Editor’s note: This is the first of a three-part series that discusses skin protectants. Here, the practical challenges to skin protection efficacy are discussed. Part two, in the November 2014 C&T, reviewed theoretical aspects of skin protection and introduce insights in the area of perfluoropolyethers. Finally, part three will explore a solution based on the concept of shielding skin.
Many have likely observed a similar demonstration of the following: At an exhibition, visitors are impressed by the performance of an individual who immerses one hand, pre-treated with a skin protectant, into a diluted hydrochloric acid solution—apparently without suffering any negative consequences. This sort of test was popular years ago when foam skin protectants were used in various service activities and industrial sectors, informally earning such products the classification of “invisible gloves.”
Such demonstrations raise several questions. What exactly does it mean to protect the skin? How should an effective skin protectant work? How can its effectiveness be measured? How is it regulated? What are the obstacles to innovation in this area? These and other issues concerning skin protectants will be reviewed in this article. Follow-up articles in November 2014 and May 2015 will introduce an idea to improve their efficacy and explore this new idea in greater depth.
Skin Exposure Complexities
In order to illustrate the complexity of skin protectants, it is necessary to consider three different exposure situations and their consequences: First, a single-incident contact of the skin with a strong irritant, which causes an almost immediate injury that may heal within a few days; next, limited contact of the skin, i.e., a few times, with a weak irritant, which usually is harmless (or appears to be); and, lastly, the combination of repeated skin contact over time with weak or substances deemed as potentially irritating, in addition to internal or external adverse conditions. This latter combination may have severe consequences. While some form of dermatitis generally results from each of these occurrences, this last situation is a chronic pathology, usually of the hands, that takes weeks, months or even years to develop and requires a comparable length of time to heal. Only in this latter case, i.e., to prevent pathology, are protective products used, typically in the form of creams or lotions.
Maintaining good skin health clearly is important, since the primary function of skin is to protect the human body against physical, chemical and microbiological insults. A first skin defense, although weak, is represented by a hydrolipidic film composed of an emulsion of sweat and sebum. A more important defense is the stratum corneum (SC), which also prevents uncontrolled water loss. The moisture condition of the SC influences the skin’s defense as well. Therefore, moisturization is a means to improve it.
Subjective and environmental factors, often in combination with mechanical trauma, may facilitate the infiltration and action of noxious substances, and repeated skin exposure to these conditions produce dryness, cracking, fissuring, hyperkeratosis, premature aging and, ultimately, chronic irritant dermatitis. This diagnosis is clinical and based on an individual’s history of exposures to known substances, as well as on negative patch test results to exclude allergic dermatitis.
Dermatitis
Atopic dermatitis, often referred to as eczema, is a general term used to describe differentiated inflammatory skin conditions; a simplified classification is shown in Figure 1. The origins of these pathologies can be exogenous or endogenous. Exogenous dermatitis is generated by contact with a substance, hence contact dermatitis (CD) is its common denomination.
There are two main types of CD. Irritant contact dermatitis (ICD), localized in the area of contact, is caused by irritants such as acids, alkalis, solvents, surfactants, lubricants, powders, etc. Contact with the irritant is necessary to produce ICD, but the probability and the severity of the reaction depend on individual factors and environmental conditions (see Figure 2). Many lists of occupations at risk for ICD have been published, although they are not exhaustive. ICD of the hands occurs most often in those whose hands are frequently washed or are in contact with water/wet materials—including cooks, hair dressers, gardeners, construction workers, mechanics, dentists and medical professionals, to name a few.
The other type of CD, allergic contact dermatitis (ACD), is due to contact with allergens such as salts, metals, fragrances, preservatives, dyes, resins, drugs, pesticides, etc. ACD, diagnosed by patch testing, may occur on any part of the body regardless of the site of contact by the allergen.
Regulatory Implications of Treatments
Beyond the complexities of dermatitis itself, complications in its treatment arise due to U.S. and European regulations. In the United States, skin protectants, often referred to as anti-irritants, are considered drugs, similar to sunscreens, since they are intended for the prevention of disease, i.e., dermatitis. As defined by Section 201 of the U.S. Federal Food, Drug and Cosmetic Act, Monograph 21 C.F.R., Part 347, “Skin protectant drug products [are] for over-the-counter human use.” Therefore, these products must comply with the Monograph, and their manufacture should be in conformance with Current Good Manufacturing Practices (CGMP).
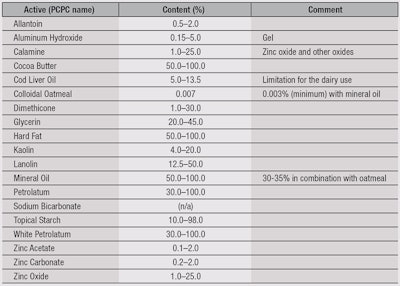
Also similar to drugs, active ingredients should be distinguished from inactive ingredients (i.e., excipients) on the product label; the most recent Monograph contains a list of 19 permitted actives and their concentrations (see Table 1). Products for the lips are included, which explains why some are permitted at such high levels. Note that the Monograph and this list do not imply that skin protectants should be registered and approved by the U.S. Food and Drug Administration (FDA) before marketing.
The positives list includes three groups of actives, and only combinations from within the same group are permitted. In particular, allantoin, cocoa butter, cod liver oil, dimethicone, glycerin, hard fat, lanolin, mineral oil, petrolatum and white petrolatum can be combined with one other. Besides these combinations, actives can be combined with actives from other positive lists, such as analgesics and sunscreens.
This author believes that classifying these active ingredients as drugs is incorrect and inappropriate. And while this positives list for permitted skin protectants exists, these ingredients alone do not provide skin protection; the formulation as a whole should provide the desired benefit. The overall scope of the FDA Monograph is that “an over-the-counter skin protectant drug product, in a form suitable for topical administration, [be] generally recognized as safe and effective.” While the scope for safety is certainly achieved, there are reasonable doubts concerning effectiveness.
First, the positives list appears to include no innovative chemistries; in particular polymer chemistries. There is only one synthetic polymer—dimethicone, also known as polydimethylsiloxane—and it represents the first generation of silicon-based polymers developed in the 1950s. Ironically, and not surprisingly, many companies prefer not to use the claim of “skin protectant” or even exploit the claim of “silicone-free” for products containing dimethicone. This may be due to consumers’ negative impression of silicone oils. Thus, companies are left to either develop an effective product using ingredients that are not included in the positives list, which will not support skin protection claims, or to use dimethicone for its known efficacy and face potential negative consumer perception. (As an aside, dimethicone is a rather common ingredient, appreciated for its safety and sensorial profile.)
It is also important to note that although the United States treats skin protectants as drugs, these actives are not necessarily superior to similar products in Europe and in other regions, where they are classified as cosmetics. Outside of the United States, the working mechanism of these products and conditions of the skin are the primary considerations when determining their regulation. Another point to notice, in both the United States and Europe, there appears to be no remarkable difference in performance between products distributed through special channels in professional and industrial sectors and products sold in stores as cosmetics. The fact that it is possible to have different claims without different performance demonstrates a shortcoming of the regulations. (Editor’s note: for more on the FDA and skin protectant claims, read the “FDA Sidebar” accompanying this article.)
This is further highlighted by “invisible glove” products, which had similar commercial success in the United States and in Europe. On the one hand, more stringent regulations make no guarantee for better performance (and it is difficult to believe the situation would be better in less severely regulated markets); on the other hand, without regulation, skin protectants are cosmetics historically, technically and by logical market identification, so why include effective, more expensive ingredients in products with potential efficacy claims that would be no better than poorly performing products? It is difficult to say which is better (or worse). It seems that changes are necessary, in both the United States and Europe, favoring greater uniformity and revising the definition of what constitutes an “active.”
Evaluation Protocols
Test methods to evaluate and compare skin protectants also are a serious and unresolved problem. In a laboratory, it is practically impossible to reproduce the real life conditions that cause ICD. This situation is different for other types of skin protectants, e.g., sunscreens, whose UV filters can be evaluated for efficacy via in vitro and in vivo methods. Simple methods to test the efficacy of skin protectants have been proposed since the 1950s1 and involve submitting the skin to different markers—i.e., indicator reagents, dyes or irritants with and without the pre-application of a protective product for 5-60 min, depending on the nature of the marker. In these cases, each experiment can be regarded as an acute exposure to a well-defined penetrating substance, often high in concentration. This type of investigation is generally demanding, as far as the performance of the skin protectant, and it has poor value since, as stated, it does not successfully reproduce real life conditions.
To compare skin protectants against a single water-soluble irritant such as sodium lauryl sulfate, the hand immersion test is often used. In it, volunteers immerse their hands for a few minutes in an aqueous solution containing the irritant. Before immersion, like the “invisible glove” test, one hand is treated with a product while the other hand serves as the control. The protective properties are evaluated by measurements of trans-epidermal water loss (TEWL), Laser Doppler Flowmetry (LDF) or other bioengineering techniques. These protocols involve various levels of complexity including repeated applications on volunteers, internal and external controls and statistical analyses, but usually are conceived to evaluate skin protectants against a single irritant, thus conclusions regarding broader protection cannot be reached.
In an effort to create a test that better simulates real life factors, Frosch developed a test method applied first to guinea pigs then to human volunteers.2-5 This method involved nine separate applications of four standard irritants: 1% sodium hydroxide, 10% sodium lauryl sulfate, 30% lactic acid and pure toluene. One application consisted of an occlusive patch test for 30 min, and these applications were carried out for two weeks. Quantification of the irritation was measured by TEWL; evaluation of erythema was determined by visual scoring, from 0 = none, to 5 = very severe. The protocol of Frosch was modified by Elsner, who used more diluted irritants, such as 0.5% sodium hydroxide, 5% sodium lauryl sulfate, 15% lactic acid, in addition to pure toluene. Elsner also used test sites on the mid-backs of the 20 volunteers, instead of their forearms.6
Again to emphasize, alternative to standard methods, tests that better simulate real conditions should be conducted. Consider that rather than treating healthy skin with irritants to evaluate protection, it is more convenient to treat dermatis-afflicted patients to evaluate regression of their condition. Further, these volunteers may continue their normal daily routines under the conditions that produced the dermatitis. In relation, investigations of chronic ICD regression of volunteers treated with skin protectants assume the prevention and treatment of dermatitis address the same issue. These test protocols, conceived for homogeneous groups, should be adapted to specific characteristics and conditions of the volunteers and their environment. A good moisturizer for a German woman is not the ideal for an African woman, for example.
At any rate, neither the test methods or investigations using volunteers can be adopted routinely for screening experimental products, development work or for comparing commercial products. On the whole, however, these methods and investigations have helped to elucidate some theoretical aspects of skin protection, which will be described in part two of this series.
References
- RR Suskind, The present status of silicone protective creams, Indust Med Surg 24 413-416 (1955)
- PJ Frosch et al, Efficacy of skin barrier creams: The repetitive irritation test (RIT) in guinea pig, Contact Dermatitis 28 94-100 (1993)
- PJ Frosch et al, Efficacy of skin barrier creams: Ineffectiveness of a popular skin protector against various irritants in guinea pig, Contact Dermatitis 29 74-77 (1993)
- PJ Frosch, A Kurte and B Pilz, Efficacy of skin barrier creams: The repetitive irritation test (RIT) in humans, Contact Dermatitis 29 113-118 (1994)
- PJ Frosch and A Kurte, Efficacy of skin barrier creams: The repetitive irritation test (RIT) with a set of four standard irritants, Contact Dermatitis 31 161-168 (1994)
- P Elsner, W Wigger-Alberti and G Pantini, Perfluoropolyethers in the prevention of irritant contact dermatitis, Dermatology 197(2) 141-145 (1998)