to a multitude of mechanical and chemical insults. Therefore, nail polish formulation has historically included ways to improve its wear.1
The challenge with traditional nitrocellulose-based nail polishes is that the coating is a combination of individual polymer molecules—nitrocellulose and modifying polymers, plasticizers, pigments, effect pigments and other additives. The coating even contains some residual solvents with higher boiling points. There is no internal structure to the film, i.e., no cross-linking to hold the polymers and other materials together. As such, mechanical and chemical insults can lead to cracks, which in turn lead to chipping, flaking and loss of gloss.
Another aspect of wear is adhesion of the nitrocellulose and other polymers to the nail plate, which can be affected by a number of factors. Physical differences in nails such as flexibility, water content and surface roughness/smoothness can play a role, as demonstrated by some individuals exhibiting only minor chipping after four to five days, while others show much greater wear after just one or two days. Also key is the preparation of the nail before application; nails should be as clean as possible, with no oily residue remaining, for improved wear.
Attempts to improve wear include adding acrylate/methacrylate copolymers and terpolymers that contain pendant polar groups to nail polish formulations.2 This is because polar groups such as hydroxyl from acrylic acid (see Figure 1), and acetoacetoxy from acetoacetoxyethyl methacrylate (see Figure 2), presumably provide better interaction between the polymer and nail plate. While the addition of polar polymers showed some improvement in wear, it also required a topcoat in some cases because the acrylic/methacrylate terpolymers softened the nitrocellulose film. The topcoat therefore protected the nail polish much like the clear coat on car paint.
Despite these efforts, consumers continued to demand improved wear and gloss. In relation, nail salons moved toward two-part liquid/powder systems in which the liquid contains acrylate and methacrylate monomers, and the powder is a polymer containing the initiator(s) and other additives. This initiator is usually heat-sensitive, and upon exposure to heat from the hands, it generates free radicals that can initiate free-radical polymerization of the liquid monomers. A good example is the polymerization of methyl methacrylate, initiated by benzoyl peroxide. Exposure to heat causes the O-O bond to cleave, generating two free radicals that initiate the polymerization (see Figure 3).
Gel nails provide advantages over two-part systems. First, the photoinitiators were incorporated into the system rather than being added at the time of application. Thermally activated initiators could not be used in this way, as exposure to heat prior to application to the nails would start the polymerization reaction in the container, hardening the material and rendering it useless. The other advantage was the greater control over the final polymer structure in terms of composition, degree of cross-linking and three-dimensional structure.
Gel Nail Mechanism
Gels have been around since the 1980s;3 they can be applied over a nail extension or over the individual nail. Hard gel nails require mechanical means to remove, whereas soft gel nails can be removed by soaking in acetone. In either case, although they may contain solvents, gel nails do not dry by the evaporation of a solvent upon application. Instead, the gel is cured by exposure to UV light to convert it from a fluid to a solid state. The UV light interacts with a photoinitiator to generate free radicals. For photoinitiators to function, they must absorb enough energy (UV light) to create an excited state. The molecules then dissociate or rearrange to generate free radicals. These free radicals interact with the monomers and oligomers in the gel to initiate polymerization (see Figure 4).
Monomers and Oligomers
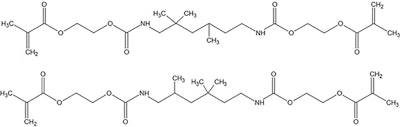
In monomers such as ethyl methacrylate, acrylic acid and others, only one double bond is available for polymerization. As such, they can only form linear polymers (see Figure 5). To form two or three-dimensional structures, functional molecules with two, three or more double bonds are required for polymerization; Figure 6 shows such a schematic. Examples of difunctional molecules include ethylene glycol dimethacrylate (see Figure 7) and homologues of polyethylene glycol diacrylate (see Figure 8); other functionalized and difunctional molecules are shown in Figure 9.
Increasing the hardness of the film further requires a higher cross-link density using trifunctional acrylates and methacrylates such as trimethylolpropane tricarylate and trimethacrylate (see Figure 10 and Figure 11). However, note that using trifunctional acrylates and methacrylates at too high a level can lead to an inflexible and brittle film.
Different approaches can provide a polymerized gel film that exhibits both sufficient hardness and flexibility. One is an optimized blend of monomers and di- and tri-functional acrylates and methacrylates. Here, monomers provide chain length, which increases flexibility, while the multifunctional acrylates and methacrylates give strength and hardness.
Judicious use of acrylates and methacrylates alone also can affect the final polymer’s properties. Methacrylate polymers tend to be more brittle than acrylate polymers, as evidenced by the difference in glass transition temperatures (Tg). The glass transition temperature for polymethyl methacrylate is 105°C, while the glass transition temperature of polymethyl acrylate is 10°C. What a difference a methyl group makes.
Challenges to using monomers include their odor and potential for irritation. To avoid these issues, gel manufacturers have mixed monomers with oligomers. From the prefix oligo meaning few, and mer(s) meaning part(s)—oligomers are short polymer chains with acrylate or methacrylate groups on one or both ends and a variety of different short-chain polymers in the middle. One particular class is polyurethane end-capped with acrylate functionality. These oligomers also give added toughness to the cured film.
The film-forming ingredients that cure in GelColor by OPI,4 per its MSDS (see Table 1), include di-hema trimethylhexyl dicarbamate (see Figure 12), hydroxyethyl methacrylate (HEMA) (see Figure 13) and hydroxypropyl methacrylate (see Figure 14). Another example is Creative Nail Design’s Brisa Gloss Gel Top Coat (see Table 2).5 This formulation also contains di-hema trimethylhexyl dicarbamate and hydroxypropyl methacrylate, but also contains neopentyl glycol dimethacrylate and isopropylidenediphenol PEG-2 dimethacrylate. Since this is a high-gloss top coat, the isopropylidenediphenol PEG-2 dimethacrylate increases the refractive index due to its phenyl functionality, leading to increased gloss. Figure 15 graphically shows how the photoinitiator interacts with the multifunctional monomers and functionalized polymers or oligomers to produce a three-dimensional (3D) polymer network.
Photoinitiators
As noted, free radicals must be generated to initiate polymerization, and photoinitiators accomplish this by absorbing enough energy (UV light) to create an excited state. The photophysics of many commercially available photoinitiators has been studied extensively.6-8 Once the excited state is formed, the molecule can dissociate, rearrange or abstract a hydrogen to generate a free radical. It is this free radical that interacts with the monomers and oligomers to initiate polymerization (see Figure 15). Examples of photoinitiators include hydroxycyclohexyl phenyl ketone (see Figure 16), phenyldimethoxyacetophenone (see Figure 17), and trimethylbenzoyl diphenylphosphine oxide (see Figure 18).
The molecular structure of the photoinitiator determines the wavelengths of UV light that will be absorbed, as well as the amount. Hydroxycyclohexyl phenyl ketone has a maximum absorption at approximately 240 nm, and smaller absorption bands at 280 nm and 330 nm. Trimethylbenzoyl diphenylphosphine oxide has a maximum absorption at 295 nm, and smaller absorption bands at 380 nm and 393 nm. Higher levels of aromatic structure increase the amount of UV light absorbed, and increased conjugation moves the absorption closer to the visible spectrum.
An important reason for using combinations of photoinitiators with different absorption spectra is to ensure the gel nail is completely cured throughout the thickness of the film, especially when working with colors. This is important because added pigments reduce the penetration depth of UV light due to scattering. A comparison of UVA, UVB and UVC wavelength and depth of curing (see Table 3) shows that photoinitiators with different absorption maxima are required to cure through the whole film. Certain photoinitiators will stay in their excited state longer than others, providing more opportunity to initiate a polymerization.
An important challenge to consider when working with photoinitiators is that their excited states often are quenched by oxygen before they can generate radicals; or, the radicals generated react with oxygen. This explains why, after curing, there is a sticky layer on the surface that requires removal. The excited photoinitiators at the surface of the film do not participate in polymerization. Likewise, polymerization can be affected by oxygen at the surface, leaving a partially polymerized layer on the surface.
Using the correct amount of photoinitiator is also critical when formulating gel systems. Too little leads to insufficient curing, leaving too soft a film on the nails. Too much will generate an excess of polymer chains that are too short or cross-linked, resulting in a film that is too hard and brittle, and not flexible enough.
UV vs. LED Curing
One area of confusion is UV vs. light-emitting diode (LED) curing. Both types use UV light; the difference is the output of the two lamps. UV curing uses a compact fluorescent light (CFL) to produce UV light when an electric current is passed through a mercury vapor. LED curing uses a semiconductor diode to produce UV light when an electric current is applied through the device.
The big difference is the spectra of the UV light produced. Mercury vapor lamps have a broad distribution of peaks in the UV spectrum, while LED lamps produce a narrow band of UV light. This is why LED-curing gels must be used with the appropriated LED light source. If the spectral output of the LED lamp does not match the absorption spectrum of the photoinitiator, the photoinitiator will not be excited and polymerization will not occur, or will occur only slightly. Since mercury CFL lamps produce UV light over a broader range, they can cure LED systems—although not as efficiently as an LED lamp. On the other hand, LED lamps are smaller than CFL mercury lamps, so more can be incorporated into UV-curing units. Additionally, LED lamps do not generate as much heat as mercury lamps.
In relation to UV exposure, one concern has been whether the amount of light used to cure gel nail systems poses health risks, particularly cancer. A recent study published in Photochemistry and Photobiology concluded that “the risk of developing non-melanoma skin cancer was 11-46 times lower than the risks of being exposed to natural midday sunlight.”9 The study did not mention; however, that individuals taking medication requiring them to avoid natural sunlight without proper protection should be cautious when using UV nail lamps.9 An announcement from the Professional Beauty Association’s Nail Manufacturers Council on Safety also supported this study, and referenced two previous studies that further support the safety of LED and UV lamps.10
Overview and Outlook
Formulating a UV or LED-curable gel requires detailed knowledge of monomers, photoinitiators and additives, as well pigments and effect pigments. The final function, whether a hard gel or soft gel, extension or not, will determine which types, combinations and levels of monomers, multifunctional acrylates and methacrylates, oligomers and photoinitiators are required to achieve the desired result.
Soft gels, also referred to as soak-off gels, were developed to avoid one of the drawbacks of hard gels—removal. Hard gels require mechanical means, i.e., grinding, to remove them, whereas soft gels are removed by soaking them in acetone. Obviously, soft gels have a lower cross-link density and different composition than hard gels.
There has been a recent trend for gel systems that apply more like traditional nail polish, rather than a heavier, more viscous product that requires sculpting. Numerous products are available both commercially and for salon use. Examples include products from Sally Hansen, OPI and Creative Nail Design. Most of these are provided as systems, which include a gel base, color coat and top coat or sealer—all of which must be cured. This also explains the popularity of LED gels, which cure faster than UV gels.
An interesting new gel polish that does not require a base coat or top coat, and applies like a traditional nail polish but without solvent, is Origin All-in-One-Gel Polish.11 This patent-pending product12 is produced by California Chemical Specialties. Another relatively new development is Sally Hansen Miracle Gel. This product combines a color coat and top coat that cure in natural light. Both coats contain an oligomer, and the top coat contains a photoinitiator, which after application and drying, initiates photopolymerization upon exposure to natural light.13
Conclusion
Gel nails have come a long way from the 1980s when they first were used. These products have undergone a tremendous evolution in terms of types of products available and ease of use. With the rapid advances in technology and chemistry, who knows what the future holds for gel nails? One thing is for sure—it will be exciting.
References
- FC Pagano, A review of nail polish: The industrial cosmetic, Cosm & Toil 126 5 372-380 (May 2011)
- US Pat 5,772,988, Nail enamel compositions from acetoacetoxy methacrylate copolymer, FC Pagano, AA Patil, RW Sandewicz, WL Anton and HJ Spinelli, assigned to Revlon Consumer Products Corp (May 10, 1996)
- www.nailsmag.com/article/108201/hard-vs-soft-a-closer-look-at-gels (Accessed Sep 30, 2014)
- www.intbeauty.com.au/MSDS/OPI_MSDS/GelColor_All_Shades_OPI_MSDS-246_IBS.pdf (Accessed Dec 8, 2014)
- www.cnd.com/pro-products/enhancements/gel-system/prep-finish/brisa-gloss# (login required; Accessed Nov 13, 2014)
- NJ Turro et al, Probing the reactivity of photoinitiators for free radical polymerization: Time-resolved infrared spectroscopic study of benzoyl radicals, J Am Chem Soc 124(50) 14952–14958 (2002)
- Y Yagci, S Jockusch and NJ Turro, Photoinitiated polymerization: Advances, challenges and opportunities, Macromolecules 43(15) 6245–6260 (Jun 16, 2010)
- NJ Turro et al, Photochemistry and photophysics of α-hydroxy ketones, Macromolecules 34(6) 1619–1626 (2001)
- JC Dowdy and RM Sayre, Photobiological safety evaluation of UV nail lamps, Photochemistry and Photobiology 89 961-967 (2013)
- www.probeauty.org/docs/nmc/PBA%20-%20DrSayre%20UV%20Lamps%20Release%20v3.pdf (Accessed Nov 13, 2014)
- www.cachemspecialty.com/products/29-gel-polish/origin-all-in-one-gel-polish (Accessed Nov 13, 2014)
- US Pat App 20130263875, Curable nail enhancements, T Burgess and AB Louis, assigned to ESSchem Inc. (Oct 10, 2013)
- www.sallyhansen.com/sites/default/files/global_miracle_gel_faq.pdf (Accessed Oct 12, 2014)