Today, nanotechnology is recognized as enabling the manipulation of matter at the atomic and molecular level to create new materials with functional characteristics that are markedly different from conventional materials.1 Nanoemulsions, for example, can be used to control the rate at which assets are delivered to the skin. This control is achieved by incorporating materials within the nanoemulsion according to their compatibility with it, which obviously relies on their physical-chemical structure. Indeed, a great deal of attention has been dedicated recently to colloidal systems for the delivery of active ingredients because they significantly reduce the side effects of drugs and increase their bioavailability.2
Besides delivery, nanoemulsions exhibit improved stability over conventional systems, such as liposomes or solid lipid nanoparticles, since their small particle sizes are less affected by gravity and less inclined to settle during storage, thus preventing flocculation and increasing shelf life.3 The particles also prevent coalescence due to their uniform shape. Furthermore, the significant thickness of the particle film, relative to the particle diameter, prevents its thinning or rupture; and wetting, spreading and penetration can be improved as a result of the low surface tension.4
The size of nanoemulsion particles is a key factor in their efficient transdermal delivery;5, 6 their large surface area and small size ensure uniform deposition on skin, thus increasing the rate of skin absorption and hydration potential.5 In order to reduce particle size, physical means are used. High pressure homogenization is the preferred technology since it can be used on a wide range of compositions. This pressure typically is applied in two stages—high pressure at first, followed by low pressure.7 High pressure homogenizers are manufactured by various companies and the end product will vary with the brand and model used.8
Besides pressure, the number of homogenization cycles or passes used during the two stages can affect the particle size in the end product. The chemical composition and concentration of emulsifier used can also impact the nanoemulsion.9 Finally, the temperature at which the nanoemulsion is homogenized influences its production; Aubrun et al. and Liedtke et al. observed that higher homogenization temperatures resulted in smaller particles.4, 8 Thus, the objective of the present work was to examine formulations prepared with similar oil fractions but different emulsifier systems and process parameters to determine why their behaviors differ. Understanding these dynamics would thus determine which conditions, among those studied, is optimal for formulating nanoemulsions.
Materials
O/W emulsions were prepared using three emulsifying systems with different molecular weights to evaluate their behavior in each system. The first consisted of stearyl alcohol polyethylene glycol ether etherified with ethylene oxide (steareth-2) and cetearylic alcohol polyethylene glycol ether (radicals derived from cetylic and stearylic alcohols) etherified with ethylene oxide (ceteareth-20). The second system consisted of inulin, a polymer blend of different fructose types, which was obtained from chicory and chemically modified by the insertion of lauric groups. The third system consisted solely of the emulsifier potassium cetyl phosphate, a potassium salt of a complex mixture of phosphoric acid esters and cetyl alcohol. The oil fraction used in all the three formulations was a mixture of stearyl alcohol, cetearyl alcohol, almond oil, caprylic/capric triglycerides, tocopherol acetate, and mineral oil. The aqueous phase was purified water and 0.1% EDTA (w/v); benzyl alcohol served as the preservative.
Methods
The water and oil phases used in the preparation of the nanoemulsions were mixed at 75°C. All samples were homogenized with the aid of a mechanical agitator equipped with a notched spindlea at 2000 rpm for 5 min, and also with a turbo-agitatorb at 14000 rpm for 3 min.
Following the mechanical agitation step, samples were cooled to room temperature (20–25°C) and transferred to a high pressure homogenizerc. Additional samples were prepared by varying the type of emulsifier, the first stage homogenizing pressure (60 MPa and 120 MPa), and the number of cycles to investigate the effects of these variables on nanoemulsion characterization (see Table 1).
Analysis of particle diameter, size distribution: Particle diameter and size distribution were determined by laser beam diffractiond in a module for fluids. All samples were diluted in watere (20 uL/2 mL) that was freed of air by ultrasound treatmentf for 10 min.
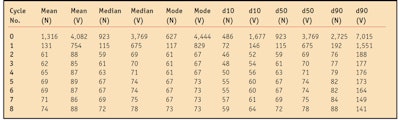
Statistical analysis: The determination of particle diameter and size distribution was performed in triplicate, in terms of number and volume, and is presented here in terms of volume. The statistical parameters used were the mean, median and mode, which represents the largest particle size cumulative found in 10%, 50% and 90% of the sample, respectively. Sample characterization was based on analysis of the distribution graphs of particle sizes combined with the interpretation of statistical parameters. Figure 1 shows a comparison of the particle size by volume and number of homogenization cycles at a pressure of 120 MPa for the emulsifiers used.
Viscosity testing: The viscosity of the samples was determined using a viscosimeterg at 20 rpm/20°C. Figure 2 shows the viscosity values for the ethoxylated system. Trials of the ethoxylated emulsifier system showed that the viscosity of the products tested decreased with an increasing number of passes, which may be linked to better compression of the particles of oil and wax inside the product tested. The viscosity results from tests conducted at a pressure of 60 MPa were higher than the viscosity at 120 MPa, and may be related to the particle size. Further detailed rheological analyses are necessary for a better understanding.
Results
Number of cycles: The number of cycles of homogenization was found to significantly affect the distribution of particle size, as shown in Figure 1 and Table 2. However, the data showed a plateau for the mean particle size and distribution after only three cycles, with an average size of approximately 70 nm.
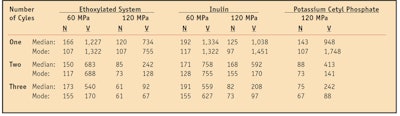
Particle size distribution: Particle size distribution results are expressed in terms of number and volume for the three test emulsifier systems, with and without homogenization. One of the objectives of the current study was to interpret the effect of the number of runs on particle size distribution, specifically with respect to the homogeneity of particle sizes present in the sample, as shown in Table 3.
As can be seen, the particle size was reduced as the number of homogenization cycles increased. This reduction in particle size was observed in all samples. The results also showed that the 60 MPa pressure produced predominantly larger particles (nm), when compared with particles obtained at 120 MPa.
Discussion
Number of cycles vs. particle size distribution: The present study showed that a single pass was not sufficient to significantly reduce the size of the particles. From the second homogenization cycle, however, the analysis to number and volume showed a reduction in particle size, in comparison with the first cycle. As might be expected, the results were consistent with literature findings indicating that a pressure of 60 MPa generated larger particles than 120 MPa; i.e., higher pressures are more effective at producing smaller particles.2, 8-10
Another important factor identified was that any further pass beyond the second cycle produced no significant reduction in particle size; the samples had stabilized, which is consistent with the literature.2, 8, 9
Pressure vs. particle size distribution: In the formulation using the ethoxylated emulsifier system, the average particle size resulting from three homogenization cycles at 60 MPa was 173 nm, in contrast to the much smaller average particle size of 120 nm produced in a single cycle under a pressure of 120 MPa. In terms of volume, three cycles at 60 MPa showed particles sizes with an average diameter of 540 nm, which is approximately six times larger than the average value obtained with the same number of cycles at 120 MPa (92 nm). Higher pressures also prove to be more effective since they require a lower number of cycles to decrease the particle size of nanoemulsions. This is a dynamic system that tends to reorganize itself more effectively by reducing the size of particles. Thus, both the size and distribution of the particles were found to be affected by the pressure applied to the system.
Emulsifier choice vs. particle size distribution: The present study also provided a clearer understanding of the behavior of different molecules in the high-pressure homogenization process, allowing for the determination of their feasibility for the production of nanoemulsions. The results showed that the sample using the ethoxylated emulsifier produced particles with the smallest diameters.
The sample based on inulin—highly effective as a surfactant because it is firmly absorbed on the micelles, in addition to being used in electrolyte-rich solutions and in even lower concentrations than ethoxylates11, 12—was found to be more effective than the sample using potassium cetyl phosphate, a molecule characterized by a relatively high molecular weight, which produced a particle size slightly larger than inulin in this process.
It seems there is little doubt that the characterization of a nanoemulsion is affected not only by the size of its molecules, but also by the emulsifier concentration in the nanoemulsion, by its ability to stabilize the o/w interface. In work on the influence of emulsifier types on high-pressure homogenization processes, Yuan et al. found that high hydrophilic-lipophilic balance (HLB) values provided better conditions for emulsifiers to stabilize the nanoemulsion system.7
Conclusions
Studies such as these are important to formulators during the exploration process. The present work demonstrates that ethoxylated emulsifier systems worked better in the preparation of nanoemulsions than the other two test systems. Generally, ethoxylated surfactants and simpler molecules of relatively low molecular weight produced the most satisfactory results for the formation of nanoemulsifiers. However, if the goal is not to create nanoemulsions, it is possible to obtain more homogeneous formulations at pressures of 60 MPa or even smaller. Higher pressures did prove to be more efficient for producing nanometer-scale particles with fewer passes, which could optimize time and resources. Finally, beyond a certain number of cycles—two or three, for the test samples in this study—high pressures did not improve the quality of the product to any significant extent.
References
Send e-mail to [email protected].
- K Neves, Nanotechnology in cosmetics, Cosm & Toil–Brazil, 20 (1) 22-7 (Jan/Feb, 2008)
- D Mou et al, Hydrogel-thickened nanoemulsions system for topical delivery of lipophilic drugs, Int J of Pharmaceuticals 353(1-2) 270-6 (Apr 2, 2008)
- TP Formariz, MCC Urban, AAJ Silva, PD Gremião and AG Oliveira, Microemulsions and crystalline liquid phases as drug delivery systems, Brazilian Pharmaceutical Sciences Review 41(3) 301-13 (Jul/Sep, 2005)
- OS Aubrun, JT Simonnet and F L’Alloret, Nanoemulsions: A new vehicle for skin care products, Advances in Colloid and Interface Science 108-109(1) 145-9 (May 20, 2004)
- RH Müller, RD Petersen, A Hommoss and J Pardeike, Nanoestructured lipid carriers (NLC) in cosmetic dermal products, Advanced Drug Delivery Reviews 59(1) 522-30, (May 17, 2007)
- AK Kohli and HO Alpar, Potential use of nanoparticles for transcutaneous vaccine delivery: Effect of particle size and charge, Int J of Pharmaceutics 275(1-2) 13-7 (Mar 19, 2004)
- Niro Soavi Inc., Niro Soavi: VHP Homogenizer Technology, available at www.niro-soavi.it (accessed May 1, 2008)
- S Liedtke, RH Wissing and K Mäder, Influence of high-pressure homogenization equipment on nanodispersions characteristics, Int J of Pharmaceutic 196(2) 183-5 (2000)
- Y Yuan, Y Gao, J Zhao and L Mao, Characterization and stability evaluation of β-carotene nanoemulsions prepared by high pressure homogenization under various emulsifying conditions, Food Research Int 41(1) 61-8 (2008)
- CP Tan and M Nakajima, β-carotene nanodispersions: Preparation, characterization and stability evaluation, Food Chem 92(4) 661-71 (Nov 23, 2004)
- TF Tadros, A Vandamme, B Levecke, K Booten and CV Stevens, Stabilization of emulsions using polymeric surfactants based on inulin, Advanced Colloid and Interface Science 108-109(1) 207-226 (May 20, 2004)
- TF Tadros, A Vandamme, K Booten, LB Evecke and CV Stevens, Stabilization of emulsions using hydrophobically modified inulin (polyfructose), Colloids and Surfaces A: Physicochemical and Engineering Aspects 250(1-3) 133-40 (Dec 10, 2004)