Human skin, more specifically facial skin, periodically needs a deep cleansing to remove not only the oily particles resulting from secretions, but also dead skin caused by desquamation of the epidermis.1 Cleansers are designed to remove dirt, sweat, sebum and oils from the skin, which helps to promote normal exfoliation and thereby rejuvenates the skin.2 However, the use of cleansers can lead to a reduction in the level of the natural moisturizing factor (NMF) of skin.3 Factors that reduce the water content can lead to changes in skin´s viscoelasticity.4 Further, harsh cleansers such as soaps can induce dryness, leading to scaly and rough skin.5 These effects may be much more severe during winter months when the air is cold and dry.
Visible skin dryness has been found to correlate positively with a lack of surface hydration.2 Minimizing damage caused by cleansers is therefore the first step toward cleansing without compromise. The second step is compensating for the level of damage caused by cleansers by providing moisturizing benefits during washing.2 By moisturizing skin, optimal levels of hydration and plasticization are maintained, allowing skin to retain its normal viscoelasticity.6
Current technologies that compensate for cleanser damage and provide benefits include those that deposit oils, lipids and humectants during washing.2 The pH is also an important factor to maintain both the structure and function of the stratum corneum, as any small and sustained pH increase adversely influences the barrier repair mechanism.7 In addition to moisturizing skin and maintaining pH, exfoliation is an important aspect to cleansing. It involves the removal of “old” skin to speed up the renewal of “young,” new skin. To provide exfoliation, compositions have been proposed that contain abrasives and/or insoluble abrasive particles of an appropriate size and shape; for example, finely ground quartz, polyethylene beads, aluminium oxide and ground fruit pits. After being used on skin, these substances are removed by wiping or rinsing with water. Such compositions, despite being particularly effective, can be highly irritating8 due to the abrasion mechanism itself. This may be more or less pronounced depending on the type of abrasive substance, the particle size and shape, the formulation and the skin condition. Abrasive solid particles should therefore be of a particle size appropriate to exfoliate in a nonirritating manner. In addition, exfoliation of the skin should only be performed by consumers one to two times per week, depending on skin type.
Pumice stone has been used for centuries to remove rough and hardened skin, particularly on the feet. Pumice is a frothy volcanic rock formed during explosive volcanic eruptions of acidic and highly viscous magma. Bubble-shaped openings, i.e., vesicles, are formed in it by the expansion of entrapped gases, thus it has a very low density and in some cases, floats on water. This fragile, natural acicular glass consists of 60–75% silica content and minerals; in particular, feldspars. Pumice ground into micro-fine grains provides an excellent exfoliating medium.9, 10
Based on its known use in raw form, i.e., rock, to exfoliate the feet, the authors sought to evaluate different particle sizes of pumice for its effects on formula texture and rheology, as well as its potential for use elsewhere on the body. This natural source exists in abundance in the Azores archipelago islands of volcanic origin, and can be obtained at low cost. Pumice-containing soaps and gels were developed, some of which were optimized for cutaneous application, stability, rheology, texture, pH, etc. Optimizing parameters such as rheology allows for easy application of the formulation over the skin, and gives the product formulators an idea of the force necessary to spread the product uniformly over the skin. The exfoliating efficacy was then tested for the optimized soap and gel formulas containing pumice particles between 125–250 µm in size, which provide abrasive action but are appropriate for the face, where the skin is thinner.
Materials and Methods
Materials: In addition to pumice, sampled from Sabreira do Pedregal, Pisão in São Miguel’s island, Azores archipelago, test formulations incorporated perfluorocarbon (PFC) gel, aloe vera and imidazolidinyl ureaa, in addition to stearic acid, glycerine and triethanolamineb, and propylene glycolc. PFC is a type of carbomer or carboxyvinyl polymer that has the ability to gel water at a neutral pH. Since this excipient shows acid characteristics, it was necessary to add a base, in this case triethanolamine, until pH 7.11
Formula preparation: Compositions of both a gel and soap are shown in Formulas ">1 and ">2 on Page 782. For the gel preparation, imidazolidinyl urea, a preservative, was dispersed in water; the emollients and moisturizers propylene glycol and aloe vera were then added. PFC was dispersed into the mixture with a mechanical stirrer and neutralized with triethanolamine. The pumice was added next, as well as color and fragrance compounds. For textural and rheological studies, additional preparations included a PFC gel base; gel base formulations with 5% w/w pumice of grain sizes > 500 µm (GP 500), 250–500 µm (GP 250) and 125–250 µm (GP 125); and optimized gels with pumice having grain sizes of 250–500 µm (EP 250) and 125–250 µm (EP 125).
Soap was prepared by a saponification reaction of stearic acid with 38% w/v sodium hydroxide solution. The mixture of stearic acid and propylene glycol was heated at 70°C until the stearic acid melted. The mixture of glycerine with 38% w/v NaOH solution was heated to the same temperature, then the hydrophilic mixture was added slowly to the oil mixture under constant stirring, until homogenous. The preparation was stirred until it reached 50°C and the previously colored and scented pumice was added; this preparation was poured into appropriate molds.
Texture, rheology analysis: Texture analysis for quality control purposes has significantly increased in the pharmaceuticals field over the past several years.12 One test used to assess textural properties is the penetration test, whereby an analytical probe is depressed into the sample at a defined rate and to a desired depth. From the resultant force-distance curve, the mechanical parameters of firmness and adhesiveness may be derived. Firmness is defined as the force necessary to attain a given deformation, and adhesiveness is regarded as the work necessary to overcome the attractive forces between the surfaces of the sample and of the probe with which it comes into contact.13, 14
As a review, the study of the flow properties of matter and its deformation are termed rheology, which constitutes an integral branch of physics, physiology and pharmaceutical fields.15 The flow properties of matter influence each step of the pharmaceutical development process, such as filling, mixing, packing and removal from the container, and define its in vivo behavior.16 Depending on their flow properties, materials can be classified as Newtonian and non-Newtonian. Newtonian flow is characterized by a constant viscosity regardless of the applied force, while non-Newtonian flow is characterized by a change in apparent viscosity with increasing shear stress; non-Newtonian types of flow include plastic, reofluidificant and dilatant.17 Reofluidificant behavior is characterized by a decrease in apparent viscosity as stirring is applied to the system. This type of behavior is the most common in dermocosmetic formulations because it allows easy spreading.
Thixotropy is a rheological manifestation in which viscosity changes are reversible and time-dependent. Thixotropy is a term to describe an isothermal system in which the apparent viscosity decreases under shear stress, followed by a gradual recovery when the stress is removed.15, 18 In stability tests, which are usually conducted at constant temperature, the samples are periodically checked for changes in important features. In the present work, the gel formulations were submitted to textural and rheological analyses at 48 hr, 15 days and 30 days after their preparation. These formulations were maintained at 20°C throughout the test period. Textural analysis of the samples was performed in compression mode using a texturometerd by carrying out a penetration test using a load cell of 5 kg, a 13-mm diameter cylindrical probe, a penetration depth of 5 mm at velocities of 3 mm/s, and a trigger force of 0.049 N.
After penetrating the sample, the probe returned to a position 90 mm above the platform surface. From the graphic force versus time obtained, the maximum force (firmness) and the negative area (adhesiveness) were calculated; measurements were performed in triplicate. Rheology tests were performed using a rotational viscometerd with needle number 6. Soap hardness was determined also using a texturometere, with a load cell of 30 kg and a cylindrical probe with a 2 mm-diameter. The pH of the soap also was evaluated from a 1% w/v solution of soap dissolved in neutral water using a pH meterf.
Efficacy measurements: Determinations of skin hydration, pH, lipids and skin relief were performed before and after test formulas EP 125 or SP 125 were applied to the forehead and rubbed circularly 30 times. The same person applied the product in all volunteers, after training, in order to maintain the same pressure. Hydration and pH values as well as the sebum content were determined with multiprobe equipmentg using a corneometerh, skin pH meterj and sebumeterk probes. Skin surface images were obtained with a UVA light video cameram and softwaren specially designed to characterize skin’s relief by different variables (see Skin Characterization Variables on Page 784). In this study, the variables analyzed were: surface, volume, energy (NRJ), scaliness (SEsc), roughness (SEr), wrinkles (SEw) and smoothness (SEsm). Thus, before and after treatment comparisons of the same skin site enabled the comparison of changes in skin condition.19
Test conditions: Tests were conducted in a laboratory environment, at a temperature of around 20°C and mean relative humidity of 60%. A total of seven healthy Caucasian volunteers, three men and four women, ages 20–27 years, were enrolled in the study. All volunteers received oral and written information about the study modalities, including the experimental protocol. Written informed consent was obtained.
In vivo protocol: The volunteers applied the exfoliating preparation, i.e., soap or gel, chosen randomly, in the test area using a circular motion for 30 sec (see ">Figure 1). After application, the product was removed with water and the skin was gently dried with a towel. The formulations used in the efficacy study were EP 125 and the SP 125 due to smaller particle sizes of the pumice, which are less irritating for facial application. Final measurements were made 30 min after application of the product. Statistical analyses were performed with software and differences were considered significant when p < 0.05.
Results and Discussion
Texture and rheology: Figures ">2 and ">3 show the results for firmness and adhesiveness, respectively, determined 48 hr, 15 days and 30 days after preparation and storage at 20°C.
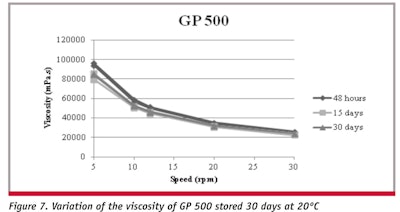
Preparations GP 125, GP 250 and GP 500 showed higher firmness than the respective base. Optimized formulations EP 125 and EP 250 showed less firmness than formulations GP 125 and GP 250 after 15 days. A decrease was found in firmness and stabilization after 30 days at 20°C. Regarding adhesiveness, only GP 125 and EP 125 showed a significant decrease after 15 and 30 days. Figures ">4; ">5 and ">6; ">7 and ">8; and ">9 show the apparent viscosity of all gel formulations, determined 48 hr, 15 days and 30 days after their preparation and storage at 20°C.
Analyses of the graphs of apparent viscosity versus rotational speed show that all gel formulations presented reofluidificant behavior without thixotropy. All formulations presented a high yield value, i.e., required a certain minimum pressure to begin flow, which is characteristic of preparations containing carbomers. This property confers the ability to maintain the uniform dispersion of solid particles, like pumice, without sedimentation.11 EP 125 and EP 250 showed lower apparent viscosities than all the other formulations. Although the gel base and EP 125 presented a decrease of apparent viscosity after 15 days, no changes were found in this parameter after 30 days of storage, like the other formulations studied. The SP 125 test formula showed a hardness value of 7.15 ± 0.38 N and a pH value of 10.25.
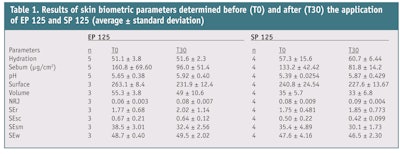
Exfoliant efficacy: With the purpose to demonstrate which formulation shows the greatest exfoliant efficacy, skin biometric measurements were performed. ">Table 1 shows the results and average ± standard deviation of the parameters determined before (T0) and 30 min after (T30) application of the exfoliating products. The biometric parameters that correlate better with exfoliant efficacy, taking into account the obtained results, are sebum, surface and energy. By the analyses of box plot graphs (see Figures ">10, ">11, ">12, ">13, ">14), it can be seen that after exfoliation with the EP 125 gel, a significant improvement in several biometric parameters was observed, such as sebum, surface and energy (NRJ). A decrease in the sebum content was expected because exfoliation removes the lipid content of the stratum corneum. A reduction in surface is also indicative of skin renewal; this parameter relates real topography with the same surface after it is made smoother by stretching, so it is normal to observe a reduced surface after exfoliation. Smoothness (SEsm) decreases as a consequence of cell desquamation promoted by exfoliation. Abrasives are intended to make the skin smooth by mechanical removal of the upper layers of the stratum corneum; however, immediately after treatment, the opposite is observed.20-22
Energy (NRJ) significantly increased after exfoliation. This parameter correlates with the level of moisture on the skin surface; the greater the energy, the more hydrated the skin. After exfoliation with soap SP 125, no significant improvement was observed in any of the biometric parameters evaluated, which means that gel EP 125 showed better exfoliating efficacy.
Conclusions
In order to evaluate different particle sizes of pumice for their effects on formula texture and rheology, as well as their potential use elsewhere on the body, several formulations were developed and tested. Formulations GP 125, EP 125 and SP 125 showed textural and rheological characteristics appropriate for cosmetic use. However, EP125 gel showed the best exfoliation efficacy. This gel formulation containing pumice with particle sizes within the range 125–250 µm allowed for the removal of dirt, sweat, sebum and dead cells without damaging the facial skin, as assessed visually. Therefore, this formulation could be an effective and interesting natural exfoliating product for oily and combination skin.
Acknowledgements: The technical and scientific research described were supported by Instituto de Inovação Tecnológico dos Açores (INOVA) under the project “Preparation of Formulation Development Studies and Applications: Dermocosmetic Pumice and Mud Baths,” funded by the project TERMAZ.
References
- US Patent 5,431,913, Skin cleansing and exfoliant composition and method of treating skin, E J Phillips (1994)
- KP Ananthapadmanabhan, DJ Moore, K Subrqmanyan, M Misra and F Meyer, Cleansing without compromise: The impact of cleansers on the skin barrier and the technology of mild cleansing, Dermatologic Therapy 17, 16–25 (2004)
- C Prottey and T Ferguson, Factors which determine the skin irritation potential of soaps and detergents, J Soc Cosmet Chem 26, 29–46 (1975)
- M Kawai and G Imokawa, The induction of skin tightness by surfactant, J Soc Cosmet Chem 35, 147–156 (1984).
- FA Simion, Human in vivo methods for assessing the irritation potential of cleansing systems, in Surfactants in Cosmetics, MM Riger and LD Rhein, eds, Marcel Dekker Inc, New York (1997) pp 519–532
- W Matthies, Dermatological observations, in Anionic surfactants, C Gloxhuber and K Kunstter, eds, Marcel Dekker Inc, New York (1992)
- JW Fluhr, J Kao, M Jain, SK Ahn, KR Feingold and PM Elias, Generation of free fatty acids from phospholipids regulates stratum corneum acidification and integrity, J Invest Dermatol 117, 52–58 (2001)
- US Patent 4,675,526, Anhydrous skin cleansing composition containing an oil phase, an emulsifying agent and particulate water soluble polymeric abrasive particles, A Zabotto and J-C Contamin, assigned to Societe Anonyme dite: L'Oréal, Paris (1987)
- Cosmetic ingredients, Dermaxime Bio-cellular skin products, available at www.dermaxime.com/pumice.ht (Accessed Dec 3, 2011)
- N Michalun and M V Michalun, Milady’s Skin Care and Cosmetic Ingredients Dictionary, 3rd edn, Cengage Learning, New York (2010)
- LN Prista, AC Alves, R Morgado and J Sousa Lobo, Tecnologia Farmacêutica–II Volume, 5th edn, Fundação Calouste Gulbenkian: Lisboa (2006)
- H Li and X Gu, Correlation between drug dissolution and polymer hydration: A study using texture analysis, Int J Pharm 342, 18–25 (2007)
- IF Almeida and M F Bahia, Evaluation of the physical stability of two oleogels, Int J Pharm 327, 73–77 (2006)
- MC Bourne, Texture profile analysis, Food Technol 32, 62–72 (1978)
- CH Lee, V Moturi and Y Lee, Thixotropic property in pharmaceutical formulation, J Control Release 136, 88–98 (2009)
- HA Barnes, Thixotropy–A review, J Non-Newton Fluid Mech 70, 1–33 (1997)
- LV Allen, NG Popovich and HC Ansel, Formas Farmacêuticas e Sistemas de Libertação de Fármacos, 8th edn, Porto Alegre: Artmed (2007)
- J Mewis and NJ Wagner, Thixotropy, Adv Colloid Interface Sci 147–148, 214–227 (2009)
- E Berardesca, N Cameli, G Primavera and M Carreira, Clinical and instrumental evaluation of skin improvement after treatment with a new 50% pyruvic acid peel, Dermatol Surg 32, 526–531 (2006)
- MRP Ferreira, PC Costa and FM Bahia, Efficacy of anti-wrinkle products in skin surface appearance: A comparative study using non-invasive methods, Skin Res Technol 16, 444–449 (2010)
- M Tan, JM Spencer, LM Pires, J Ajmeri and G Skover, The Evaluation of aluminum oxides crystal microdermabrasion for photodamage, Dermatol Surg 27, 943–949 (2001)
- M Lodén and A Bengtsson, Mechanical removal of the superficial portion of the stratum corneum by a scrub cream: Methods for the objective assessment of the scrubs, J Soc Cosmet Chem 41,111–121 (1990)