Editor’s note: This article is the first in a two-part series describing the anti-aging effects of a natural dipeptide. The first installment presents in vitro studies; the second considers clinical efficacy.
Peter T. Pugliese, MD, refers to elastin as “the youth protein” and boldly states that “there is probably no single protein that has as much to do with a person’s appearance and well-being as the protein elastin.”1 The skin’s elasticity and firmness are naturally maintained by a complex system of cross-linked proteins that strengthen the skin’s resistance to stretching. The most important protein structure for maintaining this system is the elastic fiber, which is made up mostly of elastin and associated proteins.
Natural or induced facial aging is characterized by ptosis of the skin, which progresses under the effects of 20,000 daily muscle movements, shocks associated with walking and jumping, gravity, fat overload and reduced activity of the supporting and maintenance proteins due to UV and reactive oxygen species (ROS)-induced damage. Such effects cause the network to break down, loosen and ultimately sag along the jaw, eyebrows and lips, imparting the expression of sadness and coarsening the features of the face.2
Elastic Fibers
As noted, elastin is an essential component of the extracellular matrix (ECM) or connective tissue of the skin and is responsible for its elastic and viscoelastic mechanical properties. Elastin makes up 1–3% of the dry weight of the dermis1, 2 but is highly resistant to chemical and physical stresses thanks to its high content of hydrophobic amino acids, e.g., Akam Cakm Ile, Pro, Gly, etc., and cross-linking between certain residues of the elastin molecules. This resiliency is exemplified by the billions of extension and recoil cycles elastin undergoes in the aorta without incurring damage.3
When the skin is stretched, elastin uncoils, exposing its hydrophobic residues to the rather hydrophilic environmental medium, resulting in an unstable structure. The return of the skin to a balanced, stable state is automatic thanks to the elastic fibers, which like springs return the skin perfectly to its former position.4 In contrast to collagen, the extension of elastin is reversible.1
Production of Elastin
While elastin is the major component of elastic fibers, ~90%, it is a little-known fact that the protein is not effective if produced alone; elastin is only functional when present in the form of elastic fibers.5 The biological production of the elastic fiber is a complex process involving several players in parallel, resulting in the formation of an effective mature fiber. A review by Wagenseil and Mecham5 explains the present knowledge of elastic fiber assembly in detail.
Briefly, the process begins with the stimulation or activation of the ELN gene, concomitantly with the Microfibril Associated Glyco Proteins (MAGP) genes MAGP1, MAGP2 and fibrillin-1 and 2 (FBN1, FBN2). Tropoelastin, a 60–70 kDa protein, is secreted by the ribosome in fibroblasts, organized into small aggregates, and cross-linked by a lysyl-oxydase enzyme (LOX). Interaction with fibulin proteins of the five species Fib1 to Fib5, with the latter seemingly playing the crucial role in assembly, leads to bigger aggregates. These aggregates transport across the cell membrane and align on the microfibrils, where further processing by LOX leads to coalescence and formation of the complete elastic fiber, attached to the cell via fibulin-5 and integrins.
Transglutaminase is also an important enzyme in the elastic fiber formation process. Transglutaminase forms the cross-links or desmosines, and enables production of the strong links between elastin and fibrillin-1,6 thus stabilizing the primary scaffolding.
Scanning electron microscopy has shown that the elastic matrix of skin cells undergoes changes with age;7 disorganization and fragmentation of the fibers is observed. The same picture is observed in fibulin-5-deficient mice, whose general appearance suggests either elastolysis or cutis laxa, a disease in which, after stretching, the skin resumes its initial shape with great difficulty.
Finally, decorin, a leucine-rich proteoglycan that regulates the assembly of certain tissues, is associated with tropoelastin and microfibrils of importance at the ECM level. It acts as a companion to the collagen fibers that cohabit with elastic fibers to yield resistant skin with appropriate elastic and mechanical properties (see Figure 1).
Cosmetically Addressing Damage
Compared to research dedicated to collagen stimulation, maintenance and repair in skin fibroblasts during the past 20 years, efforts to investigate elastin and elastic fiber maintenance in skin have been small, in part due to methodological difficulties.8 A few plant extracts have been described in scientific and commercial literature as elastase enzyme inhibitors, whether of human leukocyte elastase (HLE) or various MMPs.9–11 Retinoic acid (RA) has been reported to stimulate elastin synthesis12 but it is a drug and is not permitted in cosmetic products. Also, Mondon et al. reported13 that certain oligosaccharides stimulate tropoelastin synthesis in human fibroblasts.
In relation, historic research14 has indicated that elastin present in skin having undergone actinic elastosis shows changes in its amino acid composition; specifically, in its proportions of tyrosine (Tyr) and arginine (Arg). This, coupled with the fact that fragments of fibrillin-1 and fibulin-5 contain the sequence “Tyr-Arg,” and that triggering the coordinated synthesis of various proteins and enzymes is involved in the production of elastic fibers, led the authors to investigate the potential bioactivity of the natural dipeptide Kyotorphin (Tyr-Arg),15 albeit modified to N-Acetyl-Tyr-Arg-O-Hexadecyl Ester (NATAH) (see Figure 2) for reasons of topical application and improved bioavailability. These in vitro studies are described here; extensive clinical trials were subsequently carried out as well, which will be described in the second part of this article.
Materials and Methods
Elastin and tropoelastin synthesis: Normal human dermal fibroblasts (NHDF) in DMEM were cultured to confluence. The cells were then exposed or not (negative control) to the NATAH lipopeptide at various concentrations in the micromolar (μM) range. TGF-β1 was used as the positive control. After three days, the culture supernatants were assayed to determine the quantity of elastin and tropoelastin using an enzyme-linked immunosorbent assay (ELISA) sandwich method; note that the antibody used recognizes both forms of elastin. After 18 days, immunolabeling and quantitative image analysis were conducted to evaluate the quantity of fully formed elastin fibers deposited in the ECM.
Fibrillin-1 synthesis: Cells incubated with NATAH for 18 days as described previously were labeled to detect fibrillin- 1 by immunofluorescence. The standardized photographs enabled imaging and quantification of fibrillin-1 synthesis by image analysis (n = 4) with softwarea.
Fibulin-5 synthesis: After 15 days of incubation with the peptide, the NHFD cultured as described above were lysed and analyzed using the Western blot method. Fibulin-5 synthesis was detected using a specific antibody. The resulting bands were quantified using appropriate image analysis software.
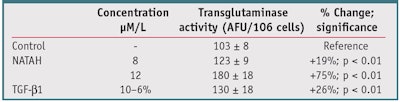
Transglutaminase activity: After three days of incubation as previously described, cell layers were exposed to dansyl cadaverine (DC) for 18 hr—the time necessary for metabolism and binding of DC by the enzyme. The fluorescence obtained is proportional to transglutaminase activity (n = 6) and was evaluated using a fluorescence reader.
Decorin synthesis: Decorin assay was conducted on cell extracts after three days of incubation as described above by an ELISA method.
3D skin model experiments: The synthesis of elastin/tropoelastin also was studied in a reconstructed skin modelb. Experimentally induced aging was accomplished by applying 0.05% betamethasone twice, initially (D0) and at the beginning of the first day (D1), to the surface of the substrate. In the evening of D1, then daily for four days, NATAH was applied at a concentration of 6 ppm in a gel to the surface of the chemically aged and non-aged skin models. The skin specimens were frozen on Day 4 for immunohistochemical labeling. With this detection method, elastin and tropoelastin are colored brown. Two skin specimens were used per condition and six sections per specimen were analyzed.
The same 3D skin model was used without the experimental aging procedure to measure LOXL-1 synthesis by immunofluorescent labeling and quantification on the skin sections—six per specimen.
Results
Elastin synthesis: Cell culture experiments, both monolayer supernatant and ECM buildup, as well as the 3D skin model studies demonstrated that incubation of the cells with the peptide and/or application of NATAH to the skin led to a highly significant increase in tropoelastin and/or elastin synthesis. The monolayer cell culture supernatant (see Table 1) showed concentration doseresponse effects; the other experiments were conducted with 12 μM/L peptide.
Elastin synthesis after application of the peptide was evaluated subsequent to immunohistochemical labeling of elastin in the dermis using image analysis. Each image (n = 6) showed a normal distribution, from 0 to 256 levels, of all the brown pixels of the selection. The distributions were subsequently averaged and two average distributions were thus obtained; one for the peptide and the other for the control. The shift in the mean distributions obtained was compared over the range of 104–155. The change in distribution was found to be directly proportional to the presence of elastin in the skin models.
The images in Figure 3 reveal that the NATAH peptide stimulates the release and binding of elastin to its matrix. The increased abundance of elastin fibers (+94% relative to control), shown in violet, in the presence of the peptide did not entail any toxic effect after 18 days of exposure, as reflected by the similar number of nuclei in the two specimens, shown as blue ovals. The 3D skin modelb has the advantage of containing both dermis and epidermis. This model, more complex than NHDF in monolayer culture, confirmed the results obtained on fibroblast monolayers.
Figure 4 reveals the stimulation of elastin synthesis in both untreated and artificially aged skin. Elastin synthesis after the application of the peptide was evaluated subsequent to immunohistochemical labeling of elastin in the dermis and showed a normal distribution (n = 6), from 0 to 256 levels, of all the brown pixels of the selection. The shift in the mean distributions thus obtained was compared over the range of 104–155. The change in distribution was directly proportional to the presence of elastin in the skin models.
The results of the analysis showed that the stimulation of elastin was approximately the same in both untreated (+14.6%; p < 0.01) and artificially aged (+14.8%; p < 0.01) skin models exposed to NATAH in comparison with the control skin specimens (see Figure 5).
Associated Protein Synthesis
As stated in the introduction, the simple stimulation of elastin or tropoelastin synthesis is not necessarily sufficient for a viable effect on tissue in human skin. Converting tropoelastin molecules into functional elastic fibers requires the concerted activity of several specific proteins and enzymes. The authors therefore investigated the main components of the cascade of events described in Figure 1, and in part two of this article, in the various in vivo models used.
The incubation of NHDF with the NATAH peptide resulted in a markedly strong, dose-dependent increase in fibrillin-1 deposition in the cell culture medium (see Table 2), with values approaching those obtained with TGF-β1. Fibrillin-1 is the first and major essential component, next to tropoelastin, for the construction of microfibers. However, tropoelastin and fibrillin-1 must be linked by chemical bonds,5, 8 a process that is governed by the two enzymes—transglutaminase (TG) and lysyl-oxydase, or its equivalent “Lysyloxydase- like” 1 (LOXL-1)—leading to the formation of desmosine. NATAH was shown to significantly stimulate both TG, assayed in monolayer culture = +75% increase, and LOXL-1, assayed in the 3D model = +135% (p < 0.01) (see Table 3 and Figure 6). It must be noted that stimulation, like that obtained with TGF-β1, occurs in both compartments of the reconstructed skin specimens. This is consistent with the findings of Noblesse et al.,16 who showed the presence of LOXL in the two upper strata of the skin but have yet to explain the role of LOXL in the epidermis.
Figure 1, although schematic and somewhat simplified, indicates that two further protein mediators have strategic roles in maintaining elasticity through the anchoring of elastic fibers to cells and the ECM. These mediators are fibulin-5, which anchor the desmosine structures to the fibroblast cell wall as an attachment to integrins, and decorin, an apparent coordinating protein whose precise role requires further elucidation.17 The data presented indicates that in normal human skin fibroblasts, NATAH, at the concentration of 12 x 10-6 M/L, increases fibulin-5 synthesis by 59% (p < 0.01) and decorin synthesis by 68% (p < 0.01) (see Figure 7). These figures compare well with those obtained with TGF- β1 (55%, p < 0.01), the usual positive control.
Part I Discussion and Conclusion
Biochemical and biological processes are always more complicated than initially expected and commonly described. Nevertheless, the knowledge of the cascade of events intervening in the deposition of elastic fibers in connective tissue, including the human skin, has become increasingly detailed. The logical sequence of events—from the release of soluble linear tropoelastin, to the anchoring of elastin fibers at specific cell surface sites via the intermediate stages of microfibril assembly, desmosine formation affected by TG and LOXL1, fibulin interaction with integrins, and decorin mediation—appears now to be increasingly well-understood.
On the basis of the results obtained using several complementary in vitro models described above, the authors conclude that the NATAH peptide stimulates the entire complex and interconnected cell machinery of NHDFs involved in producing elastic fibers. It is clearly of interest to have identified a defined molecule of peptide structure that appears to act specifically and precisely on these steps, and whose sequence moreover resembles fragments of fibrillin and fibulin proteins, thus allowing the assumption of a natural connection. This is similar to the matrikine concept, i.e., that a proteolytic fragment of one protein possessing retrofeedback stimulates activity to repair and maintain other structural tissular proteins.18 The observation that NATAH possesses these activities at concentrations close to physiological levels—most peptide messengers are active in the μM, i.e., ppm, range19—further corroborates this peptide’s specificity.
As always, questions remain: What is the specific target of NATAH on or in the fibroblast cell? Is a single message to the promoter of the ELN gene sufficient to induce the cascade of other genes’ activation, or does the peptide act on several gene loci? What other as-yetundetected factors influence the quantity and quality of elastic fibers produced? Continued research will supply some of the answers in the future. In the meantime, it is nevertheless important to ask: How pertinent are these results in view of cosmetic, topical application? Do these in vitro activities translate into measurable and perceivable skin care benefits? The clinical studies, described in part two of this paper, will show that this is indeed the case.
References
- PT Pugliese, Physiology of the Skin II, Allured Business Media, Carol Stream, IL USA (2006) p 149
- AJ Bailey, Molecular mechanisms of aging in connective tissues, Mech Aging Dev 122 735–755 (2001)
- FW Keeley, CM Bellingham and KA Woodhouse, Elastin as a self-organizing biomaterial: Use of recombinantly expressed human elastin polypeptides as a model for investigations of structure and self-assembly of elastin, Philos Trans R Soc Lond B Biol Sci. 28 357(1418) 185–189 (Feb 2002)
- JE Wagenseil and RP Mecham, New insights into elastic fiber assembly, Birth Defects Res C Embryo Today 81(4) 229–40 (Dec 2007)
- MJ Rock et al, Molecular basis of elastic fiber formation. Critical interactions and a tropoelastin-fibrillin-1 cross-link, J Biol Chem 279 23748–23758 (2004)
- M Hirai et al, Fibulin-5/DANCE has an elastogenic organizer activity that is abrogated by proteolytic cleavage in vivo, J Cell Biol 176, 1061–1071 (2007)
- RB Rucker RB and MA Dubick, Elastin metabolism and chemistry: Potential poles in lung development and structure, Environmental Health Perspectives 55 179–191 (1984)
- FR 2723313, Centaurium erythraea extract compositions, D Greff and D Fontanel, assigned to Sederman and Fontanel (1994)
- R Alasbahi and M Melzig, The in vitro inhibition of human neutrophil elastase activity by some Yemeni medicinal plants, Sci Pharm 76 471–483 (2008)
- TS Thring, P Hili and DP Naughton, Anti-collagenase, anti-elastase and antioxidant activities of extracts from 21 plants, BMC Complement Altern Med 4 9 27 (Aug 2009)
- S Tajima, A Hayashi and T Suzuki, Elastin expression is up-regulated by retinoic acid but not by retinol in chick embryonic skin fibroblasts, J Dermatol Sci 15(3) 166–172 (Sep 1997)
- P Mondon, C Mas-Chamberlin and K Lintner, Glycokines: Sugar molecules with specific messenger activity improve tissular cohesion as measured with a novel, noninvasive, non-touch “elasticity” technology, proceedings of the SCC Annual Conference, New York (2008)
- JG Smith, EA Davidson and RL Hill, Composition of normal and pathological cutaneous elastin, Nature 197, 1108–1109 (1963)
- H Takagi et al, Morphine-like analgesia by a new dipeptide, L-tyrosyl-L-arginine (Kyotorphin) and its analogue, Eur J Pharmacol 55(1) 109–111 (1979)
- E Noblesse et al, Lysyl oxidase-like and lysyl oxidase are present in the dermis and epidermis of a skin equivalent and in human skin and are associated to elastic fibers, J Invest Derm 122, 621–630 (2004)
- B Reinboth, E Hanssen, EG Cleary and MA Gibson, Molecular interactions of biglycan and decorin with elastic fiber components: Biglycan forms a ternary complex with tropoelastin and microfibril-associated glycoprotein 1, J Biol Chem 277, 3950–3957 (2002)
- FX Maquart, A Simeon, S Pascoand JC Monboisse, Regulation of cell activity by the extracellular matrix: The concept of matrikines, J Soc Biol 193 (45) 423–428 (1999)
- K Lintner K and O Peschard, Biologically active peptides: From a lab bench curiosity to a functional skin care product, Int J Cosm Sci 22 207–218 (2000)
- FX Maquart et al, Stimulation of collagen synthesis in fibroblast cultures by the tripeptide-copper complex glycyl-L-histidyl-Llysine- Cu2+, FEBS Lett 238(2) 343–346 (1998)