Facial aging is a progressive phenomenon known to be the cumulative result of intrinsic and extrinsic factors. It leads to changes in visible skin color due to the loss of vascular and pigmentation homogeneity, resulting in age spots, telangectasia, a sallow yellow color and loss of a pink glow. It also leads to changes in surface texture, with the appearance of fine lines progressing into deeper folds and creases, worsened by the loss of skin elasticity.1, 2 The type and severity of aging-associated skin changes and their time of occurrence and relative appearance differ among individuals. This is particularly visible when assessing different ethnic groups, as shown in various studies comparing aging processes in Caucasian women versus other skin types.3-5
It is difficult to clearly separate the participation of intrinsic vs. extrinsic factors in facial aging. Intrinsic factors include genetic traits such as melanin type, bone structure, skin texture and hormone homeostasis.6, 7 A variety of exogenous factors linked to lifestyle, including cultural inheritance and socio-economics, as well as adaptation to ecological and climatic conditions, have been described as responsible for impacting skin aging. UV exposure and smoking are the most understood and prominent accelerators.8, 9 Temperature and humidity also have been shown to influence aging. Seasonal changes in skin composition have been measured,10-12 and individuals having similar skin types but located in two different areas of the same country—thus exposed to different external factors—were shown to age differently.13 Therefore, the place and country of residence, closely associated with geographic and cultural environments, are overall key factors in understanding aging and its characteristics.
Although clinical signs of facial aging are well-characterized, investigations mostly have been performed in populations considered to have a Caucasian skin type. This characterization, in fact, refers predominantly to individuals of white skin living in Western Europe and North America.14, 15As of this writing, no data has been published on facial skin aging in Eastern European, in particular Russian, individuals. The objective of this study was, therefore, to assess and quantify 23 facial aging features (including wrinkle number and depth, skin texture and skin color) in Russian women ages 30 to 65 years, using severity grading and image analyses of high resolution standard photographs.
Materials and Methods
Population: Two hundred and three healthy Russian women, ages 30–65, were enrolled in the study conducted in Moscow (58⁰N; 38⁰E) in January 2011. The women recruited had never had surgical intervention on their faces, e.g., cosmetic fillers or face-lifts, and had not received professional resurfacing treatments within the three years preceding the study. Subjects were divided into seven subgroups of 29 volunteers each, arranged in five-year age clusters.
Measurements: Volunteers arrived for the study with clean faces, using no particular cleanser, and having applied no makeup. After a 30-min acclimation to control conditions, front and 37-degree facial profile views were photographed using a standard imaging high resolution digital cameraa under standard white and cross-polarized lighting conditions.
Grading of photographed facial wrinkles: Photographs obtained under general white light were cropped and resized using an image editing softwareb to enable expert grading of facial wrinkles using the photographic scales published by Bazin et al.16 For features present on both the front and 37-degree facial profiles (i.e., crow’s feet, wrinkles at the corners of the mouth, nasolabial fold wrinkles and under-eye wrinkles), cropped photographs were randomly assigned from left or right, then mirrored so that all features appeared from the left side of the face. All photographs were observed in a randomized order by three expert graders under standard conditions. To validate the robustness of the grading, several photographs were presented twice. The scores of the expert graders were averaged to give a clinical assessment of severity for each chosen attribute for each volunteer.
Image analysis: Cross-polarized photographs were evaluated using image analysis softwareb customized by a proprietary color-measuring macro designed to measure the three L*, a*, b* coordinates of the standard CIE lab colorspace used for skin color measurements.17 Seven regions of interest (ROI) defined by the investigators were analyzed to evaluate the skin color Individual Typology Angle (ITAº), skin clarity (L*), color intensity (Chroma C), cheek redness (a*), facial redness (a*), skin sallowness (b*), under-eye dark circle severity (ΔE) and facial color unevenness (ΔE).17, 18
Additional facial feature information was obtained from the front view photographs by the automatic detection capability of the softwareb in a masked area that was predetermined for all panelists according to key facial landmarks; e.g., left and right corners of eye, bridge, nose and corner of the mouth. Skin roughness was calculated by the number of variations in surface topography from the analysis of the color gradation from the surrounding skin tone on the general white light photographs. Pores were analyzed in the same way; the software automatically taking only negative volume compared to the analysis of skin roughness. From the cross-polarized photographs, data on red areas and brown spots was generated separately using the built-in imaging technology that transforms red, green and blue channel (RGB) photographs into the RBX colorspace, where the red and brown channels represent hemoglobin and melanin distributions.19 The number and the severity, i.e., total size and intensity of detected instances, of those features were automatically determined once the area of interest was defined and corrected.
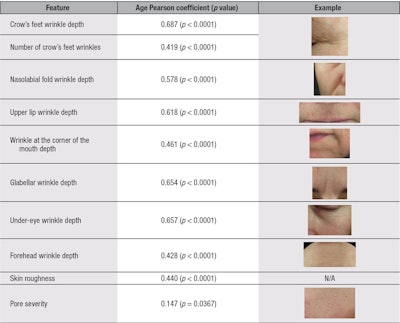
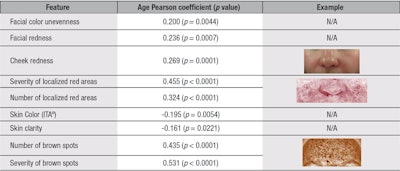
Statistical analyses: In total, twenty-two facial features were measured from the photographs captured (see Table 1). Linear Pearson correlation coefficients were calculatedc to determine how each of the measured skin attributes change with chronological age. Results were divided into two clinical clusters: wrinkles and texture (see Table 2) and skin color (see Table 3). Only parameters correlating with chronological age are shown. Furthermore, all strong correlating attributes having a Pearson Correlation factor above 0.4 were further studied by stratifying the panel into five-year age clusters to understand the pattern of change through the aging process (see Figure 1, Figure 2 and Figure 3). Analysis of variance at one factor, followed by a Tukey comparison test, was used to compare values associated with the five-year age clusters within each attribute.
Results and Discussion
Analysis of the correlation coefficients demonstrated that the severity of all measured wrinkles was found to significantly increase with chronological age. Within the same wrinkle/texture category, the number of crow’s feet wrinkles, skin roughness and pore severity were also found to correlate with increasing age (see Table 2). In the skin color category, pigmentation disorders (i.e., the number of brown spots and their severity; uneven skin tone associated with facial color unevenness; and vascular disorders based on both localized red area counts, and severity and diffuse facial and cheek redness) were found to significantly increase with age, as shown in Table 3. Furthermore, the following parameters were found to decrease with chronological age: skin clarity and color (ITAº), indicating the skin becomes duller and darker with age, respectively (see Table 3). However, under-eye dark circle severity, skin sallowness, skin color intensity and number of pores were not found to correlate with age (data not shown).
Having divided the panel of volunteers into age subgroups, the authors could study the evolution of the twelve strongly correlated attributes shown in Figure 1, Figure 2 and Figure 3. Observed were three age-dependent kinetic aspects for these facial attributes. The first kinetic corresponded to an overall linear increase in severity from subjects 30 to 65 years; forehead wrinkle depth, nasolabial wrinkle depth and the number of brown spots exhibited this profile (see Figure 1). Nasolabial wrinkle depth was further characterized by a slight slow-down in the late 40s (45–49 years) before picking up strongly again at 50 years.
The second kinetic was associated with a gradual increase in severity in subjects 30 to 49 years, followed by acceleration at 50 years, then a leveling off at 55+ years. Crow’s feet, under-eye and glabellar wrinkle depths fell into this group (see Figure 2). The accelerated wrinkle deepening observed at 50 years of age was found to be statistically significant for glabellar wrinkles. The number of crow’s feet wrinkles exhibited nearly the same profile—with a constant and strong increase from 30 to 44 years, then gradual leveling off at 45+ years, reaching an average grade of three wrinkles for the 60 to 65 year-olds group (see Figure 2).
Finally, the third kinetic tended toward an exponential profile, with a late onset around 45 to 50 years old. This was observed for a combination of wrinkle- and color-related features; corner of the mouth and upper lip wrinkle depths, and severity of brown spots and localized red areas followed this profile, although there was a lesser rate of change overall for the latter.
Within the age-correlating facial features measured, and according to the second kinetic, wrinkles from the eye area (i.e., crow’s feet and under-eye wrinkles) and glabellar wrinkles were found to be the quickest wrinkles to deepen with age in Russian women, starting from 30 years old—although forehead and corner of the mouth wrinkles were associated with the highest mean grade for the youngest age group. Beyond the three kinetics described, it was observed that most of the signs of facial aging shown in Figure 1, Figure 2 and Figure 3 exhibited a common severity increase between the ages of 45 to 50 years. Although this age-dependent increase in feature severity was only statistically significant for the glabellar wrinkle (see Figure 2), the eye area wrinkles, mouth area wrinkles and pigmentary spots followed the same trend.
Menopause has been associated with a decrease in skin collagen content, reduced vascularity, loss of firmness and elasticity, as well as deep wrinkling and thinner skin.20 The average age for menopause in Russian women has been reported as 49 years and could correspond to the underlying cause of this key measured facial aging event.21 In this study, although no measurements of skin elasticity or firmness were performed, the nasolabial fold depth could be used as an indicator of facial sagging.22 Its kinetic profile indicates a gradual but slow deepening, compared with other wrinkles, but reaching a noticeable increase in severity from mild to moderate depth (between grade 2 and 3) from 50+ years (see Figure 1).
Interestingly, the average nasolabial fold depth was less severe in subjects 30 to 65 years in this study than in one by Bazin et al. on a panel of French women, which could indicate Russian women are less prone to sagging.16 Furthermore, in that same panel, the 45 to 50 year-old severity shift in facial aging was not observed. However, it would be difficult to comment on this difference without a complete understanding of the panel characteristics related to menopause and any hormone replacement therapy.
The late onset observed for the pigmentary changes may also be explained by chronic photo-damage.2 The lack of significant age-related change in skin yellowness (b*) in the Russian women panel confirmed findings by Rigal et al. in a Caucasian population living in Chicago.23 However, here, both diffuse redness parameters—i.e., cheek and facial redness–significantly correlated with increasing age, which differs from Rigal et al.’s findings where skin redness on both the forehead and cheeks was reported to decrease.23
Also, the present results showed that skin color unevenness did not correlate with age, which differs from findings from Rigal et al. The methodology described here provides a measurement of “macro” skin color unevenness, i.e., the difference between five large facial areas (ΔE between ROI on the nose, chin, cheeks and forehead), and is not directly comparable to the technique used in the Rigal et al. study. As per the L* and the ITA° significant correlations with increasing age, the present results indicated that aging facial skin becomes darker and duller over the years. This confirms previous work on the photo-exposed skin of Caucasian subjects.24 However, it should be noted that value changes found here were minimal (ΔL*= -1.49) between the youngest and the oldest age group, on average, and that these changes were not significant. One could speculate these changes to be linked to the sun exposure profiles of the panelists.
Due to differences in study settings and the skin aging features analyzed, it is challenging to highlight further regional differences within the “Caucasian” skin type. A comparison of two distinct Caucasian panels within the same study would be interesting. Nevertheless, this study of facial aging features in Russian women showed the relevance of key aging features, their prevalence and their characteristic kinetics over the years. Wrinkles in the eye area and the glabella were shown to develop first and the fastest from 30 years, while the mouth area wrinkles had a later onset and exponential kinetic at around 45 to 50 years. Furthermore, the peri-menopausal age was shown to be associated with a key increase in severity for all areas of the face for both color and wrinkle-related parameters. Pigmentary changes were of particular relevance in this panel of Russian women, with the increase in cheek and facial redness (diffuse) as well as localized red areas, which could be associated to the prevalence of telangectasia.
Conclusion
In this study, the authors measured twenty-three facial features in Russian women related to wrinkles and skin pigmentation on high resolution standard photographs using various techniques. In total, more than 200 Russian women ages 30 to 65 years were included, and as many as nineteen facial attributes were shown to significantly correlate with age. Studying the patterns of change by five-year age clusters for the key facial aging features enabled the identification of three kinetic trends, further highlighting key facial aging events in Russian women.
To the best of these authors’ knowledge, this is the first published work to date that enables a better understanding of the visible facial aging features of Russian women; further, to known “Caucasian” western European individuals. It also offers more key information for the development of relevant anti aging products dedicated to Russian women’s skin care needs from the ages of 30 to 65 years.
References
- RG Glogau, Physiologic and structural changes associated with aging skin, Dermatol Clin 15(4) 555-9 (1997)
- R Jackson, Elderly and sun-affected skin. Distinguishing between changes caused by aging and changes caused by habitual exposure to sun, Can Fam Physician 47 1236-1243 (2001)
- K Tsukahara et al, Comparison of age-related changes in wrinkling and sagging of the skin in Caucasian females and in Japanese females, J Cosmet Sci 55(4) 351-371 (2004)
- GG Hillebrand, MJ Levine and K Miyamoto, The age-dependent changes in skin condition in African Americans, Asian Indians, Caucasians, East Asians and Latinos, IFSCC Magazine (4) 259-266 (2001)
- S Nouveau-Richard et al, Skin aging: A comparison between Chinese and European populations. A pilot study, J Dermatol Sci 40(3) 187-193 (2005)
- BA Gilchrest, A review of skin aging and its medical therapy, Br J Dermatol 135(6) 867-875 (1996)
- S Verdier-Sevrai, F Bonte and B Gilchrest, Biology of estrogens in skin: Implications for skin aging, Exp Dermatol 15(2) 83-94 (2006)
- C Kennedy, MT Bastiaens, CD Bajdik, R Willemze, RG Westendorp and JN Bouwes Bavinck, Effect of smoking and sun on the aging skin, J Invest Dermatol 120(4) 548-554 (2003)
- WC Leung and I Harvey, Is skin aging in the elderly caused by sun exposure or smoking? Br J Dermatol 147(6) 1187-1191 (2002)
- R McCallion and A Li Wan Po, Dry and photo-aged skin: Manifestations and management, J Clin Pharm Ther 18(1) 15-32 (1993)
- L Declercq, N Muizzudin and L Hellemans, Adaptation response to human skin barrier to a hot and dry climate, J Invest Dermatol 119(3) 716 (2002)
- J Rogers, C Harding, A Mayo, J Banks and A Rawlings, Stratum corneum lipids: The effect of aging and the seasons, Arch Dermatol Res 288(12) 765-770 (1996)
- GG Hillebrand, K Miyamoto, B Schnell, M Ichihashi, R Shinkura and S Akiba, Quantitative evaluation of skin condition in an epidemiological survey of females living in northern versus southern Japan, J Dermatol Sci 27(supp 1) 42-53 (2001)
- AV Rawlings, Ethnic skin types: Are there differences in skin structure and function? Int J Cosmet Sci 28 79-93 (2006)
- K Holubar, What is Caucasian? J Invest Dermatol 106:800 (1996)
- R Bazin and E Doublet, Skin Aging Atlas, volume 1, Caucasian Type, MED’COM, Paris (2007)
- IL Weatherall and BD Coombs, Skin color measurements in term of CIELab color space values, J Invest Dermatol 99 468-473 (1992)
- Guideline for the Colorimetric Determination of Skin Color Typing and Prediction of the Minimal Erythemal Dose (MED) Without UV Exposure, Colipa, Brussels (2007)
- www.canfieldsci.com/FileLibrary/RBX tech overview-LoRz1.pdf (Accessed Jan 23, 2014)
- G Hall and TJ Phillips, Estrogen and skin: The effects of estrogen, menopause and hormone replacement therapy on the skin, Am Acad Dermatol 53(4) 555-68 (2005)
- F Thomas, F Renaud, E Benefice, T de Meeüs and JF Guegan, International variability of ages at menarche and menopause: Patterns and main determinants, Human Biology 73(2) 271-290 (2001)
- T Ezure and S Amano, Involvement of upper cheek sagging in nasolabial fold formation, Skin Res Technol 18(3) 259-264 (2012)
- J de Rigal et al, the effect of age on skin color and color heterogeneity in four ethnic groups, Skin Res Tech 16 168-178 (2010)
- M Ramos-E Silva and SCDS Carneiro, Cosmetics for the elderly, Clin Dermatol 19 413-423 (2001)