Editor's note: What’s the difference between the INCI name of a silicone surfactant and its properties? In this edition of “Comparatively Speaking,” Tony O’Lenick explains.
First, it is important to understand there are a plethora of silicone compounds available to the formulator. All of these function in formulations based upon their specific structure. This means International Nomenclature Cosmetic Ingredient (INCI) names, which are rather generalized, are of limited importance for choosing ingredients. Indeed, testing must be performed in formulations to ensure maximum effects.
Second, even small differenences in structure can have a profound effect upon formulations. This is important when one looks to aesthetics since critical effects are caused by surface active silicones interacting with the many raw materials in a formulation.
INCI names are designed for the purpose of providing a compliant label for cosmetic raw materials and the formulations that contain them. They are not designed to provide full structural or compositional information on the products they describe. This can make the task of duplicating a formulation based soley on the label very difficult.
It becomes even more complex if the formulation contains polymers. Polymers are the reaction products of monomers and their ratios can be altered to obtain desired characteristics in formulations.
Case Study: PEG-8 Dimethicone
One powerful example is provided by several closely related polymers, all of which are referred to as PEG-8 dimethicone. This structure is shown in Figure 1.1
In examining the structure, one can see the ratio of a to b clearly is important. As the ratio of a relative to b goes up, the water-soluble groups make up less of the total and the polymer becomes less water soluble. However, even when the ratio of a:b is kept constant, and the total values for both a and b increase, dramatic differences occur in some important properties. Consider that while the ratio of a to b units may remain the same, since the total number of both are increased, there is an overall increase in molecular weight.
These differences emerge in properties such as surface tension, aqueous foam and wetting times. Reductions in surface tension are required to get foam and wetting. Note that not all surface tension-reducing polymers result in good wetting and/or foam; however, all good foamers and wetters lower the surface tension. The following considers these properties.
Surface Tension
Surface tension can be conceptualized in various ways but perhaps the best description is to consider it the result of the forces of attraction existing between the molecules in a liquid. This is measured by the force per unit length acting in the surface at right angles to an element of any line drawn in the surface (mN/m).2
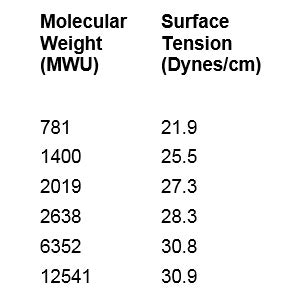
Table 1 and Figure 2 provide molecular weights and surface tension values for 0.1% w/w concentrations of different PEG-8 dimethicones in deionized water at room temperature. Measurements and determinations were made on a tensiometer.3
Foam Properties
Foam is a complicated physical phenomenon that is made up of at least two distinct phases: a continuous liquid phase in which there is a surfactant and other formula ingredients; and a gas phase, commonly air. In some instances, a third oil phase may be present.
The formula ingredients all interact with one other and any soil or impurities present, along with the physical methods used for agitation. This determines the foam properties. Another variable that affects foam structure is the container in which the foam is held. Indeed, many factors are responsible for foam quality, including some over which the formulator has no control; for example, the qualitative and quantitative aspects of the soil present on the hair will affect the foam properties delivered by a shampoo.
The bulk properties of foam are crucial to formulation success. Cosmetic elegance is determined by the size, shape and longevity of foam provided by formulations. Foam instability is even a factor, as the consumer expects the foam to be easily and effectively removed by rinsing once the action of the foam is complete. For these reasons, the ultimate determination of a particular foam’s suitability for a given formulation will be determined by the consumer. Therefore, salon testing is the best measure of foam quality.
Basic Foam Test
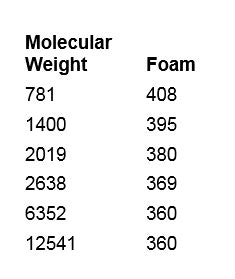
A cylinder shake test can be used as a basic means to assess foam.4 Here, a 1% sample in de-ionized water or other solution is shaken for 5 sec in a 100-mL graduated cylinder. The height of the foam is then measured initially after shaking, and after 1 min and 3 min of standing. Table 2 and Figure 3 illustrate the molecular weight vs. foam values of PEG-8 dimethicone.
Wetting Time
Wetting refers to the displacement of one liquid from a substrate surface by another liquid. In general, the latter liquid must have the same or a lower surface tension than the substrate; otherwise, no wetting is achieved. An equal or lower surface tension produces a positive spreading coefficient, which in turn leads to good wetting. Thus, lowering the surface tension enables the wetting of the substrate.5
Wetting Tests
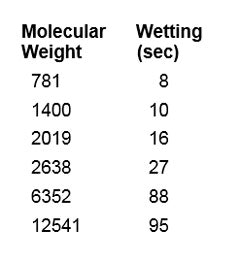
Measurements for wetting capabilities can be made using the Draves Wetting Test Method.6, 7 Table 3 and Figure 4 provide examples taken of PEG-8 dimethicone at 0.1% and room temperature, vs. molecular weight.
Conclusions
The addition of a silicone to a formulation must provide consumer-perceivable benefits in a cost-effective manner. This means the silicone polymer that provides the best properties at the lowest concentration must be identified, which requires product testing.
Fortunately, the foremost property needed for functionality is a reduction in surface tension, and this property is easily measured in formulations. This should be assessed for all formulations. Simply put, if there is no surface tension reduction, then satisfactory foam, wetting and aesthetics will not be achieved.