Cationic polymers are often used in shampoo and body wash formulations to provide conditioning and antistatic properties, and to aid in the deposition of other ingredients. Polymers of this type may be entirely synthetic or derived from bio-based renewable resources. The perceived benefits are delivered for both skin and hair due to these ingredients' substantive adsorption to the substrates.4, 5
Polyquaternium-10 (PQ-10) is one such cationic polymer, which is a quaternary ammonium derivative of hydroxyethyl cellulose typically used at concentrations of 0.1–5.0%. PQ-10 is used extensively within the personal care industry in hair care products and to a limited extent in lotions and makeup, with a predominant focus on rinse-off cleansing and conditioning applictions.1, 2, 4 The polymer finds use as a conditioner, humectant, viscosity control agent and deposition aid that, as noted, can adsorb onto proteinaceous surfaces such as skin and hair.1, 2, 4, 7
PQ-10 may be incorporated into surfactant-based systems to provide consumer-acceptable conditioning profiles, adjustable visual aesthetics and additional viscosity benefits. It requires solubilization into surfactant-based systems to achieve stability and desired aesthetics. When the surfactant concentration is high enough, micelles form along the polyion chain, solubilizing the polymer into the system and providing a visually clear product.
The conditioning benefits from cationic polymers in shampoos and body washes are achieved through the dilution precipitation mechanism, which occurs during in-use product rinsing. The mechanism of delivery for cationic polymers from surfactant-based systems is generally via coulombic attraction between the anionic functions of the surfactant and the cationic groups on the polymer, which results in precipitation of a charge-neutral complex when the formula is diluted to the point of charge equivalence.7, 15 PQ-10 forms clear films once deposited onto the substrate, and the improvement conferred to hair's appearance has been ascribed to such deposits.
Process and Manufacture
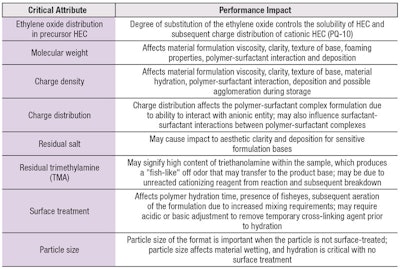
PQ-10 is a cationic derivative of hydroxyethyl cellulose with the precursor being cellulose, a polysaccharide that can be derived from a variety of natural sources such as cotton and wood. In its native form, cellulose is comprised mainly of β-linked D-(1,4)-glucose monosaccharide units. The material is further treated to undergo ethoxylation via reactions with the alkylating reagent ethylene oxide and with heat and alkaline conditions to produce a water-soluble cellulose derivative known as hydroxyethyl cellulose (HEC).15, 17 The ethoxylation of the original cellulose reduces the inherent crystallinity of the structure and enables water solubility to facilitate additional derivation of the backbone during the quaternization process. Differences in the degree of substitution (DS) for the HEC may affect the homogeneity of the charge distribution during the quaternization of the backbone.18
Quaternization of the backbone may be conducted using either the chlorohydrin compound 3-chloro-2-hydroxypropyltrimethylammonium chloride (CHPTMAC) or its epoxide analog, 2,3-epoxypropyltrimethylammonium chloride (EPTMAC).19, 20 CHPTMAC and EPTMAC are cationizing reagents that, in the presence of a base catalyst, react with the available hydroxyl (-OH) pendant functional groups on the cellulose backbone to create cationic hydroxyethyl cellulose, as seen in Figure 1.20, 21 Placement of the charge and distance from the cellulosic backbone affect the polymer-surfactant interactions and flocculation of the material.22 Depending on the distribution of the charge density along the backbone, there may also be differences in performance of the final product.
A heterogenous reaction is often conducted, in which the hydroxyethyl cellulose is dispersed in a water and organic solvent slurry mixture. The introduction of organic solvents such as isopropyl alcohol reduce potential side reactions, such as hydrolysis of EPTMAC to create cationic glycols (e.g 2,3-dihydroxypropyl-N,N,N-trimethylammonium chloride). The organic solvent has the additional benefit of controlling the amount of swelling of HEC during the reaction, allowing for higher reaction temperatures, which can shorten reaction times compared with aqueous reactions.19, 20
Both NaOH and CHPTMAC are added to the slurry, converting the CHPTMAC to the reactive epoxide EPTMAC; the NaOH also catalyzes the etherification reaction by deprotonating the HEC (−OH) groups on the anhydrous glucose repeat units to form alkoxide ions, which react with the EPTMAC epoxide ring to yield the hydroxypropyltrimonium ethers.19, 20
The final step of PQ-10 production is purification, in which PQ-10 is filtered and washed several times to remove unreacted cationizing reagent, organic solvent, nitrogen, NaCl and other impurities. Prior to washing and filtration, the cationic HEC polymers are usually cross-linked with pH-responsive cross-linking reagents, such as sodium borate at alkaline pH or glyoxal at acidic pH.34 The cross-linking step is particularly important for end use to prevent clumping during long-term material storage, polymer hydration and formulating.23, 35
The washed cationic HEC polymers are then dried and milled to yield PQ-10 as a fine powder. Depending on the washing, filtration and product drying, there may be a moderate amine odor based on residual trimethyl amine (TMA) from the cationizing reagent trapped in the powder.1
PQ-10's flexibility allows formulations to be customized to the current needs of the market.
PQ-10 Properties
PQ-10 is considered seminatural, with portions of the structure derived from renewable resources, i.e., cellulose. However, the polymer is synthetically modified to create a cationic polysaccharide comprised of a nonionic cellulose backbone functionalized through reaction with EPTMAC.1 PQ-10 is soluble in water and insoluble in alcohol and nonpolar organic solvents. The material is also stable over a wide range pH (4–10) and is generally provided in a powdered preservative-free format.1 The cationic DS for personal care grade PQ-10 generally ranges from 0.7–1.25 meq/g.8, 9 The cationic charge density may also be reported in terms of nitrogen content (% N), which typically ranges from 0.8–2.2% N.4, 8, 11
PQ-10 usually exhibits medium to high molecular weight (MW) values ranging from 4.0 × 105 g/mol to 9.0 × 105 g/mol; however, lower MW versions (2.5 ×105 g/mol) are available. Also, based on variations in the reaction conditions, oxidatively and/or enzymatically degraded PQ-10 are available.12-16 Performance attributes and product compatibility may vary depending on the method of manufacture.
Typical grades for PQ-10 are low, medium and high molecular weight ranges, with high or low charge densities. Several viscosity grades are offered depending on the molecular weight and level of cationic substitutions.2 Note that PQ-10 from two different grades could lead to performance differences in apparently similar products. The residuals, molecular weight, charge distribution and charge density of the PQ-10 will also affect the final product clarity, viscosity and deposition within surfactant-based systems due to polymer-surfactant interactions. The distribution of the charge density on the cellulosic backbone may also lead to inconsistent performance or differences in product turbidity. The critical attributes for PQ-10 are summarized in Table 1.
Deposition Mechanism and Structure/Function
When PQ-10 is incorporated into a surfactant-based system, anionic surfactant monomers will bind to the cationic sites on the backbone of the structure, creating a polymer-surfactant complex that solubilizes at high surfactant loads (see Figure 2).15 Depending on the molecular weight, charge distribution and density, the polymer-surfactant interactions and surfactant-surfactant interactions will affect product clarity, viscosity and deposition.15
The mechanism of conditioning from the system depends upon the reformation and phase separation of this polymer-surfactant complex during rinsing. Oppositely charged surfactant ions interact by binding with charged sites along the backbone of the polymer.7 Once the solubilized complex is diluted near the isoelectric point—i.e., the point where the concentration of cationic and anionic charges is equivalent, then phase separation, also known as coacervation or the “Lochhead Effect,” occurs. PQ-10 is delivered in this way to provide conditioning benefits for hair and skin.
Broad performance attributes, formulation flexibility and derivation from renewable resources make PQ-10 an industry staple.
However, when PQ-10 is in a system delivering insoluble ingredients, coacervation provides a targeted system to impart both PQ-10 and the other ingredients onto the substrate during rinsing.2, 4, 6, 10, 24 Depending on the application, these ingredients may include triglycerides, silicones, zinc pyrithione, etc.
The properties, delivery and performance of PQ-10 may be adjusted by the structure of the polymer. Parameters such as molecular weight and polymeric charge density may be tuned to affect the deposition and conditioning performance of the product. For instance, as stated, higher charge density and higher molecular weight polymers require increased surfactant loads to solubilize the polymer-surfactant complex and achieve clarity and stability; however, insufficient levels may cause turbidity or precipitation prior to product dilution.25, 26
The increased surfactant binding on the backbone of the higher charged polymer will also increase the viscosity of the product through aggregation of surfactant monomer on the polyion. The binding of surfactant on the polyion effectively shifts the point at which critical micelle concentration would be achieved.27 There may also be an increase in coacervate produced and a shift in its deposition to later stages in the rinsing process.
Conversely, lower molecular weight polymers and charge densities can be less substantive due to reduced polymer-surfactant interactions.25 Lower molecular weight polymers generally have less impact on the formulation viscosity and require lower surfactant loading to achieve polymer-surfactant complex solubility and formulation clarity.28
PQ-10 performance also may be influenced by the ingredient type and concentrations used in the formula. Non-ethoxylated surfactants such as sodium lauryl sulfate may have an inhibiting effect on PQ-10 coacervate formation and deposition performance, compared with ethoxylated alternatives such as sodium laureth sulfate. This is due to solubility differences between surfactant monomers affecting polymer-surfactant interactions.26
Alkyl ether carboxylic acid salts have been found to increase PQ-10 coacervation from systems.24 Adjusting the type of anionic surfactant or addition of amphoteric surfactant content may also aid in increasing the total amount of PQ-10 coacervated through increased hemimicelle packing onto charged polymer sites. Nonionic ingredients may be another option to enhance the delivery of PQ-10 and actives. Further, high molecular weight nonionic polyethylene oxide may enable a more uniform flocculation of the complex by reducing agglomerates and promoting the more even deposition of the active and polymer
coating.26, 29
The order of addition is also a critical factor when formulating with PQ-10 from different sources. As stated, there may be differences in molecular weight and charge density, which could cause precipitation during manufacturing or a hazy final product due strong polymer-surfactant interactions. This can be mitigated by the adding NaCl or amphoteric surfactant prior to adding the anionic surfactant, which allows screening of the cationic charge from anionic, in turn mitigating complexation. However, doing so may impact coacervate deposition, as this increases the concentration of hemimicelles on the backbone and can increase solution viscosity.24, 26, 30
Finally, other parameters that may influence coacervate formation include concentration, ionic strength of the solution, change density of the interacting components, pH and
temperature.31, 32, 33
PQ-10 can be effectively formulated in synergy with a range of materials.
Technology and Applications
When properly formulated, PQ-10 can offer consumer-acceptable conditioning for hair and skin, enhanced delivery of actives and visually clear surfactant-based formulations—among other benefits—and may also impart viscosity to the final product. PQ-10 of high charge density also forms solid-like gels over a limited concentration range, whereas low charge-density species form a liquid-like gel over a much broader concentration range in surfactant-based systems, which allows for a range of final product viscosities and rheologies.35
The polymer is water soluble however may reach the limits of compatibility for systems using high levels of alcohol due to insolubility in nonpolar organic solvents. The various grades of PQ-10 enable formulators to enhance the conditioning and delivery benefits for PQ-10 for specific surfactant systems and applications. In addition, adjustments for the surfactant ratio and remainder of ingredients allow further enhancement of inherent benefits of PQ-10 delivery and product attributes.2, 11 This flexibility allows formulations to be customized to the current needs of the market.
PQ-10 can be effectively formulated in synergy with a range of materials, not limited to anionic and amphoteric surfactants, nonionic ingredients and added electrolytes to meet consumer acceptable conditioning, viscosity, aesthetics and deposition. The polymer is also derived from renewable resources, which is a desirable feature.1 The robust nature of this ingredient, effective conditioning and tangible benefits for active delivery have an advantage for use in personal care products. Thus, due to the broad performance attributes, formulation flexibility and renewable sourcing, PQ-10 continues to be an industry staple.1-3
References
- Final report on the safety assessment of Polyquaternium-10, International Journal of Toxicology 7(Supplement 3) 335–351 (1988)
- msdssearch.dow.com/PublishedLiteratureDOWCOM/dh_096d/0901b8038096db77.pdf?filepath=productsafety/pdfs/noreg/233-00333.pdf
- lubrizol.com/Personal-Care/Products/Product-Finder/Products-Data/280
- korea.in-cosmetics.com/__novadocuments/226508?v=635937864279730000
- AO Barel, HI Maibach and M Paye, Handbook of Cosmetic Science and Technology, Third Edition, pp 364–368 (2010)
- R Lochead, The Role of Polymers in Cosmetics: Recent Trends, American Chemical Society Symposium Series (2007) pp 1–56
- Evaluation of Novel Synthetic Conditioning Polymers for Shampoo, Journal of Cosmetic Science, 60 239–250 (2009)
- US Pat 6451300, Anti-dandruff and conditioning shampoos containing polyalkylene glycols and cationic polymers, DS Dunlop et al., assigned to The Proctor and Gamble Corporation (Sept. 17, 2002)
- US Pat 9040474B2, Granulated Foam Composition Compromising a Polyol Ester and Cationic Conditioning Polymer, D Creutz et al., assigned to Dow Corning Corporation (May 26, 2015)
- US Pat 6451300, Anti-dandruff and conditioning shampoos containing polyakylene glycols and cationic polymers, DS Dunlop, SM Guskey, VE Leyba and DA Royce, assigned to Procter & Gamble (Sep, 17 2002)
- univar.com/~/media/PDFs/US%20Corp%20Region%20PDFs/PC/Dow%20UCARE%20Polymers%20from%20Univar.ashx
- US Pat 3391135, Process for the Manufacture of Low Molecular Weight Cellulose Derivatives, O Shigeru et al., assigned to Shin-Etsu Chemical Company (July 2, 1968)
- US Pat 3709876, Water soluble hydroxyethyl methyl cellulose ether thickener for latex paint, R Glomski, L Davis, J Glover, assigned to Dow Chemical Co (January 9, 1973)
- WO Pat 2003105793A2, Composition containing a cationic polymer with a high charge density and a conditioning agent, NW Geary, ES Johnson, assigned to The Procter & Gamble Company (March 11, 2004)
- JV Gruber, Polysaccharide Based Polymers in Cosmetics, in Principles of Polymer Science and Technology in Cosmetics and Personal Care, CRC Press, London (1999) pp 130–368
- JL Amos et al., Effect of Hydrophobic Substitution on Conditioning Polymers, American Chemical Society Symposium Series (2007) pp 59–71
- dowconstructionchemicals.com/na/en/pdfs/832-00226.pdf
- D Satas and AA Traction, Coatings Technology Handbook, 2nd Edition, CRC Press (2001) pp 547–554
- quab.com/files/Personal_Care_Article.pdf
- quab.com/docs/QUAB_Brochure.pdf
- cosmeticsandtoiletries.com/formulating/function/feelenhancer/premium-Guar-Hydroxypropyltrimonium-Chloride-199604791.html
- K Esumi, Bridging Of Aqueous Polyelectrolytes And Other Complex Systems, in Polymer Interfaces and Emulsions, Marcel Dekker, New York, NY (1999) pp 284–286
- US9643031, Dispersible cationic polygalactomannan polymers for use in personal care and household care applications, AN Chan et al., assigned to Hercules LLC (Nov. 2, 2007)
- kaochemicals-eu.com/sites/default/files/related_files/business_unit/Akypo%20Coacervate%20ENG%20A4%20%282010-03%29.pdf
- L Wing and SLP Jordan, Cationic Cellulosic Polymers with Multifunctional and Outstanding Performance for Personal Care, in Cosmetics and Toiletries Manufacture Worldwide (2003)
- ulprospector.com/documents/1130117.pdf?bs=76&b=204357&st=20&r=na&ind=personalcare
- JCT Kwak, Polymer-Surfactant Systems, Volume 77 (1998) pp 1–4
- XJ Loh, Polymers for Personal Care Products and Cosmetics, Royal Society of Chemistry (2016) pp 78–84
- J Amos et al., Synergistic effects of high molecular weight polyethylene oxide (PEO) and cationic cellulosic polymers on conditioning properties of hair care products, J Cosmet Sci 55(1) 123–124 (2004)
- B Kopiczak and Z Matras, The Effect of Surfactant and High Molecular Weight Polymer Addition on Pressure Drop Reduction in Pipe Flow, Brazilian Journal of Chemical Engineering (33) 933–943 (2016)
- J Caelles, Anionic and cationic compounds in mixed systems, Cosmetics & Toiletries 106(4) (1991)
- CJ van Oss, Coacervation, complex coacervation and flocculation, J Dispersion Sci Technol (9) 561 (1988–1989)
- F Burgess, Practical analysis of complex coacervate systems, J Colloid Interface Sci (40) 227 (1990)
- msdssearch.dow.com/PublishedLiteratureDOWCOM/dh_0981/0901b8038098195b.pdf?filepath=rds/RDS_00084959.pdf&fromPage=GetDoc
- cosmeticsandtoiletries.com/formulating/function/surfactant/a-brief-review-of-polymer-surfactant-interaction-290085341.html