Tony O’Lenick explores the difference between germicidal and conditioning quats.
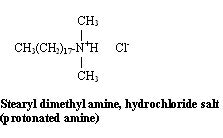
Quat is surfactant jargon for a quaternary ammonium compound. This class of compounds includes nitrogen-containing materials in which the nitrogen has four nonhydrogens surrounding them. Quats by virtue of their fatty group and their cationic group are substantive to hair, skin and cell walls of bacteria. These substrates are fatty in nature and generally have a negative charge, the result of oxidation of sulfur-bearing amino acids.
The nature of the interaction between hair, skin and bacteria cell walls is critical to the function of the quat at the surface. If the quat coats the hair and skin with minimal penetration, it is mild and conditioning. If the quat is made to maximize the disruption of the bacteria cell wall, it will be antimicrobial.
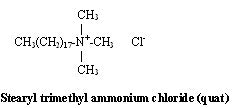
Antimicrobial quats are registered with the EPA and have very specific alkyl chains and an aromatic group. They are commonly encountered as swimming pool algaecides and hard surface spray disinfectants.
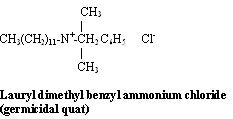
CH2 C6H5 is benzyl.
The introduction of an amido group into the molecule increases mildness and conditioning.
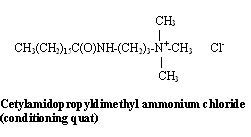
Conditioning quats provide very different cosmetic properties than do germicidal quats.










