Natural ingredients have always been important in the cosmetics market, and the demand for the use of greener processes is becoming more important to both formulators and consumers. In the present article-and to emphasize the distinction-natural refers to the source of raw materials, an example set by the Natural Products Association, while green refers to the process used to convert starting materials to a finished ingredient.
The US Environmental Protection Agency (EPA) has adopted a series of principles to guide the development of environmentally responsible and sustainable manufacturing, called “The 12 Principles of ‘Green’ Chemistry.”1 These principles have guided the development of the current biocatalytic process.
The green nature of biocatalysis- along with its mild reaction conditions, high and sometimes unique reaction selectivities, and the potential to work solvent-free-suggest that biocatalysis is a natural fit for the green preparation of cosmetic ingredients. Since petroleum-based organic solvents are undesirable for green processes, biocatalytic processes (enzymatic or microbial transformations) can instead be used since they generally are considered green.
Green Production of Esters
Green processing is especially relevant in the manufacture of naturally derived cosmetic ingredients. Natural starting materials typically are derived from plants or microbes via fermentation with minimal processing. Cold-pressed seed oils are examples of plant-derived starting materials with broad cosmetic utility. Oils (triglycerides) can be converted to the humectant glycerol or to fatty acids—both of which are starting materials for esters.
Esters are an important class of cosmetic ingredients, encompassing actives, emollients, emulsifiers and surfactants. They are employed in antiaging ingredients such as retinyl palmitate, in emollients such as fatty alcohol/fatty acid esters, and in emulsifiers such as mono- and diglycerides. Traditionally, esters are made by condensation of an alcohol and an acid in the presence of a strong acid catalyst and elevated to temperatures to both drive the reaction and remove the water by-product.2
Acid-catalyzed, high-temperature esterification reactions are energy-intensive and the reaction conditions are harmful to many starting materials, such as unsaturated fatty acids. Under harsh conditions, these starting materials produce undesirable color, odor and by-products that impact yield. In addition, further process steps must be included to remove the acid catalyst.2
In contrast, biocatalytic processes involve mild reaction conditions that do not degrade sensitive reactants or products, resulting in a reduced number of by-products as well as improved color and odor production.2 Numerous reports of biocatalytically prepared cosmetic esters exist. A few examples include plant monoterpenes as fragrance molecules (geranyl acetate),3 sugar fatty acid ester surfactants4 and fatty acid esters of plant phenolic antioxidants such as rutin and naringin.5 However, these examples are not green since they require the use of organic solvents for both the reaction and post-reaction processing to purify the final product.
A breakthrough in the deeper “greening” of biocatalytic processes, described here, is the elimination of organic solvents. Reactions similar to those described above have been performed in the absence of organic solvents and address the same classes of cosmetic ingredients: plant monoterpenes as fragrance molecules (citronellyl acetate),6 sugar fatty acid ester surfactants7 and fatty acid esters of plant phenolic antioxidants (ferulic acid).8
Eliminating the solvent from a biocatalytic reaction can have a significant environmental impact. In the authors’ experience, when solvent and solvent-free biocatalytic processes for manufacturing fatty acid esters are compared, the solvent-free biocatalytic process saves more than 10 L organic solvent/kg of product in reaction and post-processing waste. Solvent-free systems also offer better volumetric production than solvent-borne reactions and in many cases, the products are sufficiently pure so that no post-reaction processing is required.2, 9, 10
Solvent-free biocatalytic reactions can be carried out continuously2 or as batch reactions.11 A typical stirred tank batch reaction scenario is depicted in Figure 1. The two reactants are loaded along with the biocatalyst, and the mixture is heated to the desired temperature. The reaction is then driven to high conversion by removal of the volatile by-products such as water or a lower alcohol by one of several methods including evaporation, pervaporation or by purging the reaction mixture with inert gas (shown in Figure 1). Removal of these by-products perturbs the equilibrium of the enzymatic reaction and affords high conversion without the need for large excess of either reactant. A semi-batch or continuous process, along with an evaporative method to remove the water by-product of the reaction, has been described for the economical production of higher-volume cosmetic ingredients.2, 10
Case Studies: Esters for Skin Illuminating and Emulsifying
Skin-illuminating ingredients: Inhibitors of tyrosinase-a key enzyme in melanin biosynthesis from tyrosine in mammalian skin-are useful for reducing undesirable pigmentation of the skin, resulting in a more even skin tone. The ability of a compound to address skin pigmentation can be effectively predicted in vitro by measuring the tyrosinase-inhibiting activity.12
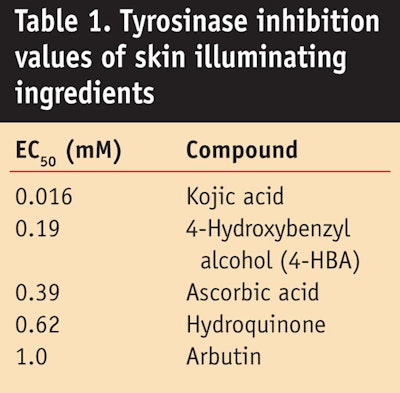
Different tyrosinase inhibitors exist on the market, including hydroquinone, kojic acid, ascorbic acid and arbutin. Kojic acid is a natural product formed by fermentation with a history of use as a cosmetic ingredient for achieving even skin tone. Structurally related to kojic acid, 4-hydroxybenzyl alcohol (4-HBA) is a generally regarded as safe (GRAS) flavoring substance (Flavor and Extract Manufacturers Association substance No. 3987) that has also shown potential as a ingredient for evening skin tone. The EC50 or concentration of these ingredients at which the tyrosinase exhibits only 50% of its normal activity is shown in Table 1. A low EC50 value indicates the material is an effective inhibitor. The activity of 4-HBA was found to be higher than ascorbic acid, hydroquinone and arbutin in the tyrosinase inhibition assay.
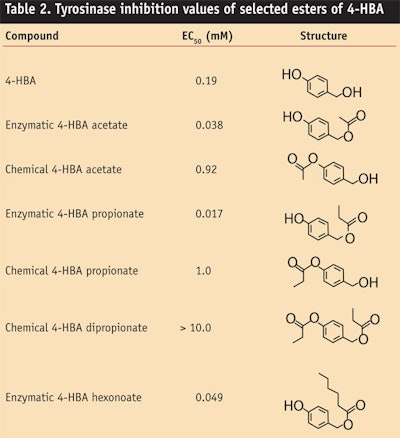
Two distinct monoesters can be prepared from 4-HBA since the parent molecule has both a phenol and a primary alcohol. Esterification using standard chemical means such as acid chlorides or anhydrides selectively occurs at the phenol because this is the most acidic hydroxyl. However, enzymatic esterification of 4-HBA is selective for the benzyl alcohol, often resulting in > 98% selectivity for the primary ester.
Using the protocol shown in Figure 1, 4-HBA can be esterified enzymatically with an acid or ester reactant under solvent-free conditions and with a high conversion to ester, which is achieved by purging the mixture with nitrogen to remove the water or alcohol by-product. After removal of the enzyme, 4-HBA esters of high purity are obtained. These enzymatically prepared esters function as highly effective tyrosinase inhibitors (seeTable 2), demonstrating enhanced tyrosinase inhibiting activity as compared to the parent diol and the chemically prepared ester (4-hydroxymethylphenyl ester) and diester.
The esters are also significantly more lipophilic than the parent molecule, as demonstrated by their increased solubility in non-polar media. Fatty acid esters of skin-illuminating ingredients improve permeability, activity, pH and heat stability of an ingredient in a formulation. These performance benefits have been demonstrated for kojic acid13, and can be inferred for structurally related 4-HBA.
Emulsifiers: Plant oil-derived esters can form the basis for emulsifier and emollient esters, and are also targets for a green biocatalytic production process. The naturally occurring mixture of fatty acids in the starting plant oil is reflected in the composition of the final product. As fatty acid chain length and degree of unsaturation alter the properties of the final product, the specific needs of the formulator can be addressed and tailored by judicious choice of plant oil. In addition, mono- and poly-unsaturated fatty acids, such as conjugated linoleic acid and gamma linolenic acid, can be processed via biocatalysis without compromising the structural integrity of the fatty acid or producing undesired color in the reaction product. 11
Emulsifiers are an important class of cosmetic ingredients used to stabilize creams and lotions. Lecithin is the only green and natural emulsifier used in cosmetic formulations today. More familiar in food applications, lecithin is a naturally derived mixture of compounds including phospholipids, which impart the emulsifying properties. Lecithin is typically separated from soybean oil by steam precipitation, qualifying the product as a green and natural ingredient. However, more versatility in the portfolio of natural and green emulsifiers for cosmetics is needed.
One class of emulsifiers commonly used in food and cosmetic products is glycerides. These fatty acid esters of glycerol currently are available as synthetically produced ingredients, but solvent-free enzymatic synthesis of fatty acid mono- and diglycerides as emulsifiers has also been reported.9-11, 14
Mixtures of glyceride esters can be prepared from glycerine and fatty acid ethyl esters derived from plant oils by biocatalytic, solvent-free transesterification. These reactions are pushed to high conversion by removal of the ethanol liberated in the transesterification using a nitrogen purge, resulting in glycerides with > 95% purity (based on gas chromatographic analysis for residual fatty acid ethyl ester).
To evaluate the glycerides, the rheological properties of a simple 1:1 mixture of golden jojoba oil and water emulsified with 5% w/w of each glyceride were investigated. Glycerides have a unique property among emulsifiers in their ability to interact with polysaccharides and polypeptides to dramatically change the rheological properties of the emulsion. This interaction is well-known in the food industry and can be leveraged in the formulation of cosmetic emulsions as well. To demonstrate the interaction of glycerides with polysaccharides, samples were also prepared where the water phase contained 1% w/w soluble potato starch (dissolved by heating). The mixtures were each homogenized and held at RT. The viscosities of the resulting emulsions were measured at 23°C using a viscometera.
In Figure 2, viscosity is plotted as a function of shear rate. The solid lines depict the broad range of emulsion viscosities—a function of the natural fatty acid composition of the oil (related to chain length and degree of unsaturation) and any natural minor components present in the oil. Glyceryl oleate resulted in the thickest emulsion, while glyceryl pomegranate had a thinner, cream-like consistency. When 1% w/w soluble potato starch was added to the water phase, the viscosity of the emulsions increased (dotted lines). An interaction between the glyceride and an additive was also apparent in the presence of other biological polymers such as agar and lactalbumin. The interaction between glycerides and polysaccharides/proteins varies with the additive and suggests that a wide and interesting variety of textures could be achieved.
Conclusion
Consumers are becoming much more educated about the efficient use of resources, particularly as expressed by natural ingredients prepared using green chemistry. In addition, many consumer product companies are developing and communicating a sustainability strategy that includes more environmentally responsible manufacturing practices. Good communication between suppliers and customers is crucial to developing greener ingredients, and suppliers will be challenged to come up with creative ways of communicating a green story to their customers.
A comprehensive picture will compare the overall ecological impact of green ingredients with more traditional synthetic counterparts. This picture will include such diverse considerations as the geographical source of raw materials, annual renewability, competition with the food supply, processes used in manufacture, solvents used in manufacture, manufacturing energy used and its source, processes used to clean and maintain the manufacturing equipment, choice of ingredient packaging, mode of shipping, and shelf-life and biodegradability of the ingredient, among others.
The biocatalytic production of cosmetic ingredients has advantages. Communicating the green story will require an in-depth analysis of the biocatalytic process and a comparison with the corresponding synthetic processes in terms of energy saved and solvent/other waste eliminated. The widespread use of biocatalysis for the production of green and natural ingredients has yet to occur, although a combination of biocatalyst technology and process engineering developments will lead to more uses for these green processes in cosmetics, and eventually, across all markets.
References
1. P Anastas and J Warner, Green Chemistry: Theory and Practice, Oxford University Press: New York (1998)
2. T Veit, Biocatalysis for the production of cosmetic ingredients, Eng Life Sci 4(6) 508–511 (2004)
3. A Trusek-Holownia, A Noworyta, An integrated process: Ester synthesis in an enzymatic membrane reactor and water sorption, J Biotechnol 130, 47–56 (2007)
4. US Pat 5,505,938, Straight chain saturated or unsaturated C8-C18 alkyl aldonolactone esters and an enzymatic process for their preparation, DJ Pocalyko, AJ Carchi, B Harichian andR Vermeer, assigned to Lever Brothers Company (Apr 9, 1996)
5. E Passicos, X Santarelli, D Coulon, Regioselective acylation of flavonoids catalyzed by immobilized Candida Antarctica lipase under reduced pressure, Biotechnol Lett 26(13) 1073–1076 (2004)
6. F Fonteyn, C Blecker, G Lognay, M Marlier and M Severin, Optimization of lipase-catalyzed synthesis of citronellyl acetate in solvent-free medium, Biotechnology Letters 16(7) 693–696 (1994)
7. M-P Bousquet, R-M Willemot, P Monsan, and E Boures, Enzymatic synthesis of unsaturated fatty acid glucoside esters for dermo-cosmetic applications, Biotechnol Bioeng 63(6) 730–736 (1999)
8. JA Laszlo, DL Compton, FJ Eller, SL Taylor,TA Isbell, Packed bed reactor synthesis of feruloyl monoacyl- and diacylglycerols: clean production of a green sunscreen, Green Chemistry 5, 382–386 (2003)
9. P Tufvesson, A Annerling, R Hatti-Kaul, D Adlercreutz, Solvent-free enzymatic synthesis of fatty alkanolamides, Biotechnol Bioeng 97(3), 447–453 (2006)
10. J Aracil, M Martinez, and R Soriano, Valorisation of glycerol. Enzymatic synthesis of fatty acid monoglycerides, Proceedings of 1st World Conference on Biomass for Energy and Industry, London, James & James, Ltd. (2001) pp 1047–1050
11. Z Guo and Y Sun, Solvent-free production of 1,3-diglyceride of CLA: Strategy consideration and protocol design, Food Chemistry 100(3) 1076–1084 (2007)
12. SJ Um, MS Park, SH Park, HS Han, KJ Kwan, HS Sin, Synthesis of new glycerrhetinic acid (GA) derivatives and their effects on tyrosinase activity, Bioorganic & Medicinal Chemistry 11(24) 5345–5352 (2003)
13. US Pat 4,369,174, Cosmetic composition containing kojic acid ester, S Nagai and T Isumi, assigned to Sansho Pharmaceutical Co, Ltd (Jan 18, 1983)
14. AM Fureby, P Adlercreutz and B Mattiasson, Glyceride synthesis in a solvent-free system Journal of the American Oil Chemists Society 73(11) 1489–1495 (1996)