Emulsions are thermodynamically unstable systems defined as microscopic dispersions of liquid droplets contained within another liquid, with a diameter ranging from 0.5 mm to 100 mm.1 Emulsions usually consist of mixtures of an aqueous phase with various oils or waxes. The liquid that is broken up into droplets is termed the internal or disperse phase, whereas liquid surrounding the droplets is known as the external or continuous phase. Both phases are stabilized by a third component, the surfactant.2
The two most familiar types of emulsions are readily distinguished as o/w and w/o.3 The majority of skin care products and a significant percentage of toiletry products are emulsions.4 The basic components of these formulations are emulsifiers, emollients and consistency enhancers. Additional value can be given to these formulations by including active ingredients with specific cosmetic or dermatological effects. Particularly advantageous cosmetic emulsion preparations are obtained when antioxidants are used as active ingredients.
Based on a perceived safety benefit, the worldwide trend toward using natural additives has spurred interest in natural antioxidants found in plants.7 Extract from grapefruit is rich in natural antioxidants8 and provides some cosmetic benefits for the skin such as reduction in skin melanin, increase in skin moisture and antiwrinkle effects.7 The best natural antioxidants present in grapefruit extract are ascorbic acid or vitamin C,9 flavonoids,10 beta carotene and lycopene.11 Thus, the current work aimed to formulate a stable w/o emulsion containing grapefruit extract by entrapping the extract in the inner aqueous phase. Tests were performed on sample formulations to measure their effects on different physiological characteristics such as melanin, erythema, moisture, sebum, pH and TEWL.
Materials
Paraffin oil was used in the oily phase of a test formulation containing 1% grapefruit extract, as well as in the control (see Formula 1). Paraffin oil is a synthetic that is often preferred because of its benefits including being nontoxic and nonirritant to skin, as well as its ability to form an elegant white emulsion. It is a mixture of refined liquid saturated aliphatic (C14–C18) and cyclic hydrocarbons obtained from petroleum.12
In addition to the paraffin, cetyl PEG/PPG-10/1 dimethiconea was chosen as the lipophilic emulsifier to form a w/o emulsion in both the test formulation and the control.13
Beeswax was incorporated in both formulas to increase the consistency of the cream and to stabilize the w/o emulsion.14
Grapefruit extract is water-soluble and thus was used as an aqueous extract in the test formula. During this study, 30 different test formulations were prepared but in the present work, data is presented comparing only one test formulation with the control.
Preparing the Emulsion
The w/o test emulsion was prepared by adding the aqueous phase incorporating the grapefruit extract to the oily phase with continuous agitation (see Formula 1).15
The oily and aqueous phases were separately heated to 75C ± 1C, and one to two drops of citric acid were added to the aqueous phase to adjust the pH before heating. After heating, the aqueous phase was added to the oily phase, drop by drop.
Stirring was continued at 2000 rpm by the mechanical mixer for about 10 min until the complete aqueous phase was added. Two to three drops of lemon oil were added during this stirring time to give the emulsion a fragrance.
After complete addition of the aqueous phase, the speed of the mixer was reduced to 1000 rpm for homogenization for a period of 5 min, then the speed was further reduced to 500 rpm for another 5 min for complete homogenization, until the emulsion cooled to room temperature. The control formula, without grapefruit extract, was prepared in the same manner.
Emulsion Analysis
The test and control emulsions were visually analyzed both organoleptically to test color, thickness, look and feel, and physically to measure creaming and phase separation.
Stability: In the cosmetics industry, product stability is one of the most important quality criteria.16 Final acceptance of an emulsion by the consumer depends on its stability and appearance,17 and one readily apparent requirement in a well-formulated emulsion is that the emulsion possesses adequate physical stability.18
In the present study, the base and test formulation each were divided into four samples separately and these samples were kept at different storage conditions—i.e., at 8˚C in a refrigerator, at 25˚C, 40˚C, and at 40˚C + 75% relative humidity (RH) in stability chambers. These samples were observed with respect to change in color, liquefaction and phase separation for a period of 28 days at definite time intervals.
Color: The freshly prepared base and test formulation appeared elegant white in color and no change in color was observed for either the control or test formula, up to an observation period of 28 days. This showed both emulsions were stable at the different storage conditions throughout the 28 days.
Liquefaction: The viscosity of an external oil phase is the key factor contributing to the formation of stable emulsions.19 According to Stoke’s law, increased viscosity of the external phase is associated with an improved shelf-life of emulsions.20 Cosmetic creams appear as stable, concentrated emulsions21 but as soon as an emulsion has been prepared, time- and temperature-dependent processes occur to affect its separation, leading to the decreased viscosity, which results in increased liquefaction.18
No liquefaction was observed with the naked eye in the control and test formulation samples stored at 8˚C and 25˚C during the 28 days but slight liquefaction occurred in the samples of the control kept at 40˚C and 40˚C + 75% RH from the 21st day of observation. No further increase in liquefaction was noted until the end of the study. On the other hand, a slight liquefaction was observed in test formulation samples stored at 40˚C and 40˚C + 75% RH at the 28th day of the observation period.
Stoke’s law states that the rate of creaming is inversely proportional to the viscosity. As creaming increased, the viscosities of the base and the formulation gradually decreased at rising temperatures, resulting in liquefaction.20
Phase separation: The instability of emulsions is explained by phase separation,16—i.e., any emulsion reverts back to the separate bulk phases. The separated phase can either cream or sediment.18 Destabilization is compounded mostly by coalescence and gives a first indication through extension of droplets.16 The two instability processes, coalescence and Ostwald ripening, result in droplet size growth with time.2 According to Stoke’s law, larger droplets cream much more rapidly than smaller particles.20 The concentration of the disperse phase and the droplet size are key parameters in determining the type and time scale of the instability process.2
In the case of the control and test formulations, no phase separation was observed in any of the samples up to 28 days, indicating their stability.
Centrifugation: Centrifugation, if used judiciously, is an extremely useful tool for evaluating and predicting the shelf-lives of emulsions.18 The cream volume or the separation of phases at a given time is taken as a measure of the physical stability of emulsion.21 However, these are examples of situations that exist in any accelerated test—namely the tendency to “overkill” emulsions because the tests used introduce a new mechanism of instability by causing unreasonably high stress.18
In the described study, 5–10 g samples of the test and control formulas were rotated at 5000 rpm for 5 min. The test was performed for the formulations kept at the different storage conditions for up to 28 days at definite time intervals. No phase separation on centrifugation was noted in any of the samples thus indicating that both the test and control emulsions were stable for 28 days.
Electrical conductivity: Conductivity differences arise when an emulsion creams, the proportion of oil increases in the separated upper portion of the emulsion, and the proportion of water increases in the lower part of the emulsion. By measuring conductivity differences in the upper and lower parts as a function of storage time, instability can be determined.21 Conductivity tests are also used to distinguish between emulsion types22—o/w emulsions will conduct because water is the continuous phase, and since oils are poor conductors, w/o emulsions conduct poorly.23
In the present study, a conductivity test was performed for all samples of the control and test formulation. No change in electrical conductivity was noted.
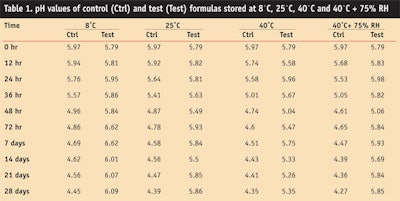
pH: pH is a significant parameter in so far as the effectiveness of the cream is concerned, and it can be used as an indicator of emulsion stability.16 For the formation of stable emulsions, the pH value of the aqueous phase is a key factor.19 The pH of skin ranges between 5 and 6; 5.5 is considered the average pH level. Therefore, formulations for application to skin should have pH close to this range.
In the present study, the pH of the freshly prepared control and test formulations was found to be 5.79 and 5.97, respectively (see Table 1).
The pH values decreased continuously from the first to the last day. On the 28th day, the pH levels of the control formulation samples were 4.45, 4.39, 4.35 and 4.27, respective to the different storage conditions. The pH of the control samples stored in different conditions showed slight variations with time, and slightly increased at the end of the study period. This could have been due to the production of metabolites; no such tendency is inherent in emulsions, but it can occur.
The pH values of the test formulation measured 6.09, 5.86, 5.35 and 5.85 on the 28th day, respective of their storage conditions—i.e., at 8˚C in a refrigerator, at 25˚C, 40˚C, and at 40˚C + 75% RH.
By using a two-way Analysis of Variance (ANOVA) technique at 5% level of significance, the change in pH of the samples of control formulation, at different levels of time and temperature, was found to be significant but there was an insignificant difference in changes of pH for samples of the test formulation.
A Least Significant Difference (LSD) test also was applied to determine the individual average effects of the pH levels of the control and test formulas at different temperatures with the passage of time. From this test it was concluded that at different storage conditions, a significant change occurred in the pH of the control formula but an insignificant change was observed in the test formulation samples.
Dermatological Tests
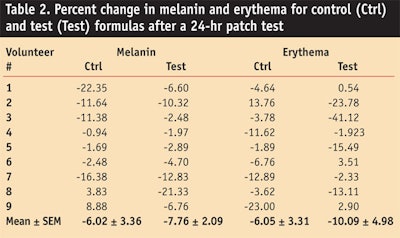
Patch test: Before application of the control and test formulations to a total of nine male and female human volunteers aged 20–25, patch tests for melanin and erythema were performed and the values measured are shown in Table 2.
After performing the patch test for 24 hr, the test and control formulations were found to produce no skin irritation; thus both creams were deemed safe for in vivo evaluation. This lack of irritation could have been attributed to the presence of a good emollient in both formulas—i.e., glycerin24—or ascorbic acid, a natural antioxidant present in the formula from the grapefruit extract,25,26 which has been shown to reduce skin erythema.27,28
Melanin
Melanocytes are present in the basal layer of the epidermis and they manufacture melanin, which is responsible for the color of skin. Melanin is formed through a series of oxidative reactions involving the amino acid tyrosine in the presence of the enzyme tyrosinase. Tyrosinase catalyzes three different reactions in the biosynthetic pathway of melanin in melanocytes.29
Antioxidants have important physiological effects on skin.30 Their main role is to scavenge free radicals such as peroxides27 that contribute to tyrosinase activation and melanin formation.31 As previously mentioned, grapefruit extract is rich in natural antioxidants32 that capture free radicals, resulting in the inhibition of melanogenesis.30
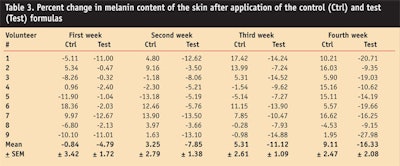
In the present study, the effect of the control and the test formulations on the production of skin melanin was examined. The amount of melanin was measuredb for four weeks at different time intervals in each test subject after application of the control and test formulation.
It was found that the control formulation increased melanin content in the skin until the end of the 28-day test period, while the test formulation incorporating grapefruit extract decreased the melanin content throughout the study (see Table 3).
An ANOVA test found the control and test formulations to produce statistically significant effects on skin melanin content in volunteers; a paired sample t-test showed a significant difference in the melanin effects of the control versus the test formulation from the second week, lasting to the fourth week of the study, confirming the two creams had different effects on melanin.
Researchers concluded that the decreased melanin content after application of the test formulation could have been attributed to the antioxidant activity of the grapefruit extract—particularly its potent antioxidant vitamin C that causes inhibition of melanogenesis.9, 30,31
Erythema
Skin irritation is caused by the direct toxicity of chemicals on cells or blood vessels in the skin and is different from contact allergy, which is caused by an immune response.28
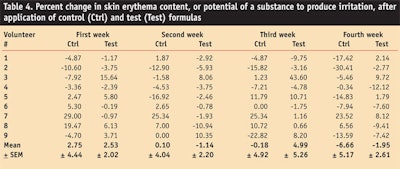
In the described in vivo study, irritation also was monitored weekly throughout the test period for both formulations. Erythema was measuredb and found to be slightly increased after application of the control formula for the first week but almost no change was observed during the second week, and a pronounced decrease was observed at the third and fourth week.
In comparison, after application of the test formulation, erythema was slightly increased after the first week; however, decreased at the second and fourth week.
An ANOVA test confirmed statistically that the control and test formulation produced insignificant effects on skin erythema. A paired sample t-test showed a significant variation in irritation with respect to the control and test formulation at the first and second week (see Table 4).
It was thus concluded that both the test and control formulations decreased erythema at the end of the study period and the overall effect of the test formulation on erythema was insignificant; thus it could be used safely without irritating skin. In addition, the active ingredient in grapefruit extract is a good source of vitamin C,33 which is used as an anti-inflammatory,28 and topical vitamin C is claimed to inhibit UV-radiation-induced damage to porcine skin since it functions as a biological co-factor and an antioxidant.29
Moisture Content
Moisturizing treatments involve factors such as repairing the skin barrier; retaining/increasing water content; reducing TEWL; restoring the lipid barrier’s ability to attract, hold and redistribute water; and maintaining skin integrity and appearance.34
A 100-g sample of grapefruit juice contains 36–40 mg of vitamin C,11 which is known to increase the collagen fibers in the dermis.35–39 With an increase in collagen, conditions for hydration are improved.40 In addition, vitamin C improves the barrier function of the stratum corneum (SC), in turn improving moisture in the skin.41
In both formulations tested, glycerin was incorporated in the internal aqueous phase to enhance the skin moisture level. Glycerin is a humectant with excellent sensorial properties that moisturizes the full thickness of the SC while creating an apparent “reservoir” of moisture in the skin. This makes the skin more resistant to dry conditions.24
In the present study, a slight increase in moisture was foundc from the first to third week after application of the control, and a slight decrease was observed at the fourth week; however, after the application of the test formulation, the skin moisture content was more pronounced from the first to the fourth week.
ANOVA testing found that the control showed an insignificant change with respect to the basic values, at 0 hr before application of creams, whereas the test formulation showed a significant variation throughout the study period of 28 days.
By applying an LSD test, the change in moisture content was found to be statistically significant after application of the test formulation. A paired sample t-test confirmed a significant difference in the moisture values was produced at the fourth week when comparing the control to the test formulation (see Table 5).
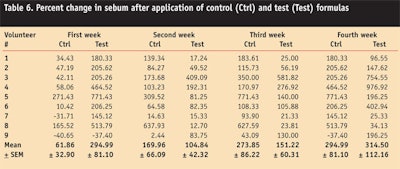
Sebum Content
Sebaceous glands, located in each hair follicle, produce sebum to lubricate and protect the skin.33 Sebum production is measured using a special opalescent plastic filmd that becomes transparent when it comes in contact with sebum lipids.42
A probe is used to press a piece of the film onto the skin for a measured length of time and sebum is adsorbed onto the film, similarly to ink on blotting paper, thus turning the film transparent.
The probe is then placed into a device that radiates a light beam onto the film. A metal mirror behind the film reflects the beam back again through the film and into an instrument called a photomultiplier, which measures the amount of light in the beam. The more sebum on the skin, the more transparent the film and the greater the amount of light reflected.
In the present study, the effects of the control and test formulations on sebum content in human facial cheeks were investigated. Sebum was measured weekly in all test subjects using the control and test formulations; both samples were found to increase sebum content from the first to fourth week of the study period.
An ANOVA test confirmed a statistically significant effect of the control and test formulation on skin sebum throughout the study period. By applying an LSD test, it was evident that significant changes in sebum content were observed at different time intervals after application of both formulations (see Table 6).
With a paired sample t-test it was found that the control and test formulations showed insignificant variations regarding the skin sebum content. Thus, researchers attributed the increase in sebum content to the oily nature of the w/o emulsions, since they contained paraffin oil.14
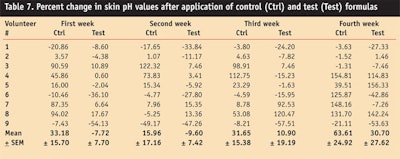
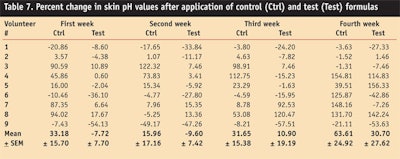
pH Values
The pH of skin ranges between 5 and 6; 5.5 is considered to be the average pH of skin. Therefore, formulations intended for application on skin should have a pH level close to this range.28
pH values in the volunteers’ skin were measured at different time intervals before and after the application of the control and test formulation for a period of four weeks (see Table 7).
After application of the control, pH values of the skin was increased throughout the study period, whereas application of the test formulation decreased pH values during the first and second week, and increased during the third and fourth week.
ANOVA testing found a significant effect on skin pH values produced by the control, whereas an insignificant effect was observed by the test formulation. A paired sample t-test concluded that the change in skin pH in volunteers by both the test and control formulation was significant in the first and fourth week.
The absence of change in the pH of skin from the test formulation could be attributed to the presence of potent natural antioxidants in the grapefruit extract,32 which can prevent the oxidative degradation of skin by scavenging free radicals, thus maintaining the natural integrity of human skin.
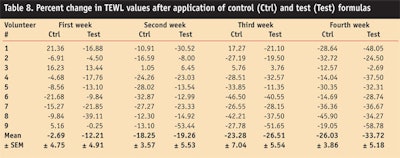
TEWL
Body loses water by constant evaporation through the skin, also known as trans epidermal water loss (TEWL).42 TEWL changes are related to SC water-binding capacity.42 Water makes up 70–75% of the basal layer weight but only 10–15% of the SC. If the water content of the SC falls below 10%, it becomes dry, less flexible and prone to damage, breakdown and infections.43
In the present study, a decrease in TEWL values was noted after the application of the control and test formulation throughout the 28-day study (see Table 8).
ANOVA testing confirmed that changes in TEWL produced by both formulations were statistically significant during the four-week study. By applying an LSD test, the change in TEWL values became significant for both formulations after the first week of application. With a paired sample t-test, insignificant variation in TEWL was found with respect to the control and test formulation throughout the study.
Researchers concluded that both formulations prevented TEWL probably due to a number of factors including their glycerin and paraffin oil content. Glycerin moisturizes the full thickness of the SC24 and paraffin oil, as mentioned previously, forms an occlusive covering on skin thus preventing TEWL. Therefore, due to moisture-retaining properties, both formulations enhanced the SC’s ability to attract, hold and redistribute water, in turn reducing TEWL.24, 34, 44
Panel Test
A questionnaire was given to each volunteer for sensory evaluation of each of the two creams. Average points were calculated for both the control and test formulations (see Figure 1).
The average points for ease of application were 4.12 and 4.08 for the control and test formulation, respectively, indicating ease of application on the skin. Points for spreadability rated the test formulation higher—4.29 for the control and 4.42 for the test formulation. Feel upon application was rated as 3.25 points for the control and 3.54 for the test formulation, an indication the test formulation felt better upon application to skin.
Average points for the 28-day application period were 3.43 and 3.5 for the control and test formulation, respectively. The numbers indicated the test formulation produced a more pleasant feeling; there also was no irritation on the skin in both formulations thus they were assigned 0.0 points for irritation by all volunteers.
Shine on skin was rated 3.35 for the control and 3.36 for the test formulation. This was expected since the two formulations contained the same quantity of paraffin oil. Similarly, the control produced a higher rating for softness of the skin; the average points was 4.74 for the base and 4.63 for the test formulation.
From a paired sample t-test, an insignificant difference was noted between the average points of sensitivity for both formulations. Thus, researchers concluded that little variation existed between the control and test formulation in regards to sensory evaluation. Both creams behaved similarly, from a sensory point of view.
Conclusion
Antioxidant-rich grapefruit extract, when used at a concentration of 1% in topical creams, was not found to realize its full skin benefit potential in a 28-period of application. It is therefore suggested, to maximize the cosmetic benefits, that the grapefruit extract be used in higher concentrations and for longer period of time.
References
1. JPF Macedo et al, Micro-emultocrit technique: A valuable tool for determination of critical HLB value of emulsions, AAPS Pharm Sci Tech 7(1) (2006)
2. K Welin-Berger, Formulations, release and skin penetration of topical anesthetics, Dissertation for the Degree of Doctor of Philosophy (Faculty of Pharmacy), Uppsala University (2001) pp 17–18
3. F Nielloud, G Marti-Mestres and MM Gilberte, Pharmaceutical Emulsions and Suspensions, New York: Marcel Dekker (2007)
4. G Kutz, P Biehl, M Waldmann-Laue and B Jackwerth, On the choice of oil-in-water emulsifiers for use in skin care products for sensitive skin, SOFW J, 123 145–150 (1997)
5. TO Ngai, SH Behrens and H Auweter, Novel emulsions stabilized by pH and temperature sensitive microgels, The Roy Soci of Chem (2004)
6. US Pat 7,138,128, Preparations of the W/O emulsion type with an increased water content, and comprising cationic polymers, B Andreas,
K Rainer and Schneider Gu, assigned to Beiersdorf AG (Hamburg, Germany) (Nov 21, 2006)
7. NV Yanishlieva, E Marinova and J Pokorny, Natural antioxidants from herbs and spices, Eu J of Lip Sci and Tech 108 (9)776–793 (2006)
8. D Roberts, Antioxidant values in fruits and vegetables, Macular Degeneration Support Web site; www.mdsupport.org (Accessed Feb 27, 2007)
9. L Curtis, Juice Up, Northbrook, IL USA: Virgo Publishing (1997)
10. FP Mary et al, A furanocoumarin-free grapefruit juice establishes furanocoumarins as the mediators of the grapefruit juice–felodipine interaction, Ameri J of Clinic Nutr, 83(5) 1097–1105 (2006)
11. Grapefruit, Wikipedia, the free encyclopedia Web site; www.en.wikipedia.org/wiki/grapefruit#column-one; Wikimedia Foundation Inc. 15(59) (Accessed Mar 28, 2007)
12. British Pharmacopoeia 2 (2004) pp 1132–1134
13. Code of Federal Regulations, U.S. Food and Drug Administration, Sec.146.132, Grapefruit juice, Department of Health and Human Services, U.S. Government Printing Office 2 (2002) pp 439–440
14. RC Rowe, PJ Sheskey and PJ Weller, Dimethicone, mineral oil, wax white; wax yellow, Handbook of Pharmaceutical Excipients, 4th ed, Chicago: The PhP London, and Washington, D.C.: the APhA Washington (2003) pp 213–214, 395–396, 687–690
15. J Swarbrick, JT Rubino and OP Rubino, Coarse dispersions, in Remington: The science and practice of pharmacy, 21st edn, Philadelphia: Lippincot Williams and Wilkins (2006) pp 316–334
16. NM Mostefa, AH Sadok, N Sabri, A Hadji, Determination of optimal cream formulation from long-term stability investigation using a surface response modelling, Int J of Cosmet Sci, 28 (3) 211–218 (2006).
17. M Muehlbach, R Brummer and R Eggers, Study on the transferability of the time temperature superposition principle to emulsion, Int J of Cosmet Sci, 28(2) 109–116 (2006)
18. HA Lieberman, MM Rieger and GS Banker, Pharmaceutical Emulsions, in Pharmaceutical Dosage Forms: Disperse Systems, New York and Basel: Marcel Dekker 1 (1988) pp 199–240, 285–288
19. L Song, X Ge, M Wang and Z Zhang, Direct preparation of silica hollow spheres in a water in oil emulsion system: The effect of pH and viscosity, J of Non-Cry Sol 352(21–22) 2230–2235 (2006)
20. L Lachman, HA Lieberman and JL Kanig, in Emulsions, The Theory and Practice of Industrial Pharmacy, 3rd edn, Bombay: Varghese Publishing House (1990) p 502
21. A Abdelbary and SA Nour, Correlation between spermicidal activity and haemolytic index of certain plant saponins, Pharmazie 34(9) 560–561 (1979)
22. K Tauer, Emulsions, in MPI Colloids and Interfaces, D-14476 Golm, Germany: Am Mühlenberg (2006)
23. ST Mabrouk, The preparation and testing of a common emulsion and personal care product: Lotion, J of Chem Edu 81(1) 83–86 (2004)
24. TL Diepgen, Professional care for dry and sensitive skin, Medical and economic costs of skin disease, in White Book of Dermatology, Heidenberg, Germany: European Dermatology Forum, University of Heidenberg (2005)
25. Grapefruit, International Cyber Business Services Inc; www.holisticonline.com (2000)
26. TE Wallis, Text book of Pharmacognosy, 5th edn, New Delhi: CBS publishers, (2004) p194
27. A Prakash, Antioxidant activity, What are antioxidants? Medallion Laboratories 19(2) (2001)
28. N Akhtar, Formulation and evaluation of a cosmetic multiple emulsion system containing macademia nut oil and two antiaging agents, Dissertation for the Degree of Doctor of Philosophy, the Department of Pharmaceutical Technology, Anadolu University (2001) pp 104,107
29. P Shoukat et al, Survey and mechanism of skin depigmenting and lightening agents, Wily Int Sci J 20(11) 921–934 (2006)
30. DG Meyers and PA Maloley, Safety of antioxidant vitamins, Arch of Int Med 156(9) 925–935 (1996)
31. CD Villarama and HI Maibach, Glutathione as a depigmenting agent: An overview, Int J of Cosmet Sci 27(3) 147–153 (2005)
32. D Roberts, Antioxidant Values in Fruits and Vegetables, Macular Degeneration Support Web site; www.mdsupport.org (Accessed Feb 15, 2007)
33. G Mateljan, Grapefruit, The World’s Healthiest Foods, Honolulu, Hawaii: McGraw Hill Professional, Science and Technology Encyclopedia, www.healthline.com (2007)
34. JN Kraft and CW Lynde, Moisturizers: What They Are and a Practical Approach to Product Selection, Skin therapy letter, indexed by the U.S. National Library of Medicine 10 (2005)
35. R Hata and H Senoo, L-ascorbic acid 2-phosphate stimulates collagen accumulation, cell proliferation and formation of a three dimensional tissue like substance by skin fibroblasts, J Cell Physiol, 138(1) 8–16 (1989)
36. S Murad, D Grove, KA Lindberg, G Reynolds,
A Sivaraja and SR Pinnel, Regulation of collagen synthesis by ascorbic acid, Proc Natl Acad Sci USA 78 2879–2882 (1981)
37. JC Geesin, D Darr, R Kaufmann, S Murad and SR Pinnel, Ascorbic acid especially increases type 1 and type III procollagen messenger RNA levels in human skin fibroblast, J Invest Derma 90(4) 420–424 (1988)
38. SR Pinnel, S Murad and D Darr, Induction of collagen synthesis by ascorbic acid: A possible mechanism, Arch Derma 123(12) 1684–1686 (1987)
39. K Scharffetter-Kochanek et al, Photoaging of the skin from phenotype mechanisms, Exp Gerontol 35(3) 307–316 (2000)
40. SJ Padayatty and M Levine, New insights into the physiology and morphology of vitamin C, Canad Med Assoc J 164(3) 353–355 (2001)
41. M Ponec et al, The formation of competent barrier lipids in reconstructed human epidermis requires the presence of vitamin C, J Invest Derm 109(3) 348–355 (1997)
42. J Gray, The world of skin care, ed 1, London: Macmillan Press (2000) p 7
43. D Mitsushiro, Influence of drug environment on epidermal functions, J Derm Sci 24(1) 22–28 (2000)
44. HK Biesalski and JUC Obermueller, UV light b-carotene and human skin beneficial and potentially harmful effect, Arch Biochem Biophys 389(1) 1–6 (2001)