Gels are formed when a compound present in a solution self-assembles to form a highly cross-linked fibrillar network in which the solvent molecules remain entrapped. The resulting material has properties that are intermediate between those of liquids and solids. Usually, gels are classified according to the nature of the solvent involved.1, 2 Thus, hydrogels refer to gels formed in the presence of water, while organogels are those in which organic solvents are involved.
Many different types of compounds have been developed and used either as hydrogelators or as organogelators; according to the foods industry, natural and nonnatural polymers originally were tested for the formation of gels. Nevertheless, the use of low molecular weight gelators has gained increasing importance in recent years. Since low molecular weight compounds possess a well-defined structure, their properties including self-assembling behavior can be more easily studied, which allows for the more efficient design and fine-tuning of gelating agents. As a consequence, a large number of technological applications have recently been reported for gels formed either in water or organic solvents through the use of low molecular weight molecules.3 In particular, gel formulations are used as penetration enhancers for drug and cosmetic actives release. This is due to the fact that incorporating the active component in a gel matrix can control its release rate, increasing the application time and consequently, the corresponding activity.4
In this regard, there are several aspects that must be taken into consideration. First of all, topical application in the form of gel increases the contact time, thus favoring the action of the active principles. In addition, it limits the application to the specific desired area, reducing the side effects on other areas, i.e., local irritation. Finally, the occlusion of the active principle in a complex matrix can control the release rate of the active principles, and the capacity of the gelator molecules to interact with other compounds via supramolecular interactions can be used to regulate this capacity.
Three main properties can be used to define the efficiency of a given gelator: the minimum concentration of the compound in a given solvent at which the gel is formed; the solvents for which the compounds are able to act as the gelator; and the stability of the gel in the presence of external stimuli such as pressure, temperature and pH changes. In general, good gelators can act in concentrations as low as 1–5% w/w, or even lower. Hydrogelators and organogelators differ in that they usually possess different chemical structural motifs including specific groups such as amide, urea and carbamate. In the case of organogelators, most can act as such only for a limited number of solvents with related structures, e.g., toluene, dimethyl sulfoxide, N,N-dimethylformamide and methanol, and if one compound is a good gelator for alcohols, it is usually is not a good gelator for ethers. Hydrogelators, on the other hand, incorporate additional polar groups, e.g., carboxylic acids, hydroxy and amino, in their chemical structure, which increases their compatibility with water.
The stability of the gels formed in the presence of external stimuli is critical for practical applications, since gels provide the structure into which additional functionally active compounds are embedded. Thus, gels must be stable in the presence of active compounds as well as during the preparation conditions for the corresponding formula and its manipulation and use. In this regard, thermal stability is often a limiting factor. Gels are usually formed by heating a solvent in the presence of the gelator and upon cooling, the gel structure spontaneously forms in a reversible process. Accordingly, many gels maintain their properties only at temperatures lower than 50–60°C.
For applications in food, pharmaceutical and cosmetic industries, three leading areas for the application of gels, biocompatibility is a fourth essential property to be considered. As in many other areas of science and technology, trial and error can be used to develop biocompatible gels but a more efficient strategy is to design gelators having structural features that could, in principle, favor their biocompatibility. Building organic molecules based on natural components and providing mechanisms for biodegradation is one of the most common approaches to achieving this target. For cosmetic applications, an important line of work involves using different lipids including cholesterol, and ionic and nonionic surfactants with an amphiphilic nature. This work led to the development by different companies of proniosomal gel formulations as penetration enhancers.5-10 However, it is important to take into account that some surfactants can damage epidermis membranes.11
An alternative approach, described herein, is the use of peptides and pseudopeptide compounds of relatively low molecular weight to produce biocompatible gelators. Short peptides and pseudopeptides contain, in their structure, a large number of functional groups and can combine hydrophobic and hydrophilic domains according to the nature of the component amino acids and the structure of the non-natural components, in the case of pseudopeptides. These structural features provide the elements for hierarchichally ordered supramolecular self-assembly, leading to the development of fibrillar networks required for the gelation process. In addition, the preparation of molecules based on the presence of peptidic bonds, i.e., amide functionalities, facilitates their biodegradation.
Peptidic and Pseudopeptidic Hydrogelators and Organogelators
Different gels based on short amphiphilic peptidic sequences have successfully been used in the area of regenerative medicine. For instance, the peptide CH3CO–NH–(Arg-Ala-Asp-Ala)4–CONH2 has been applied for brain repair and has achieved a partial vision recovery in hamsters.12 These peptides present an alternating sequence of cationic (Arg) and anionic (Asp) amino acids separated by a hydrophobic amino acid. The resulting self-complementary chain forms a β-sheet structure, where one peptidic chain is hydrogen bonded to two additional contiguous chains, giving place to a two-dimensional structure and forming a twisted sheet. This β-sheet arrangement is the second form of regular secondary structure in proteins, and these sheet structures are able to further self-assemble into nanofibers, providing the formation of stable gels under physiological conditions.13 The same strategy has also been used to favor homeostasis, cartilage repair,14 growth of endotheliar cells,15 angiogenesis16 and for drug delivery.17, 18 In some of these cases, no side effects were observed in the animals six months after the introduction of the gelling agents, revealing the high biocompatibility of these types of systems.
The first examples of amino acidbased organogelators were obtained through modification of molecules containing a single amino acid subunit. Thus, long chain alkylamides of N-carbobenzyloxy amino acids such as compounds a) and b) shown in Figure 1 were reported to act as organogelators for a variety of organic solvents, although in some instances, in particular with polar solvents like alcohols or N,N-dimethylformamide (DMF), relatively high concentrations (> 10 g L-1) were required for gelation at room temperature.19, 20 Similar behavior was observed with a similar structure derived from a dipeptide—i.e., long chain alkylamides of N-benzyloxycarbonyl-l-valyl-l-valine such as compound c), also shown in Figure 1.21 The related compound d) in Figure 1, in which the long aliphatic chain is bound to the amino acid moiety through acylation of the amino group and contains a free carboxyl group, showed more efficient gelating ability; the concentration required for the relatively apolar solvents studied were below 8 g L-1.22 Cyclo(dipeptides) such as compound e) shown in Figure 1, also have organogelating properties but in general, only for a limited number of solvents.23
In recent years, the authors have been involved in the synthesis and study of simple pseudopeptidic compounds based on the general structure f) in Figure 2. These compounds have a large potential for diversity and their synthesis is relatively simple and can be achieved in high yields. The properties of the resulting pseudopeptides can be modulated through the proper selection of the spacer and amino acid. Additionally, they can be elaborated in order to incorporate other groups, e.g., R' in compound g) (see Figure 2), or to obtain macrocyclic structures such as h) and j), providing a higher degree of preorganization, thus facilitating a more efficient supramolecular interaction with other molecules, which clearly favors the formation of hierachically ordered fibrillar structures implementing the trapping of different solvents in the resulting network of the gel.24, 25 A common feature of all structures in Figure 2 is the presence of a high degree of symmetry—i.e., C2 symmetry associated to the presence of two identical component amino acids, which facilitates not only their synthesis, but also the understanding of their resulting properties and, accordingly, the proper design for the expected applications such as drug delivery, detection of amino acids and preparation of electronic devices.
The first examples applying the pseudopeptidic compounds described in Figure 2 as organogelators were provided by Hanabusa and Bhattacharya as a step forward from initial structures a), b) and c) represented in Figure 1.20, 26 Such compounds are exemplarized by compound k) in Figure 3, which also corresponds to the general compound structure b) in Figure 1, with R' being the carboxybenzyl-protecting group (Cbz-protecting group), which is a carbamate commonly used as an amineprotecting group in organic synthesis and peptide chemistry. In this case, the hydrophobic fragment is provided by the aliphatic spacer. The compounds studied were good gelating agents for solvents of low polarity, although could also gelate some polar solvents at higher concentrations (> 10 g L-1). Interestingly, removing the Cbz-protecting group to yield the free amino group to provide compounds with the general structure f) in Figure 2 resulted in a complete loss of the organogelating behavior.
A second class of open chain pseudopeptidic organogelators is represented by bis(amino acid) oxalyl amides, such as m) in Figure 3. For some of the compounds studied, a single molecule of gelator was able to retain up to 2,000 molecules of solvent, typically polar.27 The role of preorganization provided by macrocyclic structures was clearly highlighted when simple compounds such as n) in Figure 3 were studied.28–30 These compounds formed stable gels with a variety of organic solvents, in particular aromatic solvents and esters, providing materials that were stable under different conditions.31 The high level or hierarchical organization was revealed by the observation that the intrinsic chirality of the molecule was transferred to the macroscopic level in such a way that the fibers formed had a chiral helical structure. The possibility of incorporating active ingredients or drugs into such gels without disturbing their morphology was determined with the help of fluorescence techniques.32, 33
Although compounds with the general structure of j) (see Figure 2) showed interesting self-assembling properties in the solid state,34 no gelating properties had been observed for them yet. The same happens for amphiphilic pseudopeptides like o) in Figure 3.35 Rather interestingly, a small structural modification, with the introduction of an additional nitrogen atom in the middle of the central aliphatic spacer, see p) in Figure 3, provides an appropriate chemical structure for the formation of stable gels for a variety of water/ethanol mixtures.36 Additionally, in this case, the gel is formed in situ by the addition of water over the ethanolic solution, without needing the usual heating and cooling cycle.
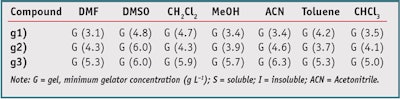
Very recently, tripodal pseudopeptidic structures such as q) in Figure 3 have also been shown to have some organogelating properties for aromatic solvents, even if relatively high concentrations were needed (> 10 g L-1).37 It must be noted that the thermal stability of those gels was significantly higher (Tg > 73°C) than that observed for most organogels. Based on the general structure g) in Figure 2, a new and interesting family of organogelating compounds is currently being studied.38 These compounds are related to structure o) in Figure 3 but incorporate a urea linkage between the hydrophobic tail and the pseudopeptidic moiety. Two main features define the properties of the resulting pseudopeptides. First, they are able to gelate a broad variety of organic solvents ranging from polar (MeOH) to relatively apolar solvents (toluene), as shown for selected examples in Table 1. In addition, all the gels formed show very high thermal stability, with gel-sol transition temperatures higher than 80°C; in some instances, it was observed that distillation of the solvent could be carried out without destroying the fibrillar structure of the gel.
As observed in Table 1, an almost universal organogelating character can be reached for some of these compounds, including with solvents such as dimethyl sulfoxide (DMSO), which has often been used in combination with different active components for skin treatments.39 It must be noted that in general, the organogelating behavior is associated with the presence of a central aliphatic spacer. This is in contrast with recent findings based on systems containing aromatic spacers but in these cases, the presence of additional functionalities on the aromatic groups seems to play a critical role for the properties of the resulting compounds.40
Pseudopeptidic Gels and Cosmetic Applications
Specific applications for pseudopeptidic gels in cosmetic formulations include where products are applied as creams or gels, in order to increase contact time and facilitate their topical application only on desired areas. Different materials, waxes for instance, have traditionally been used as a base component for cream or gel formulations, however, their use has some drawbacks. For instance, these semisolid systems often have low transparency, which can make it difficult to achieve the desired appearance in some cosmetic products. Besides, waxes and related materials must be used in relatively high amounts, in some cases 40% or even higher, to impart the semisolid property to the resulting composition. This can produce a heavy feel, difficult application and high levels of residue on the skin after application. Other alternatives have been considered, e.g., the use of emulsions or liposomes, but many of those approaches are limited due to cost constraints. Some of those limitations can be overcome by the use of organogelator and/or hydrogelator systems in place of the waxes and related materials to impart the appropriate rheological properties to the cosmetic composition.
As noted, different advantages are associated with the use of pseudopeptidic gels and in general, a good gelator must function at relatively low concentrations—certainly below 10% w/w and, advisably, below 5%. In some cases, the authors have found the amount required to be as low as 1 g L-1. This simplifies the cosmetic formulation since the gel greatly reduces the amount and variety of rheological additives required, as well as reduces residue and potential side effects. The gels obtained from low concentrations of gelating agent in particular are also very transparent and stable; the sample shown in Figure 4 was prepared by the authors from a pseudopeptidic structure.
Although few instances of amino acid-derived gelating agents for cosmetic applications are currently cited in the literature, some related structures have been used by companies in the field; for instance, in Japanese patents, the use of organogelators with a bisamide structure based on different diamines and a fatty acid, typically branched, is described.41, 42 Although the initial compounds were acknowledged for a broad range of potential applications including cosmetics, specific applications also have been described. For instance, amides of stearic acid with ethanolamine or ethylene diamine have been used for the preparation of toilet oil bars that enable the release of oil when wetted with water.43 The use of these diamides for other cosmetic and pharmaceutical applications also has been considered (see Figure 5); in particular, the preparation of new red lipsticks have been described using small amounts of compounds such as r) shown in Figure 5 (< 10%).44 More related to the pseudopeptidic structures considered here are the N-acyl amino acid amides like s) shown in Figure 5, which have been used as gelators for the preparation of antiperspirant gel sticks.45
Conclusion
In conclusion, amphiphilic peptides and pseudopeptides of relatively low molecular weight are promising candidates for the development of biocompatible hydrogels and organogels for pharmaceutical and cosmetic applications. Of particular interest are the pseudopeptidic compounds, as the incorporation of non-peptidic components allows for an easier and more effective design, and for simpler—and therefore, more economic—synthesis, a factor that can be critical for practical applications at an industrial level. By can produce gelators for essentially all kinds of solvents and for very high stabilities, which is of great importance for facilitating the manipulation of the resulting gels and to avoid the loss of gel properties upon storing, transport or use.
The biocompatibility of peptidebased gels is well-documented, having been used for tissue repair on different animals. In the case of pseudopeptides, preliminary studies on some of the described compounds also have revealed a very low level of toxicity for different microorganisms. Additionally, some compounds with the general structure h), in Figure 2, have efficiently been used as intracellular fluorescent pH probes.46, 47
It can be expected that, in the near future, peptide-based organogels and hydrogels will also be important components of the cosmetic industry, improving the aesthetic and rheological properties of the resulting compositions, facilitating the application and the release of the active principles, and reducing significantly the amount of additives required for the corresponding formulation. In addition, the high potential of these structures for being designed according to the expected application represents a key advantage for their use. It may also be envisaged that gelating compounds may be developed to selectively interact with the different active principles in the composition in such a way than a selective control of their individual release could be achieved. Finally, the development of gels with properties being regulated by external stimuli, i.e., the pH of the area of application or the presence of a specific compound, could produce intelligent cosmetic products that release the corresponding active principles only when they are required and just in the area where they are needed.
References
- RG Weiss and P Terech, Molecular Gels. Materials with Self-assembled Fibrillar Networks, Springer, Dordrecht, The Netherlands (2005)
- JL Atwood and JW Steed, Molecular Gels— Nanostructured Soft Materials in Organic Nanostructures, Wiley-VCH, Weinheim, Germany (2008) pp 111–154
- AR Hirst, B Escuder, JF Miravet and DK Smith, High-tech applications of self-assembling supramolecular nanostructured gel-phase materials: From regenerative medicine to electronic devices, Angew Chem Int Ed 47(42) 8002–8018 (2008)
- S Perrett, M Golding and WP Williams, A simple method for the preparation of liposomes for pharmaceutical applications: Characterization of the Liposomes, J Pharm Pharmacol, 43(3) 154–161 (1991)
- R Kakar, R Rao, A Goswami, S Nanda and K Saroha, Proniosomes: An emerging vesicular system in drug delivery and cosmetics, Der Pharmacia Letter 2(4) 227–239 (2010)
- S Murdan, B Bergh and G Gregoriadis, Water in sorbitan monostearate organogels (water in oil gels), J Pharm Sci, 88(6) 615–619 (1999)
- S Murdan, G Gregoriadis and AT Florence, Interaction of non-ionic surfactant based organogel with aqueous media, Int J Pharm, 180(2) 211–214 (1999)
- H Iwai, J Fukasava and T Suzuky, A liquid crystal application in skin care cosmetics, Int J Cosmet Sci, 20(2) 87–102 (1998)
- L Brinon, S Geiger, V Alard, J Doucet, JF Tranchant and G Couarraze, Percutaneous absorption of sunscreens from liquid crystalline phases, J Control Release, 60(1) 67–76 (1999)
- US Pat 4,767,625, Lamella type single phase liquid crystal composition and oil-base cosmetic compositions using the same, M Yuichiro, N Keiko and S Toshiyuki, assigned to Kao Corp (Aug 30, 1988)
- M Mezei and AKY Lee, Dermatitis effects of non-ionic surfactants IV: Phospholipid composition of normal and surfactant-treated rabbit skin, J Pharm Sci 59(6) 858–861 (2006)
- RG Ellis-Behnke et al, Nano neuro knitting: Peptide nanofiber scaffold for brain repair and axon regeneration with functional return of vision, Proc Natl Aca Sci USA 103(13) 5054–5059 (2006)
- Z Ye et al, Temperature and pH effects on biophysical and morphological properties of self-assembling peptide RADA16-I, J Pept Sci 14(2) 152–162 (2008)
- J Kisiday et al, Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair, Proc Natl Aca Sci USA 99(15) 9996–10001 (2002)
- ME Davis et al, Injectable self-assembling peptide nanofibers create intramyocardial microenvironments for endothelial cells, Circulation 111 442–450 (2005)
- K Rajangam et al, Heparin binding nanostructures to promote growth of blood vessels, Nano Lett 6(9) 2086–2090 (2006)
- S Koutsopoulos, LD Unsworth, Y Nagai and S Zhang, Controlled release of functional proteins through designer self-assembling peptide nanofiber hydrogel scaffold, Proc Natl Aca Sci USA 106(12) 4623–4628 (2009)
- X Zhao and S Zhang, Molecular designer selfassembling peptides, Chem Soc Rev 35(11) 1105–1110 (2006)
- K Hanabusa, K Hiratsuka, M Kimura and H Shirai, Easy preparation and useful character of organogel electrolytes based on low molecular weight gelator, Chem Mater 11(3) 649–655 (1999)
- S Bhattacharya and SNG Acharya, Impressive gelation in organic solvents by synthetic, low molecular mass, self-organizing urethane amides of L-Phenylalanine, Chem Mater 11(11) 3121–3132 (1999)
- K Hanabusa, J Tange, Y Taguchi, T Koyama and H Shirai, Small molecular gelling agents to harden organic liquids: Alkylamide of N-benzyloxycarbonyl-L-valyl-L-valine, J Chem Soc, Chem Commun 4 390–392 (1993)
- X Luo, B Liu and Y Liang, Self-assembled organogels formed by mono-chain-alanine derivatives, Chem Commun 17 1556–1557 (2001)
- K Hanabusa, M Matsumoto, M Kimura, A Kakehi and H Shirai, Low molecular weight gelators for organic fluids: Gelation using a family of cyclo(dipeptide)s, J Colloid Interface Sci 224(2) 231–244 (2000)
- J Becerril et al, Efficient macrocyclization of U-turn preorganized peptidomimetics: The role of intramolecular H-bond and solvophobic effects, J Am Chem Soc 125(22) 6677–6686 (2003)
- M Bru, MI Burguete, SV Luis and I Alfonso, Anion-templated syntheses of pseudopeptidic macrocycles, Angew Chem Int Ed 45(37) 6155–6159 (2006)
- K Hanabma, R Tanaka, M Suzuki, M Kimura and H Shirai, Excellent gelators for organic fluids: Simple bolaform amides derived from amino acids, Adv Mater 9(14) 1095–1097 (1997)
- J Makarevic, M Jokic, B Peric, V Tomisic, B Kojic-Prodic and M Zinic, Bis(amino acid) oxalyl amides as ambidextrous gelators of water and organic solvents: Supramolecular gels with temperature dependent assembly/ dissolution equilibrium, Chem Eur J 7(15) 3328–3341 (2001)
- J Becerril, MI Burguete, B Escuder, SV Luis, JF Miravet and M Querol, Minimalist peptidomimetic cyclophanes as strong organogelators, Chem Commun 7 738–739 (2002)
- J Becerril et al, Self-assembly of small peptidomimetic cyclophanes, Chem Eur J 10(16) 3879–3890 (2004)
- J Becerril, B Escuder, R Gavara, SV Luis and JF Miravet, Understanding the expression of molecular chirality in the self-assembly of a peptidomimetic organogelator, Eur J Org Chem 3 481–485 (2005)
- MI Burguete et al, Use of fluorescence spectroscopy to study polymeric materials with porous structure based on imprinting by selfassembled fibrillar networks, Langmuir 24(17) 9795–9803 (2008)
- F Galindo, MI Burguete, R Gavara and SV Luis, Fluorescence quenching in an organogel as a reaction medium, J Photochem Photobiol A Chem 178(1) 57–61 (2006)
- MI Burguete, MA Izquierdo, F Galindo and SV Luis, Time resolved fluorescence of naproxen in organogel medium, Chem Phys Letters 460(4) 503–506 (2008)
- I Alfonso, M Bru, MI Burguete, E García- Verdugo and SV Luis, Structural diversity in the self-assembly of pseudopeptidic macrocycles, Chem Eur J 16(4) 1246–1255 (2010)
- J Rubio, I Alfonso, MI Burguete and SV Luis, Stimulus responsive self-assembly of Gemini amphiphilic pseudopeptides, Soft Matter 7 10737–10748 (2011)
- J Rubio, I Alfonso, MI Burguete and SV Luis, Interplay between hydrophilic and hydrophobic interactions in the self-assembly of a Gemini amphiphilic pseudopeptide: From nano-spheres to hydrogels, Chem Commun 48 2210–2212 (2012)
- S Samai, J Dey and K Biradha, Amino acid based low-molecular-weight tris(bis-amido) organogelators, Soft Matter 7(5) 2121–2126 (2011)
- EU Pat 201131333, High temperature organogelating compounds, SV Luis, J Rubio, V Martí-Centelles and MI Burguete, assigned to University Jaume I (Jul 29, 2011)
- A Casiraghi, P Ardovino, P Minghetti, C Botta, A Gattini and L Montanari, Semisolid formulations containing dimethyl sulfoxide and a-tocopherol for the treatment of extravasation of antiblastic agents, Arch Dermato Res 299(4) 201–207 (2007)
- HF Chow and CH Cheng, Expanding the gelation properties of valine-based 3,5-diaminobenzoate organogelators with N-alkylurea functionalities, Beilstein J Org Chem 6 1015– 1021 (2010)
- JP 10 237034, Stabilizing compositions containing 1,2-bis(acylamino)cyclohexanes for food, cosmetics, pharmaceuticals, etc, K Hanabusa et al, assigned to Pola Chemical Industries (1998)
- JP 07 138555, Solidifying and gelling agents for organic liquid, H Goto et al, assigned to Nisshin Fine Chemical KK (1955)
- US Pat 3 857 960, Toilet oil bar, L Mackles, assigned to Bristol-Myers Co (1972)
- EP 1 174 110 A1, Pharmaceutical or cosmetic composition presented as a solid containing bis-acyl-amides, A Livorell and S Genard, assigned to L’Oréal (2002)
- WO 93/23008, Antiperspirant gel stick compositions, BD Hofrichter and JM Boudinot, assigned to Procter & Gamble (1993)
- F Galindo et al, Synthetic macrocyclic peptidomimetics as tunable pH probes for the fluorescence imaging of acidic organelles in live cells, Angew Chem Int Ed 117 (40) 6504–6508 (2005)
- MI Burguete et al, Synthesis and evaluation of pseudopeptidic fluorescence pH probes for acidic cellular organelles: In vivo monitoring of bacterial phagocytosis by multiparametric flow cytometry, Eur J Org Chem 2010(31) 5967–5979 (2010)