Read this article in its entirety in the October 2020 digital edition. . .
Editor’s note: This second part in our series on hand hygiene provides a brief history of its evolution, considers the nature of viruses, explores hand hygiene protocols and describes the formulation of hand sanitizer gels and foams. Future parts will cover the efficacy of hand sanitizers, regulatory aspects, IP and litigation, and more. Follow along in our upcoming November issue. (See Part I and Part III).
Do Hand Sanitizers Work?
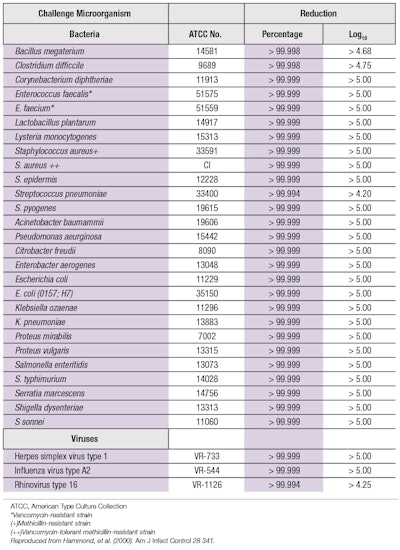
The antimicrobial efficacy of an ethanol-based hand sanitizer is shown in Table 1. Alcohol-based treatments are preferred because they generally act more rapidly than other antiseptics. They also spread rapidly over the skin and quickly evaporate to leave no residuals that could cause the emergence of multi-drug resistant microorganisms.68, 69 Moreover, after the alcohol has evaporated, the resident skin flora can be restored in a relatively short time. Alcohol-based sanitizers can be placed or carried almost anywhere for easy use and availability, which is more convenient than a permanently situated wash basin.
Hand sanitizer gels appeared in the late 20th century. The now-familiar gels were enabled by stimuli-responsive polymersa, carbomer and acrylates C10-30 alkyl acrylate crosspolymer.70-75
These polymers are cross-linked microgels that served to thicken hydroalcoholic solutions and confer shear-thinning rheology for facile dispensing. Upon contact with the hands, salt on the skin’s surface, as previously described, causes the microgels to immediately shrink to squeeze out fluid, sanitizing alcohol to spread and permeate the wrinkles, nooks and crannies on the hands. See this mechanism in action in a Video Demo on Page 39 of your digital edition. Ideally, a hand sanitizer should remove transient flora, especially pathogens; allow the resident skin flora (the microbiome) to recover; and should not compromise the skin barrier function.76
Comparing the performance of plain soap and water with alcohol, it has been observed that washing with the former can physically remove adherent flora but it does not always remove all the pathogens. Moreover, the skin can become irritated by alkaline soap and the resulting lesions can become colonized by microorganisms. Furthermore, Gram-negative bacteria can colonize soap.
On the other hand, alcohol rubs have a long history of successful use in Europe. The U.S. Food and Drug Administration (FDA) lists alcohol rubs as eligible for evaluation under the FDA New Rule for Antiseptic products.77 Alcohol hand rubs have excellent germicidal activity in vitro, including activity against Gram-positive and Gram-negative bacteria, and enveloped lipophilic viruses. The CDC/Healthcare Infection Control Practices Advisory Committee also states that alcohol hand rubs represent the standard of care for hand hygiene.78
The World Health Organization (WHO) has a comprehensive set of guidelines for Hand Hygiene.79 It is a tome, with 1,156 references and to reduce it to a single paragraph does not do it justice. However, I refer to this document to list one relevant set of WHO recommendations; specifically that hand hygiene protocols should:
• Compare the efficacy of products;
• Update the testing protocols and make them economically justifiable;
• Use realistic in vivo test models with appropriate test organisms and choice of subjects;
• Emphasize efficacy requirements rather than limiting goals to what is commercially available; and
• Design clinical studies for equivalence to meet a standard rather than compare products.
The WHO recommends two sanitizing hand rub formulas (see Formulas 1 and 2). It is surprising to see hydrogen peroxide and ethanol and glycerin together in these formulations because the peroxide would be expected to react with the alcohol and glycerin to reduce the efficacy of the formulation. Apparently, hydrogen peroxide and glycerol are included to eliminate bacterial spores during storage and to increase acceptability, and hydrogen peroxide is not an agent for antisepsis in these formulations.
These are non-thickened formulations. The WHO requires pharmacopoeial quality reagents because they are designed for the entire world and there is a need to be wary of unsafe contaminants. It is interesting that the WHO formulations do not meet the bacterial count log reductions in the time required by the European standards; the WHO formulations require 60 sec whereas the European standard mandates no more than 30 sec for the required log reduction of colony-forming units.
The WHO and also the CDC inform that efficacy cannot be judged merely by alcohol content but varies from formula to formula.80 It is reasonable to conclude that the variability could be related to the availability of alcohol to penetrate all the cracks and creases of the hands, and may be reliant on the ability of the formula to release alcohol upon contact with the hands.
Alcohol Hand Rubs and Virucidal Activity
Pathogenic viruses on the hands can remain viable for hours81, 82 and transferral from hands is recognized as a potential mode of direct and indirect spread of infectious diseases.83-85 Frequent hand-washing with water and soap can damage the skin barrier, however, and this can lead to a route of entry for SARS-CoV-2 (COVID -19) since the cell receptor for SARS-CoV-2 entry, angiotensin-converting enzyme 2, is abundant in blood vessels/capillaries of the epidermal basal layer, hair follicles and eccrine glands. There is thus a need to use suitable skin barrier prophylactics and therapeutics to ameliorate the damage caused by frequent hand-washing.86
Alcohol-based hand sanitizers may be less damaging than frequent hand-washing with soap and water. In fact, the CDC’s guideline on hand hygiene documents that alcohol is more effective than soap and water, and that the in vivo antimicrobial activity of alcohol antiseptics shows a fast log reduction, recovery of the microbiome and activity against enveloped viruses—recall that coronaviruses are lipid-enveloped viruses.87 Further studies are needed to find the minimum virucidal activity that interrupts cross-infection, however; in addition, alcohol is not sporicidal.
. . .Read more in the October 2020 digital edition. . .
References
- Lin, Y.-H., Liu, C.-H. and Chiu, Y.-C. (2020). Google searches for the keywords of “wash hands” predict the speed of spread of COVID-19 outbreak in 21 countries. Brain, Behavior and Immunity doi: 10.1016/j.bbi.2020.04.020
- E. Larson (1999). Skin hygiene and infection prevention. Clinical Infectious Diseases 29, 1287-94.
- WHO (2009). WHO guidelines on hand hygiene in health care. Page 6, available at http://whqlibdoc.who.int/publications/2009/9789241597906_eng.pdf?ua=1
- Conservation and Art Materials Encyclopedia Online (CAMEO) (2016). Labarraque’s solution. Available at: http://cameo.mfa.org/wiki/Labarraque%27s_solution
- Wikimedia Commons. (2014). File: A G Barraquw.jpg. Available at: https://commons.wikimedia.org/wiki/File:A_G_Barraque.jpg
- Gregory, T. (1942). The Condensed Chemical Dictionary, 3rd end. Reinhold Publishing, New York.
- Mayer R. (1969 and 1945). A Dictionary of Art Terms and Techniques. Harper and Row Publishers, New York.
- Shaikh, S. (2017, Jul 26). The contagiousness of puerperal fever (1843), by Holmes, O.W. Embryo Project Encyclopedia. Available at: http://embryo.asu.edu/handle/10776/12967
- NPR website. (2017, Oct 13). The Doctor Who Championed Hand-washing and Briefly Saved Lives. Available at: https://www.npr.org/sections/health-shots/2015/01/12/375663920/the-doctor-who-championed-hand-washing-and-saved-women-s-lives10. https://encrytedtbn0.gstatic.com/images?q=tbn%3AANd9GcTecEiKLWETZG8Dtex91AW9z4nGwGeUTQvsEJXIwSECzCtrrKJjt
- https://encrytedtbn0.gstatic.com/images?q=tbn%3AANd9GcTecEiKLWETZG8Dtex91AW9z4nGwGeUTQvsEJXIwSECzCtrrKJjt
- Wellcome Collection (1882). Use of the Lister carbolic spray, Antiseptic surgery, 1882. Attribution 4.0 International (CC BY 4.0). Available at: https://wellcomecollection.org/works/wdp236ft
- Price, P.B. (1938). The bacteriology of normal skin: A new quantitative test applied to a study of the bacterial flora and the disinfectant action of mechanical cleansing. Journal of Infectious Diseases 63(3) 301-318. Available at: https://doi.org/10.1093/infdis/63.3.301
- Boyce, J.M. and Pittet, D. (2002, Oct 25). Guideline for hand hygiene in health care settings. CDC MMWR Report 51, No. RR-16.
- Larson, E. (1999). Skin hygiene and infection prevention: More of the same or different approaches? Clin Infect Dis 29 1287-94.
- Larson, E., McGinley, K.J., Grove, G.L., Leyden, J.J. and Talbot, G.H. (1986). Physiologic, microbiologic and seasonal effects of hand-washing on the skin of health care personnel. Am J Infect Control 14 51-9.
- Ojajarvi, J., Makela, P. and Rantsalo, I. (1977). Failure of hand disinfection with frequent hand-washing: A need for prolonged field studies. J Hyg (Lond) 79 107-19.
- Mackintosh, C.A. (1983). Skin scales and microbial contamination. In: Marks, R. and Plewig, G., eds. Stratum Corneum. Berlin, Springer-Verlag 202-7.9.
- Larson, E., Friedman, C., Cohran, J., Treston-Aurand, J. and Green, S. (1997). Prevalence and correlates of skin damage on hands of nurses. Heart Lung 26 404-12.10.
- Meers, P.D. (1980). The shedding of bacteria and skin squames after handwashing. In: Newsom, S.W.B. and Caldwell, A.D.S., eds. Problems in the Control of Hospital Infection. London, Royal Society of Medicine 13-8; International Congress and Symposium Series vol 23.
- Centers for Disease Control and Prevention (2019). Antibiotic resistance threats in the United States 2019. Available at https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf
- Webb, G., D’Agata, E., Magal, P. and Ruan, S. (2005). A model of antibiotic-resistant bacterial epidemics in hospitals. Proceedings of the National Academy of Sciences 102(37) 13343-13348.
- D’Agata, E. Horn, M. Ruan, S., Webb, G. and Wares, J. (2012). Efficacy of infection interventions in reducing the spread of multi-drug resistant organisms in hospital settings. PLoS One 77 294-8.
- Boyce, J. (2013). Update on hand hygiene. Amer J Infection Control 41 594-6.
- Boyce, J. (accessed 2020, Jul 7). How is it spread? CDC health care-associated infections, diseases and organisms. Available at: https://www.cdc.gov/hai/organisms/vre/vre.html#Spread
- WHO website (2017). Available at http://www.who.int.en/
- Harrison, S. (199). Principles of virus structure. In: Fields, B.N., Krupe, D.M., et al., eds, Virology, 2nd edn. Raven Press, Ltd., New York.
- Falanga, A. Cantisani, M., Pedone, C. and Galdiero, S. (2009). Membrane fusion and fission: Enveloped viruses. Protein & Peptide Letters 16 751-759.
- Melikyan, G. (2014). HIV entry: A game of hide-and-fuse? Curr Opin Virol 4 1-7.
- Smrt, S. and Lorieau, J. (2017). Membrane fusion and infection of the influenza hemagglutinin. Adv Exp Med Biol 966 37-54.
- Hussain, S. and Gallagher, T. (2010). SARS-coronavirus protein 6 conformations required to impede protein import into the nucleus. Virus Research 153 299-304.
- Millet, J. and Whittaker, G. (2017). Physiological and molecular triggers for SARS-CoV membrane fusion and entry into host cells. Virology 517, 2018, 3-8.
- Pawlowski, P. (2020). Cationic amino acids may promote coronavirus SARS-CoV-2 fusion with the host cell. Preprint available at: https://www.researchgate.net/publication/340630406_Cationic_amino_acids_may_promote_coronavirus_SARS-CoV-2_fusion_with_the_host_cell
- Zsembery, A., Kadar, K., Jaikumpun, P., Deli, M., Jakab, F. and Dobay, O. (2020). Bicarbonate: An ancient concept to defeat pathogens in light of recent findings beneficial for COVID-19 patients. Preprint available at: https://ssrn.com/abstract=3589403
- Spriggs, C., Harwood, M. and Tsai, B. (2019). How non-enveloped viruses hijack host machinery to cause infection. Adv Virus Res 104 97-122.
- Hickman, H. and Suthar, M. (2018). Editorial overview: Viral immunology: Generating immunity to diverse pathogens. Current Opinion in Virology 28 viii-x.
- Tyrrell, D. (1988). Hot news on the common cold, Ann Rev Microbiol 42 37-47.
- Hilding, D. (1994). Literature review: The common cold. ENT Journal 73 639-647.
- Higgins, P. and Al-Nakib, W. (1986). Interferon-beta ser as prophylaxis against experimental rhinovirus infection on volunteers. J Interferon Res 6(2) 153-9.
- Warshauer, D. (1989). End the spread. Rhinovirus infections in an isolated Antarctica station. Am J Epid 129 319-40.
- D’Allesio, D. and Mechievitz, C. (1985). Short duration exposure and the transmission of rhinovirus colds. J Infect Disease 152 403-407.
- Kampf, G., Todt, D., Plaender, S. and Steinmann, E. (2020). Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hospital Infection 104 246-251.
- Ansai, S. and Springthorpe, V. (1991). Potential role of hands in the spread of respiratory viral infections; Studies with human parainfluenza virus3 and rhinovirus14. J Clin Microbiology 29 2115-2119.
- WHO (2020, Mar 29). Modes of transmission of virus causing COVID-19. Scientific brief. Available at: https://www.who.int/news-room/commentaries/detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations
- WHO (2020, Mar 9). Rational Use of personal protective equipment for coronavirus disease (COVID-19) interim guidance. Available at: https://apps.who.int/iris/bitstream/handle/10665/331498/WHO-2019-nCoV-IPCPPE_use-2020.2-eng.pdf
- ECDC (2020 Feb). Technical report, Guidelines for the use of non-pharmaceutical measures to delay and mitigate the impact of 2019-nCoV. Available at: https://www.ecdc.europa.eu/sites/default/files/documents/novel-coronavirus-guidelines-non-pharmaceutical-measures_0.pdf
- Kutter, J., Spronken, M., Fraaij, P., Fouchier, R. and Herfst, S. (2018). Transmission Routes of respiratory viruses among humans. Current Opinion in Virology 28 142-151.
- Boyce, J. (2011). Update on hand hygiene. Amer J Infection Control 41 594-596.
- Pittet, D., Hugonnet, S. and Harbarth, S. (2000). Effectiveness of a hospital-wide program to improve compliance with hand hygiene. Lancet 356 1307-1312.
- Kampf, G., Todt, D., Plaender, S. and Steinmann, E. (2020). Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hospital Infection 104, 246-251.
- Liss, G. and Sussman, G. (1999). Latex sensitization: Occupational versus general population prevalence rates. Am J Ind Med 35 196–200.2.
- Goldi, E., Perfetti, L., Biale, C., Calcagno, G., Bianchi, P. and Moscata, G. (1998). Latex allergy in clinical practice. Allergy 53 1105-6.
- McMurry, L.M., Oethinger, M. and Levy, S.B. (1998). Triclosan targets lipid synthesis [letter]. Nature 394 531.
- Russell, A.D., Hammond, S.A. and Morgan, J.R. (1986). Bacterial resistance to antiseptics and disinfectants. J Hosp Infect 7 213-25.
- Cookson, B.D., Harrelly, H., Stapleton, P., Garvey, R.P.J. and Price, M.R. (1991). Transferable resistance to triclosan in MRSA [letter]. Lancet 337 1548-9.
- Sasatsu, M., Shimizu, K., Noguchi, N. and Kong, M. (1993). Triclosan-resistant Staphylococcus aureus [letter]. Lancet 341 756.
- Vishniavsky, N. and Archer, G. (1984). The epidemiology of antibiotic-resistant coagulase-negative staphylococci in a cardiac surgery unit [abstract 465]. In: Program and Abstracts of the 34th Interscience Conference on Antimicrobial Agents and Chemotherapy (Orlando, FL). Washington DC, American Society for Microbiology p 175.
- BMJ (1999). Hand washing [editorial]. BMJ 318 686; available at: https://www.bmj.com/content/318/7185/686/rapid-responses
- Pittet, D. (2001). Improving adherence to hand hygiene practice: A multidisciplinary approach. Emerging InfectiousDiseases 7 234-240.
- Larson, E. (1999). Skin hygiene and infection prevention: More of the same or different approaches? Clin Infect Dis 29 1287-94.
- Doebbeling, B., Pfaller, M., Houston, A. and Wenzel, R. (1988). Removal of nosocomial pathogens from the contaminated glove. Ann Intern Med 109 394-8.
- Joga, M. and Palombo, E. (2012). Removal of contaminating bacteria from computers by disinfection and hand santization. Amer J Infect Control 40 189-90.
- Chin, A., Chu, J. … Poon, L., et al. (2020 May). Stability of SARS-CoV-2 in different environmental conditions. Lancet 1 e10; available at https://www.medrxiv.org/content/10.1101/2020.03.15.20036673v2
- Lee, A. (1990, Sept 11). Skin moisturizing/conditioning antimicrobial alcoholic gels. U.S. Patent 4,956,170. Assigned to SC Johnson & Sons, Inc.
- Lubrizol (2009, Sep 3). Formulating hydroalcoholic gels with Carbopol polymers. Technical Data Sheet (TDS) 255.
- Lubrizol (2011, May 31). Pharmaceutical BM. Cline; Bulletin 21, Formulating Topical Products.
- Yusuf, A. and Barnhardt, R. (2012, Dec 11). Skin sanitizing antimicrobial compositions. U.S. Patent 8,329,758. Assigned to GOJO Industries, Inc.
- Ciavarella, N. (2019, Oct 15). High quality non-aerosol hand sanitizing foam. U.S. Patent 10,441,115
- Lilly, H.A. and Lowbury, E.J.L. (1978). Transient skin flora. J Clin Pathol 31 919-22
- Lilly, H.A., Lowbury, E.J.L. and Wilkins, M.D. Limits to progressive reduction of resident skin bacteria by disinfection. J Clin Pathol 32 382-5.
- Lochhead, R.Y., Hemker, W.J., Castaneda, J.Y. and Garlen, D. (1986). Novel cosmetic emulsions. Cosm & Toil 101(11) 125. Also Lochhead, R.Y., Castaneda, J.Y. and Hemker, W.J. (1988, May 25). Stable and quick-breaking topical skin compositions from oil-in-water emulsions containing acrylic polymers. European Patent 268164 A2; also U.S. Patent 5,004,598 (1991, APr 2). Assigned to BF Goodrich.
- Lochhead, R.Y., Broadhead, H., Josephitis, C., Perritt, C. and Johnson, C. (2018 Oct). In the thick of it: A primer on polyelectrolyte crosspolymer rheology modifiers. Cosm & Toil. Available at: https://www.cosmeticsandtoiletries.com/formulating/function/aids/In-the-Thick-of-It-A-Primer-on-Polyelectrolyte-Crosspolymer-Rheology-Modifiers-494627411.html
- Lochhead, R.Y. (2017). The use of polymers in cosmetic products. In: Sakamoto, K., Lochhead, R.Y., Maibach, H. and Yamashita, Y., eds. Cosmetic Science and Technology, Scientific Principles and Applications. Elsevier, Amsterdam ch 13 p 171.
- Lochhead, R.Y., Dodwell, R. and Hemker, W. (1993). Pemulen polymeric emulsifiers: What they are, how they work. Cosm and Toil Mfr Worldwide 77.
- Lochhead, R.Y. and Rulison, C.J. (1993). Investigation of the mechanism and associative thickening by hydrophobically-modified hydroxyethylcellulose and hydrophobically-modified poly(acrylic acid). Polymer Materials Science and Engineering 69.
- Lochhead, R.Y., Davidson, J.A. and Thomas, G.M. (1989). Poly(acrylic acid) thickeners: the importance of gel microrheology and evaluation of hydrophobically modified derivatives as emulsifiers. In: Glass, J.E., ed., Polymers in Aqueous Media: Performance Through Association. Advances in Chemistry Series #223 p 113. American Chemical Society, Washington, DC.
- Boyce, J.M. and Pittet, D. (2002, Oct 25). Guideline for hand hygiene in health care settings. CDC MMWR Report 51 No. BR-16.
- FDA (2019, Apr 12). 21 CFR Part 210, safety and effectiveness of consumer antiseptic rubs: Topical antimicrobial products for over-the-counter consumer use. Federal Register 84 No. 71 p 14847-14864.
- Boyce, J.M. and Pittet, D. (2002, Oct 25). Guideline for hand hygiene in health care settings. CDC MMWR Report 51 No. RR-16.
- WHO (2009). WHO guidelines for hand hygiene in health care. Available at: https://apps.who.int/iris/bitstream/handle/10665/44102/9789241597906_eng.pdf;jsessionid=AFDFBCaF5D0C4668C97D4D5107F8A6DE?sequence=1
- Edmonds, S.L., Macinga, D., Mays -Suko, P., Dulley, C., Rutter, J., Jarvis, W. and Arbogast, J.W. (2012). Comparative efficacy of commercially available alcohol-based hand rubs and World Health Organization-recommended hand rubs: Formulation matters. American J Infect Control 40 521-5.
- Mbithi, J., Springthorpe, V. and Boulet, J. (1992). Survival of hepatitis A virus on human hands and its transfer on contact with animate and inanimate surfaces. J Clin Microbiol 30 757-763.
- Schurmann, W. and Eggers, H. (1985). An experimental study on the epidemiology of enteroviruses: Water and soap washing of poliovirus type 1-contaminated hands, its effectiveness and kinetics. Med Microbiol Immunol 174, 221-236.
- Sattar, S., Jacobsen, H. and Rahmann, H. (1994). Interruption of rotavirus spread through chemical disinfection. Infect Control Hosp Epidemiol 15, 751-760.
- Ward, R., Bernstein, D. and Knowlton, D. (1991). Prevention of surface-to-human transmission of rotaviruses by treatment with disinfectant spray. J Clin Microbiol 29, 991-1996.
- Gwaltney, J. and Hendley, J. (1982). Transmission of experimental rhinovirus infection by contaminated surfaces. Am J Epidemiol 116, 828-833.
- Cavanaugh, G. and Wambier, C. (2020 Jun). Rational hand hygiene during the coronavirus 2019 pandemic. J Amer Acad Dermatol e211. Available at: https://doi.org/10.1016/j.jaad.2020.03.090
- Boyce, J.M. and Pittet, D. (2002, Oct 25). Guideline for hand hygiene in health care settings. CDC MMWR Report 51, No. RR-16.
- Steinmann, J., Becker, B., Biscoff, B., Paulmann, D., Friesland, M., Pietschmann, T., Steinmann, J. and Steinmann, E. (2010). Virucidal activity of two alcohol-based formulations proposed as hand rubs by the World Health Organization. American J Infection Control 38 66-68.
- Lotfinejad, N., Peters, A. and Pittet, D. (2020, May 26). Hand hygiene and the novel coronaviruspandemic: The role of healthcare workers. Journal of Hospital Infection. Available at https://doi.org/10.1016/j.jhin.2020.03.017.
- WebMD. (accessed 2020, Jul 8). What are adenovirus infections? Available at: https://www.webmd.com/children/adenovirus-infections#1-2
- Kramer, A. and Dohner, L. (2000). Hand disinfectant. U.S. Patent 6,080,417.
- Kramer, A., Galabov, A., Sattar, S., Dohner, Pivert, A., Payan, C., Wolff, M., Yilmaz, A. and Steinmann, J. (2006). Virucidal activity of a new hand disinfectant with reduced ethanol content. J Hospital Infect 62, 98-106.
- Strachan, D. (1989). Hay fever, hygiene and household size. Br Med J 299, 1259-60.
- Strachan, D. (2000). Family size, infection and atopy: The first decade of the hygiene hypothesis. Thorax 55 (suppl 1) S2-S10.
- Von Mutius, E. (2007). Allergies, infections and the hygiene hypothesis—the epidemiological evidence. Immunobiology 212, 432-439. Available at: https://pubmed.ncbi.nlm.nih.gov/17544828/
- Kramer, A., Bekeschus, S., Broker, B., Schleibinger, H., Razavi, B. and Assadian, G. (2013). Maintaining health by balancing microbial exposure and prevention of infection: The hygiene hypothesis versus the hypothesis of early immune challenge. J Hospital Infect 83 529-534.
a Quick-Break Pemulen polymers, originally registered to BF Goodrich, now Lubrizol