It is well-known that excessive exposure to UV radiation contributes to the development of skin cancer and photoaging. Topical sunscreen products are widely used to protect against UV radiation, and as of the year 2009, more products with SPF levels higher than 50 have appeared on the market.1
Low-SPF products may not provide enough protection since studies have shown consumers use less than half of the quantity required; i.e., 2 mg/cm2, the amount used to test and measure SPF.2 In fact, the low quantity of sunscreen used by consumers provides approximately only one-third of the labeled SPF.3 Hence, there is a need for higher-SPF products to provide sufficient UV protection.
Higher-SPF products come at a cost, however—they require higher levels of UV filters in formulations, which creates challenges in consumer compliance and reapplication. This is because the quantity applied and reapplied is largely dependent on the aesthetics of the sunscreen formula. And, unfortunately, the increase in UV filters in formulas can cause sunscreens to feel less aesthetically pleasing, oftentimes leaving a greasy or waxy feeling on the skin.4
This unpleasing sensorial feel can be more pronounced when organic UV filters are added at high concentrations.1 In addition, high concentrations of mineral UV filters can cause undesirable whitening on the skin. Thus, ideally, the sunscreen formula should have the highest possible protection with the lowest amount of UV filters.
High concentrations of UV filters also raise concerns over skin tolerability and safety. Some UV filters can penetrate the skin and may accumulate at high concentrations, causing allergic reactions or contact dermatitis.5 As such, using a number of UV filters at a low percentage can be a better strategy than combinations of UV filters at a high percentage, in terms of safety.4 Growing concern for the health of the world’s coral reefs is also driving a trend for using less UV filters.6 Obviously, there must be a good balance between the amount of UV filters used, texture and sensorial properties, and safety profile in order to provide sufficient UV protection.
Aside from UV rays, other ranges of radiation that reach the earth’s surface include visible light (400-700 nm) and infrared light (700 nm-1 mm). In particular, the blue/purple region (400-500 nm) has been gaining attention, as the blue light emitted from LED screens is considered a pollutant and can adversely affect skin.
Studies have shown that blue light induces the down-regulation of aquaporins 1, 3 and 9 in human keratinocytes,7 and that free radicals are generated in human skin following irradiation with blue-violet light in vivo.8 In addition, blue light can cause increased pigmentation in those with darker skin types.9 Consumers are now demanding protection not only against UV, but also across the whole spectrum of light, particularly blue wavelengths.
Therefore, there is a need for ingredients that boost SPF without negatively affecting formula aesthetics or safety, while also providing blue light protection. To address this need, different spherical particles were studied as they can potentially act as a barrier to shield the skin by means of reflecting and scattering. A grade of fine, round particles of calcium sodium borosilicatea was initially screened among other types of spherical particles and found to enhance SPF. As such, this specific grade of calcium sodium borosilicate was tested in vitro, in vivo and ex vivo, as described next, to determine its potential to protect against UV and blue light.
The spherical nature of the calcium sodium borosilicate particles provides a ball-bearing effect on skin that promotes smooth spreading.
In vitro UV/Visible Light Absorption
A syringe was used to deposit 1.3 mg/cm2 of a test formula with or without the calcium sodium borosilicate (see Formula 1) onto a substrateb. The product was spread over the whole substrate surface to achieve a uniform film and allowed to equilibrate for 15 min in the dark at a controlled temperature to ensure self-leveling of the formula. The surface was then measured using a UV-visible/NIR spectrophotometerc to: measure the flow of UV energy through the product (l) and compare it with the initial flow (I0) according to the principle of any spectrophotometric method, illustrated by Equation 1:
T (λ) = I / I0 Eq. 1
with λ as the wavelength. The absorbance at wavelength λ is related to transmittance, as determined by Equation 2:
A(λ) = -log(T(λ)) Eq. 2
In vivo SPF
The static SPF on human skin was determined based on the U.S. Food and Drug Administration’s (FDA’s) Static Method according to the FDA Monograph (2011). The light sourced is detailed in this reference. It should be noted that two-to-five subjects were tested rather than the minimum of 10 cited in the monograph. Formula 2 and Formula 3 were used for in vivo tests.
Blue Light Effects in RHE
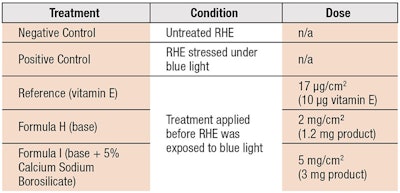
Reconstructed Human Epidermis (RHE) was produced in triplicate from keratinocytes obtained from a 21-year-old female donor; this substrate was used for morphological assessments after blue light exposure. Test Formula 4 was applied per the protocol outlined in Table 1, which details the treatment, condition and dose. Note the test formulas do not contain any mineral or organic UV filters.
After topical application with vitamin E or the variations of Formula 4, RHE was introduced in a chamber and exposed to 40 J/cm2 (427 nm) of blue light. Morphological evaluations were conducted via hemalaun eosin coloration. Biochemical evaluations were conducted using malondialdehyde (MDA) analysis with the protective index determined as follows: [Positive control – stressed + product (or reference)] / [Positive control – negative control] × 100. MDA is one of the most common products of lipid peroxidation and can therefore be used as a marker of oxidative stress that is due to blue light exposure.
Results: UV and Blue Light Absorption
The mineral sunscreen formulation with 5% calcium sodium borosilicate enhanced the absorption of UVB by 82%, UVA by 130% and blue light by 110%, compared with the same sunscreen formulation without calcium sodium borosilicate (see Figure 1).
Results: SPF Properties
Continue reading in the April 2019 Digital Edition...
References
- Wang, S. Q., Tanner, P. R., Lim, H. W., and Nash, J. F. (2013). The evolution of sunscreen products in the United States–A 12-year cross sectional study. Photochem Photobiol Sci, 12, 197.
- Autier, P., Boniol, M., Severi, G., and Doré, J. F. (2001). Quantity of sunscreen used by European students. Br J Dermatol, 144(2), 288-91.
- Faurschou, A., and Wulf, H. C. (2007). The relation between sun protection factor and amount of sunscreen applied in vivo, Br J Dermatol, 156(4), 716-9.
- Lionetti, N., and Rigano, L. (2017). The new sunscreens among formulation strategy, stability issues, changing norms, safety and efficacy evaluations. Cosmetics, 4(2), 15.
- Tampucci, S., Burgalassi, S., Chetoni, P., and Monti, D. (2018). Cutaneous permeation and penetration of sunscreens: Formulation strategies and in vitro methods. Cosmetics, 5(1), 1.
- Lim, H. W, Arellano-Mendoza, M. I, and Stengel, F. (2017). Current challenges in photoprotection. J Am Acad Dermatol, 76(3S1), S91-S99.
- Avola, R., Graziano, A. C. E., Pannuzzo, G., and Cardile, V. (2018). Blue light induces down-regulation of aquaporin 1, 3 and 9 in human keratinocytes. Cells, 3 7(11).
- Vandersee, S., Beyer, M., Lademann, J., and Darvin, M. E. (2015). Blue-violet light irradiation dose dependently decreases carotenoids in human skin, which indicates the generation of free radicals. Oxid Med Cell Longev, 579675.
- Regazzetti, C., Sormani, … Passeron, T., et al. (2018). Melanocytes sense blue light and regulate pigmentation through opsin-3. J Invest Dermatol, 138(1), 171-178.
- Becker, L. C., Bergfeld, W. F., … Anderson, F. A., et al. (2013). Safety assessment of borosilicate glasses as used in cosmetics. Int J Toxicol, 32, (supplement 3) 65S-72S.