For the complete article, click through to the October 2019 digital magazine.
The link between a healthy scalp and hair quality is now well-established.1 Poor scalp health impacts hair follicle viability, and exposure to external factors such as UV radiation and pollutants can weaken scalp skin health and cause aging. The strong anchoring of hair in the follicle, however, relies on a strong basement membrane and the presence of collagen IV—the binding protein that makes up the primary structural component of the dermal-epidermal junction (DEJ), and the most important component in follicle anchoring.2
Soluble keratin proteins have been shown in vitro to increase the ability of skin cells to produce collagen IV and, as a result, improve the integrity of the DEJ.3 Furthermore, they have been used in the development of dermatological treatments for the clinical management of wounds and the severe skin blistering disorder epidermolysis bullosa, in which the basement membrane is weakened due to a reduced expression of collagen IV.4
In addition, soluble keratin peptides are known to improve skin hydration and improve barrier function, particularly when the skin is challenged.5 A combination of these materials was therefore deemed worth considering for applications to treat scalp skin characterized by poor hydration, inflammation and, ultimately, hair fall through the poor anchoring of hair follicles. This was the focus of the present study.
Previous literature has identified, via in vitro studies of balding dermal papilla cells, the contribution of pollution and cigarette smoke to hair fall.6 Also, in France, in a study7 of 1,011 individuals, a significant 44.2% declared they suffered from a sensitive scalp, which were associated with triggers including pollution, heat, emotions and shampoos. The subjects with sensitive scalp were also significantly more likely to suffer hair loss.
Symptoms of “sensitive scalp syndrome” (SSS) include itching and prickling in the scalp, dandruff, oily scalp and pain in the hair roots. In fact, SSS has been reported to arise from exposure to increasing levels of air pollution including particulate matter, smoke, heavy metals and gases, e.g., SO2, NO2 and ammonia, that settle on the hair and scalp.8 Pollutants also can migrate into the dermis and through the hair follicle, leading to oxidative stress and hair fall.
The role that soluble keratin proteins can play in protecting hair from the oxidative effects of pollutants, specifically SO2 and cigarette smoke, has been well-documented.9 However, the question of how this protective effect impacts hair follicle health has not previously been addressed.
For one, studying follicle health requires overcoming the unique challenges of maintaining a living follicle while also allowing controlled exposure to actives and environments that represent real world conditions. Model systems in which skin structures and follicle systems are created artificially offer some promise.10 However, recent developments in donor explant methodologies have progressed to a point to allow for the maintenance of live follicles in culture conditions, facilitating the controlled study of follicle development and health in a range of environments.11
The present article outlines a study performed by an independent testing laboratorya to investigate the impact of adding keratin actives to live explants of scalp skin containing follicles. Various tests, described next, were performed to determine the effects of a specific keratin active blendb in terms of anchoring follicles and protection against damaged associated with urban pollution and cigarette smoke.
The soluble keratin extracts used are isolated using proprietary processes to control protein molecular weight and cysteine availability. The blend of oxidized keratin, keratin and hydrolyzed keratin maintains key structural characteristics from the native keratin sources to ensure the bioactivity of the resulting ingredient.
Materials and Methods
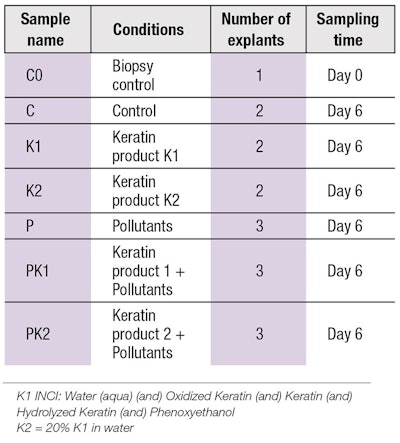
Scalp explants: Live scalp skin explants were harvested from a 58-year-old woman undergoing a facelift procedure. Sixteen shaved explants measuring 0.4 × 1.0 cm were stored in survival medium at 37°C in a humid, 5% CO2 atmosphere. The experiment was conducted for seven days; Day 0 to Day 6. Day 0 involved sampling and product application. Keratin actives were re-applied on days 2 and 5, prior to pollution exposure. Pollution exposure occurred on Day 5 and sampling for analysis was conducted on Day 6.
Keratin actives were applied at a level of 1 μL per explant and spread using a small spatula. Half the volume (0.3 mL per well) was renewed on days 2 and 5. The explants were distributed into seven batches, as summarized in Table 1.
Pollutant exposure: Explants chosen for pollution exposure, i.e., samples P, PK1, PK2 in Table 1, were placed in wells with 900 μL of Hanks’ balanced salt solution (HBSS) and exposed to a mixture of gases using the Air World Simulator Platform for 2 hr. Pollutants and levels included: SAE Dust 100 mg/m3, NO2 17 ppm, SO2 8 ppm and CO 833 ppm. Following this, the explants were exposed to exhaust gases generated with a diesel generatorc for 1 hr; the exhaust composition was as follows: PM1 500 μg/m3, NO 3.96 ppm, NO2 0.84 ppm and CO 5.8 ppm.
The explants P, PK1 and PK2 were then placed in wells with 900 μL HBSS and exposed to smoke from two cigarettes for 1 hrd. The control (nonpollution) explants C, K1 and K2 were kept in 1 mL of HBSS during this time. At the end of pollutants exposure, all explants were placed back in 600 μL of survival medium. . .
Continue reading in the October 2019 digital magazine...