Skin cells, in particular keratinocytes and fibroblasts, are the cells most exposed to environmental stressors such as UV light, smog, toxins, cigarette smoke and pollution. Once these cells are damaged, skin loses firmness and vigor, becoming weaker and more vulnerable to diseases. Skin inflammation is indeed one of the most serious consequences of environmental stress.1 Although inflammation is the first line of defense against microorganisms and external assault, it can also lead to visible signs of aging. In fact, during the inflammatory process, free radicals are generated, blood flow increases and immune cells are attracted by chemical signals to the site of injury.2, 3
In typical cases, when the inflammatory response is no longer required, the physiological events are rapidly terminated. However, in the case of prolonged and persistent environmental stress, this termination process fails, leading to chronic inflammation characterized by the continuous production of free radicals and further causing DNA damage, wrinkles and skin aging.2, 3
DNA integrity is the most important prerequisite for the fulfillment of vital cellular functions: The loss of functionally important genes compromises the health of cells and accelerates the skin aging process.4 Recent studies have demonstrated that in the complex scenario of signal transduction mechanisms activated to protect cells, there are elements that play pivotal roles to guarantee DNA integrity. Among these elements is the family of GADD45 proteins, which assists cells by preventing accumulated mutations in the nuclear DNA. These proteins act as stress sensors and are able to mediate a variety of interactions among different cellular components, triggering processes of DNA repair and protection.5
Other elements that maintain DNA integrity and stability are sirtuins—proteins that regulate cell longevity. Sirtuins are present in all multicellular organisms and regulate the activity of transcription factors and DNA-binding proteins, acting as nucleus stabilizing factors.6
Raspberry Extracts
As reported in literature by other researchers, several raspberry varieties belonging to the genuses Rubus, Fragaria, Vaccinium, Ribes, Aronia and Sorbus contain bioactive compounds that are able to attenuate skin inflammation and reduce the damage caused by reactive oxidative agents.7 Indeed, a wide spectrum of anthocyanins are present in raspberry extracts and have been associated with anti-inflammatory activity;8 the extracts also contain vitamin C and phenolics, which have a high free radical scavenging capacity. Moreover, raspberry extracts contain a high level of ellagitannins, which upon hydrolysis release ellagic acid, conferring antiviral properties and protection against certain forms of cancer.9 Finally, the extracts include the important compound resveratrol, a polyphenol whose activity has been linked to anticarcinogenic effects and the regulation of low-density lipoproteins, mediated by sirtuin activation.10
Among all the Rubus species analyzed, the wild red raspberry variety Rubus idaeus contained the highest number of anthocyanins and highest level of antioxidant compounds.11 Based on these characteristics, the authors chose wild red raspberry as the source of stem cells to develop a new cosmetic active ingredient. The leaves of this plant were used as starting material to produce stem cells, whose extract had interesting cosmetic applications.
Extract Preparation
A stem cell extract was prepared as described below, starting from the leaves of wild red raspberry since they showed the highest antioxidant content of all the plant organs. This is because pigments and other antioxidant molecules involved in photosynthesis are only present in the leaves.
Cultured stem cells were produced in a laboratory to closely control growth conditions and to limit contaminants and other biological pathogens such as viruses and bacteria. Preparing a product from sterile cultured cells standardizes the protocol, guaranteeing a reproducible yield from each batch and the desired characteristics of quality. Cosmetic active ingredients obtained from any raspberry plant organ exhibit variations in the content and quality of beneficial phenolics, anthocyanins and flavonoids,12 compared with the controlled stem cells that are consistent in content. The content and quality of these compounds also are dependent upon the fruit variety and even the age of the plant used.13 Moreover, the presence of contaminants and pollutants in the fruits, such as heavy metals, pesticides, mycotoxins, etc., may alter the quality of the product. Thus, the use of cultured raspberry stem cells to obtain the soluble extract also controls health risks to consumers.
Preparation protocol: Rubus idaeus leaves were surface sterilized in 70% EtOH for 15 min and 1% bleach for an additional 15 min, then washed three times with sterile water. The leaf explants were each cut into 0.5-cm pieces, wounded with a scalpel blade, and placed on medium AB1a supplemented with 500mg/L myo-inositol, 30mg/L sucrose, 1 mg/L of 2,4-Dichlorophenoxyacetic acid (2,4-D), 0.1mg/L kinetin, 1mg/L adenine and 7.5mg/L plant agar for callus induction.
White and friable callus was obtained after five weeks of cultivation in the dark. The callus cultures were maintained by transferring the tissues onto fresh AB1a medium every four weeks. To initiate stem cell liquid culture, 50 mg of 40–45-day-old-callus were resuspended in 25–30 mL AB1a liquid medium. The culture was incubated at 27°C in the dark under constant orbital stirring (110 rpm). The callus tissue gradually disintegrated and formed single cells or small cell aggregates after 10 days.
The suspensions were then transferred into a larger volume, where 50 mL of a dense culture was used as the starting material to inoculate 1 L of culture medium. After seven days, the cells were collected and filtered through a layer of Miracloth fabricb. The drained cells (approx. 300 g) were then resuspended in 300 mL of PBS (136 mM NaCl, 2.7 mM KCl, 12 mM NaH2PO4 and 1.76 mM KH2PO4, pH 7.4) and homogenized in a mortar. The resulting lysate was centrifuged at 10,000 rpm to precipitate the particulate fraction and isolate the soluble components. The supernatant was then collected and lyophilized and the powder obtained (approx. 4 g) was dissolved in water at the required concentrations.
Antioxidant Profile
As noted, the raspberry stem cell extract was prepared and the total homogenate centrifuged at high speed to remove all insoluble/particulate components and to ensure that only soluble material contributed to the extract composition. The extract obtained was analyzed by mass spectrometry liquid chromatography (MS-LC) (Figure 1) as described, which showed the extract to be rich in phenolic acids and flavonoids, the most abundant being ferulic acid and quercitin ramnoside.
MS-LC protocol: The chromatographic separation of phenolic compounds was performed using an HPLC apparatusc, a UV/VIS detectord set at 280 nm, and a 100 Å-columne (250 x 4.6 mm, particle size 5 μm). The eluents were 0.2% formic acid in water, and acetonitrile/methanol (60:40 v/v). The gradient program was as follows: 20–30% acetonitrile/methanol, 6 min; 30–40% acetonitrile/methanol, 10 min; 40–50% acetonitrile/methanol, 8 min; 50–90% acetonitrile/methanol, 8 min; 90–90% acetonitrile/methanol, 3 min; and 90–20% acetonitrile/methanol, 3 min, at a constant flow of 0.8 mL/min. The LC flow was split and 0.2 mL/min were sent to the mass spectrometry; the injection volume was 20 μL and three injections were performed for each sample. MS and MS/MS analyses of Rubus idaeus cell extracts were performed on a mass spectrometerf equipped with an ion source working in the negative mode.
For the anthocyanins, the chromatographic separation of phenolic compounds was performed using the same HPLC apparatusc and a UV/VIS detectord set at 520 nm; a 4.6 x 250 mm, 5 mm columng was used for separation. Elution was performed using the mobile phase A, a 5% aqueous formic acid solution; and mobile phase B, methanol. The flow rate was 1 mL/min. The gradient used was: 20% methanol, 0 min; 20% methanol, 2 min; 40% methanol, 10 min; 60% methanol, 15 min; 70% methanol, 20 min; and 20% methanol, 33 min. MS and MS/MS analyses of Rubus idaeus cell extracts were again performed on a mass spectrometerf equipped with an ion source, this time working in the positive mode.
Analysis of higher molecular weight phenolic compounds and anthocyanin content revealed the presence of ellagitannins and ellagic acid derivatives as well as pelargonidine glucoside, which were also previously shown to be present, although differentially distributed, in the extracts of raspberry fruits, leaves and flowers.14, 15
Effects on Inflammatory Genes
The presence of flavonoids and anthocyanins in the extract suggested a potential anti-inflammatory effect since these compounds have been found to repress genes relevant to inflammation.16 Thus, after treatment of macrophages with 0.0025% of the Rubus idaeus stem cell extract, the authors analyzed the expression of the COX2 and iNOS2 genes, which are involved in the pro-inflammation response.17
As shown in Figure 2, the induction of these genes caused by the bacterial inflammatory agent lipopolysaccharide (LPS) was significantly attenuated by the presence of the raspberry stem cell extract, suggesting a potential anti-inflammatory effect on skin.
Antioxidant Properties
The ability of the extract to genetically reduce the inflammatory response, coupled with the presence of high phenolic content, further suggested the extract would have a good antioxidant activity. The process of inflammation in the skin is in fact linked to the production of ROS in the cells and many compounds having high antioxidant power have also been successfully used to attenuate the inflammatory process in the skin. Therefore, to calculate the antioxidant power of the Rubus idaeus stem cell extract, the authors performed in vitro total antioxidant capacity (TAC) and oxygen radical absorbance capacity (ORAC) assays, as well as an ROS “in cell” assay.
The TAC and ORAC assays measure the total reducing power of a given compound, providing information on its potential protective effect on the skin. The ROS assay provides information about the ability of the compounds to induce antioxidative response in the cells; it can also provide clues to protective effects of the compounds in cells, as well as their capacity to keep ROS levels low inside the cell.
TAC and ORAC protocols: The TAC assay is based on a redox reaction between a test compound and copper II, Cu(II). If a test compound has reducing power, the Cu(II) is readily converted into Cu(I), which can be monitored by the addition of a chromophore, Bathocuproine (BATO). Fifty µL of Rubus idaeus cell extract in water were aliquoted into a 96-well plate, 50 µL of a BATO solution (360 µM) was added to each well, and the background absorbance at 490 nm was measured. Then, 25 µL of 100 µM CuSO4 solution was added to each sample and incubated at RT for 30 min. At the end of the incubation period, the absorbance at 490 nm was measured by a plate reader. As a reference standard, scalar dilutions of CuCl were used, ranging from 10 mM to 0.15 mM.18
The ORAC assay is based on the ability of a test compound to inhibit the oxidation of a fluorophore, generally fluorescein, by a potent oxidant, 2,2'-azobis(2-amidinopropane) dihydrochloride (AAPH).19 Twenty-five µL of Rubus idaeus cell extract dilutions in 75 mM of phosphate buffer, pH 7.4, were aliquoted into a 96-well plate and 150 µL of fluorescein solution, 8.5 nM in phosphate buffer, was added to each sample. After incubation at 37°C for 15 min, 25 µL of AAPH solution (153 mM in phosphate buffer) was piped into each well and the progress of the reaction monitored at 535 nm using a fluorescence multi-well reader. The fluorescence was measured every minute for 40 min and the antioxidant power of the mixture was calculated according to the method described by Huang.19 The net area under the curve (AUC) of the samples and standards, represented by different dilutions of 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acidh was calculated.
The standard curve was obtained by plotting 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acidh concentrations against the average net AUC of the two measurements for each concentration. Net AUC was obtained by subtracting the AUC of the blank from that of the sample or the standard. ORAC values of the samples were expressed as micromoles of 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acidh equivalents per liter.
Figure 3 shows the results of the TAC and ORAC assays, which indicate the extract has excellent antioxidant power—higher than that calculated for resveratrolj, which also is known for good antioxidant and cell protection properties.20, 21
Cytotoxicity: Before initiation of the ROS in-cell assay, the authors determined the correct dose to be used via a cytotoxicity test. Different concentrations of the Rubus idaeus extract ranging from 1% to 0.001% were tested on cultured fibroblasts and the vitality rate determined. None of the tested doses showed any toxic effects on the cells (data not shown). When fibroblasts were treated with 0.01% and 0.002% of stem cell extract, the oxidative stress induced by ROS was reduced (Figure 4). This effect was comparable to that of ascorbate, a well-known antioxidant protective agent.
DNA protection: On the basis of these antioxidant proprieties, the authors further evaluated the capacity of the extract to induce the expression of genes involved in DNA protection. As previously described, the GADD45α gene, a member of GADD45 transcription factor family,22 and the SIRT-1 gene, the most characterized member of the sirtuin protein family,23 were chosen for the study. Fibroblasts were treated with two different concentrations of the raspberry extract, 0.0025% and 0.0005%; ascorbate at 0.0044% (corresponding to 250 µM) and resveratrol at 0.5%, both as positive controls; and with and without 150 µM H2O2, which was used to induce stress on the fibroblasts and increase the basal level of GADD45.
As shown in Figure 5a, the Rubus idaeus stem cell extract induced significant expression of the GADD45α gene. In the presence of H2O2, where the expression of this gene was higher than in the unstressed cells, the induction effect of the extract was more evident and even greater than that produced by ascorbate and resveratrol. This is mainly due to the fact that the extract is a mixture of compounds; i.e., besides the presence of antioxidants such as resveratrol and phenolics, it also contains proteins and sugars, which may activate defensive signaling responses in the cell.
Analyzing the SIRT-1 gene, the authors observed that the Rubus idaeus stem cell extract, again at 0.0025% and 0.0005%, induced a 60% increase in its expression level (Figure 5b). In the presence of H2O2, which in this case inhibits SIRT-1 expression, the extract reduced the inhibitory effect caused by H2O2, confirming its protective effect against oxidative stress.
Finally, the authors verified whether the treatment of skin cells with the extract had actual effects on nuclear DNA protection. Such information can be obtained from the Comet assay, which measures the integrity level of nuclear DNA. Oxidative stress causes a fragmentation of the nuclei, which can be easily detected under the microscope after DNA staining.
Comet assay: Fibroblasts maintained in DMEM with 10% fetal calf serum under 5% CO2 at 37°C were plated at density of 1.5 x 105 per well in six-well plates. After 3 hr of incubation with the Rubus idaeus cell extract or other reagents, cells were treated with 150 μM H2O2 for 2.5 hr and detached from the plate using 1 mL/well of non-enzymatic detaching solutionk. Cells were transferred to Eppi-tubes, spun at 1.5k and washed once with phosphate buffer saline (PBS).
The cell pellet was resuspended in 10–20 mL of PBS depending upon the amount of cells recovered. Eighty mL of low melting point agarose (LPMA) 0.5% in PBS and equilibrated at 37°C was added to each tube, and the solution immediately dropped onto a normal melting agarose (NMA) pre-coated slide. Coverslip slides were placed on the top, without squeezing the cells, and the slides put on a tray in a refrigerator until the agarose layer hardened (10–15 min). Coverslips were then gently slid off without scraping the agarose layer containing the cells.
The slides were placed in cold Lysis solution for at least 2 hr at 4°C, then gently removed from the solution and placed in an electrophoresis tank filled with cold electrophoresis buffer; slides were left in alcaline buffer for 10 min to allow DNA unwinding and the expression of alkali-labile damage, then power supply was turned on to 24 volts. Slides were electrophoresed for 20 min and at the end placed in cold neutralization buffer for at least 10 min. After drying the slides, the cells were stained with a 10 ug/mL solution of ethidium bromide, covered with a coverslip and scored at a fluorescence microscope.
As shown in Figure 6, treatment with the extract clearly produced a protective effect on DNA from H2O2-induced damage. The comet tail length, an index of DNA fragmentation, was reduced by 56% in the cells pre-treated with the extract at 0.005% and this effect was even higher than that produced by resveratrol at 0.5%. This final effect observed on DNA protection was also the result of a series of events in the cells as activated by the compounds present in the extract.
Formulating Guidelines
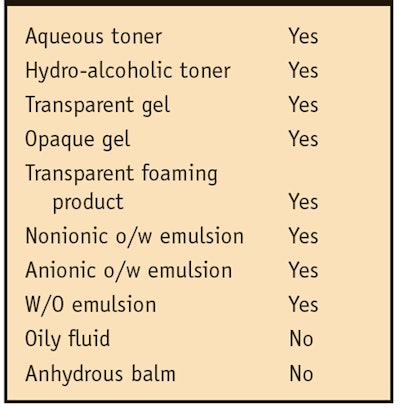
Since the described Rubus idaeus extract is composed of hydrosoluble material, it is fully soluble in aqueous medium, glycerin and ethanol. Also, as established by laboratory tests, the Rubus stem cell extract is stable at pH values ranging from 4.0 to 8.0 (data not shown) and can be added to a preparation either during the cooling phase or at the end of the formulation, as it withstands temperatures of up to 70°C. Due to its hydrophilic nature, the material is difficult to incorporate into very hydrophobic systems, such as lipogels, synthetic oils and silicones; however, it can be easily incorporated in polar or weakly polar solvents, which represent the majority of ingredients used in cosmetic formulations (Table 1).
Conclusion
In the described study, the authors discuss the development of a stem cell extract derived from Rubus idaeus cell cultures grown in liquid medium under controlled conditions. The hydrosoluble cell extract was shown to exhibit anti-inflammatory activity, mainly due to the presence of high levels of flavonoids and anthocyanins. In addition, the extract provided high antioxidant power due to the presence of natural phenolic components and induced the genes responsible for DNA protection and repair—more so than resveratrol. This stem cell culture extract could find interesting applications in skin care to promote healthy skin; for example, as shown in Formula 1 and Formula 2.
References
- S Briganti and M Picardo, Antioxidant activity, lipid peroxidation and skin diseases. What’s new? J Eur Acad Dermatol Venereol 17(6) 663–669 (2009)
- HY Ha, Y Kim, ZY Ryoo and TY Kim, Inhibition of the TPA-induced cutaneous inflammation and hyperplasia by EC-SOD, Biochem Biophys Res Comm 348 450–458 (2006)
- CR Thornfeldt, Chronic inflammation is etiology of extrinsic aging, J Cosmetic Dermatol 7(1) 78–82 (2008)
- S Pillai, C Oresajo and J Hayward, Ultraviolet radiation and skin aging: Roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation— A review, Int J Cosmet Sci 27(1) 17–34 (2005)
- B Hoffman and DA Liebermann, Gadd45 modulation of intrinsic and extrinsic stress responses in myeloid cells, J Cell Physiol 218(1) 26–31 (2009)
- S Lavu, O Boss, PJ Elliott and PD Lambert, Sirtuins–Novel therapeutic targets to treat age-associated diseases, Nature Rev 7 841–853 (2008)
- S Hakkinen, M Heinonen, S Karenlampi, H Mykkanen, J Ruuskanen and R Torronen, Screening of selected flavonoids and phenolic acids in 19 berries, Food Res Int, 32 345–353 (1999)
- S Zafra-Stone, T Yasmin, M Bagchi, A Chatterjee, JA Vinson and D Bagchi, Berry anthocyanins as novel antioxidants in human health and disease prevention, Mol Nutr Food Res, 51 675–83 (2007)
- MR Loizzo et al, Antioxidant and antiproliferative activity of Diospyros lotus L. extract and isolated Compounds, Plant Foods Hum Nutr, 64 264–270 (2009)
- Baur et al, Resveratrol improves health and survival of mice on a high-calorie diet, Nature 444 337–342 (2006)
- KR Mtt-Riihinen, AK Eldin and AR Trrnen, Identification and quantification of phenolic compounds in berries of Fragaria and Rubus species (family Rosaceae), J Agric Food Chem 52(20) 6178–6187 (2004)
- M Liu, XQ Li, C Weber, CY Lee, J Brown and RH Liu, Antioxidant and antiproliferative activities of raspberry, J Agric Food Chem 50 2926–2930 (2002)
- D Bagchi et al, Safety and whole-body antioxidant potential of a novel anthocyanin-rich formulation of edible berries, Mol Cell Biochem 281(1–2) 197–209 (2006)
- J Gudej and M Tomczyk, Determination of flavonoids, tannins and ellagic acid in leaves from Rubus L. species, Arch Pharm Res 27 1114–1119 (2004)
- SY Wang and HS Lin, Antioxidant activity in fruits and leaves of blackberry, raspberry and strawberry varies with cultivar and developmental stage, J Agric Chem 48 140–146 (2000)
- S Lee et al, Inhibitory effects of flavonoids on TNF-alpha-induced IL-8 gene expression in HEK 293 cells, BMB Rep 42(5) 265–70 (2009)
- YQ Zhang et al, Elevation of inducible nitric oxide synthase and cyclooxygenase-2 expression in the mouse brain after chronic nonylphenol exposure, Int J Mol Sci 9 1977–88 (2008)
- L Yildiz, K Sozgen, E Tutem and R Apak, Combined HPLC-CUPRAC (cupric ion reducing antioxidant capacity) assay of parsley, celery leaves and nettle, Talanta 77 304–313 (2008)
- D Huang, B Ou, M Hampsch-Woodill, JA Flanagan and RL Prior, High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format, J Agric Food Chem, 50 4437–4444 (2002)
- R Mikstacka, AM Rimando, E Ignatowicz, Antioxidant effect of trans-Resveratrol, Pterostilbene, Quercetin and their combinations in human erythrocytes in vitro, Plant Foods Hum Nutr (Jan 27, 2010)
- C Lucas-Abellan, MT Mercader-Ros, MP Zafrilla, MI Fortea, JA Gabaldon and E Nunez-Delicado, Orac-fluorescein assay to determine the oxygen radical absorbance capacity of resveratrol complexed in cyclodextrins, J Agric Food Chem 56 2254–2259 (2008)
- M Takekawa and H Saito, A family of stress-inducible GADD45-like proteins mediates activation of the stress-responsive MTK1/MEKK4 MAPKKK, Cell 95(4) 521–530 (1998)
- B Zschoernig and U Mahlknecht, Sirtuin 1: Regulating the regulator, Biochem Biophys Res Comm 376 251–255 (2008)