To read the following article in its entirety, click through to your January 2020 digital magazine.
In 1992, the International Agency for Research on Cancer (IARC) identified ultraviolet radiation (UVR) as a human carcinogen.1 Exposure to solar radiation, as the main source of UVR, is also a known contributing factor in development of skin cancer.2, 3 In relation, over the past few decades, the number of people in the United States diagnosed with skin cancer has increased. Hence, being protected from sunlight would seem essential to preventing DNA damage that leads to nonmelanoma and melanoma skin cancers; however, the sunscreen filters used for such pose some risk of their own. This review considers the safety vs. risk of consumer sunscreen use.
UVR Exposure
UV light is divided into three wavelengths: UVA, UVB and UVC. UVC (200–280 nm) is primarily absorbed by ozone in the stratosphere. In contrast, UVA (320–400 nm) and UVB (290–320 nm) can reach the earth’s surface, where they can influence humans and ecosystems.
While long-wave UVA can deeply penetrate the epidermis and dermis, causing premature aging in skin cells, UVB, which forms less than 10% of sunlight, is more active and more capable of causing sunburn. Therefore, UVB can be regarded as one of the key causes in inducing DNA damage and skin cancer. This has led to major efforts to protect human skin against the detrimental effects of sunlight—e.g., sunscreens.
Sunscreen Actives
Demand for chemo-preventive UV filters and sunscreen products has widely increased.4, 5 Sunscreens temporarily protect human skin against UV radiation and are found today in various formats; i.e., lotions, creams, water-based gels, sticks, sprays and cosmetic preparations.
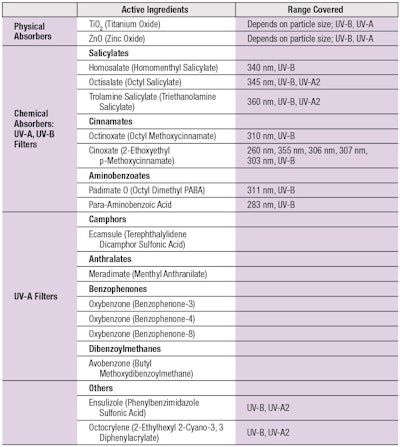
Based on their mechanism of action and chemical composition, there are two types of sunscreens: inorganic and organic. Table 1 shows the 17 active sunscreen ingredients currently approved by the U.S Food and Drug Administration (FDA).5, 6
Inorganic filters: Inorganic filters include metal oxide particles. Titanium dioxide (TiO2) and zinc oxide (ZnO) are the only ones approved for use in the United States. These materials can reflect or scatter visible, UV and infrared radiation away from skin. TiO2 and ZnO offer UVB and UVA protection, respectively.6 Combinations of both particles provide broad-spectrum UV protection. In addition, they offer advantages such as considerable stability against sunlight, the lack of skin sensitization potential, and limited skin penetration.
However, to achieve optimum effects, a thick application is required. This results in unpleasant esthetics, i.e., a visible white film on skin.7 Nanotechnology has partially solved this problem since nano-sized particle of these materials (< 100 nm) not only make them easier to apply, but also transparent on skin.8
Nonetheless, this solution has disadvantages. Reducing the particles to nano-size changes some of their properties, and they become more bioactive. For instance, some argue they can more easily penetrate skin and other tissues, which raises concerns over safety. In addition, more free radicals can be produced on the metal oxide surfaces and cause genotoxicity. This is because their photocatalytic activity, i.e., UV radiation absorption in the presence of water, increases with their reduced particle size.9
Some in vitro and in vivo studies related to the toxicity of ZnO nanoparticles (NPs) has demonstrated their penetration ability is low, unless the skin barrier is damaged due to sunburn, disease or physical damage. Nonetheless, exposure regularly to these particles might be of toxicologic concern.10
Organic filters: Organic filters, on the other hand, are aromatic structures that absorb UV. Benzophenones (BPs), camphors and cinnamates are among the well-known organics widely used in sunscreens and other cosmetic products. Organic filters can be classified by the type of UV radiation they absorb, namely: UVB, UVA or UVB-UVA filters (see Table 1).
Continue reading in the January 2020 digital magazine...