While the Internet has provided consumers with a wealth of information on a number of important issues, it also has given way to misinformation and inaccuracies about cosmetics and personal care products and their ingredients. Preservatives, for example, have come under attack and led some consumers to seek “preservative-free” products. Yet the U.S. Food and Drug Administration (FDA) states, “Cosmetics need not be sterile, however, they must not be contaminated with microorganisms that may be pathogenic, and the density of non-pathogenic microorganisms should be low. In addition, cosmetics should remain in this condition when used by consumers.”1
Because all microorganisms require water for growth, most aqueous-based, multi-use products require preservation to protect against spoilage from bacteria, yeast and fungi. Conversely, non-aqueous personal care products such as dry powders or lipsticks present a low risk for growth of microorganisms during use.2 Other types of personal care products may be inherently hostile to the growth of microorganisms due to the nature of their ingredients or the type of packaging used, which may inhibit contamination.2
Preservatives play an important role in controlling a range of microorganisms, e.g., bacteria and fungi, that may be introduced into cosmetics and other personal care products during normal use by consumers. Growth of microbes in these products, as one Personal Care Products Council video shows,3 can adversely affect consumer health and the aesthetic or functional qualities of products.4, 5 Formaldehyde-donor (FD) preservatives are one family of ingredients whose chemistry, safety, efficacy, benefits and limitations have been widely studied for the control of microbial growth in personal care products. Their properties and efficacy are reviewed here.
FD Preservatives
FD preservatives used in personal care applications include DMDM hydantoin, imidazolidinyl urea, diazolidinyl urea, sodium hydroxymethylglycinate and quaternium-15, the first three of which are the most frequently used FD preservatives in the United States and Canada.6 The use of FD preservatives by the cosmetics and personal care industry has been established over several decades. In a 2010 frequency of use study on preservatives,6 formaldehyde donors were reported in approximately 15% of the 36,811 personal care product formulations submitted to the FDA’s Voluntary Cosmetic Registration Program (VCRP).7
FD preservatives contain a carrier molecule, i.e., a formaldehyde donor, that is chemically bound to formaldehyde and is designed to release it into the aqueous phase at a known rate. Within aqueous products, an equilibrium exists among the FD preservatives, free formaldehyde and hydrated formaldehyde (methylene glycol) that favors the methylene glycol form over free formaldehyde by 10,000 fold.8, 9 The FD preservatives release small amounts of formaldehyde over time rather than all at once, which helps maintain product integrity during use.10
Safety Assessments
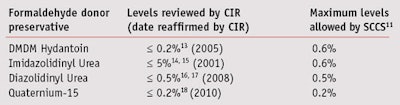
Commonly used FD preservatives have been reviewed by the Cosmetic Ingredient Review (CIR) Expert Panel11 in the United States and the Scientific Committee on Consumer Safety (SCCS, formerly the SCCP)12 in Europe (see Table 1). These reviews include scientific analysis of data published in the literature and unpublished data from preservative suppliers, among other sources. At the concentrations used in personal care products, FD preservatives have been deemed as safe; i.e., non-toxic, non-carcinogenic, non-mutagenic and not a concern for systemic toxicity. They also show a low incidence of eye and skin irritation, and contact dermatitis. Table 1 shows levels of commonly used FD preservatives determined to be safe for use in cosmetics.
Concerns about the adequacy of data on both dermal absorption and potential toxicity of quaternium-15 recently were raised by the SCCS in the European Union.19 While the CIR considered existing data sufficient for a safety determination, the SCCS requested additional dermal absorption data as well as toxicity studies in order to calculate a Margin of Safety for quaternium-15. Further, the SCCS stated the ingredient’s safety in cosmetic products could not be determined. Since that information has not been provided, the EU must decide whether action based on the SCCS opinion should be taken.
Occurrence and Measurement of Formaldehyde
Formaldehyde is a naturally occurring substance that is present in most life forms, including humans.20 All normally functioning cells in humans, animals and plants produce and use formaldehyde, but concentrations are so low that adverse health effects do not occur; for instance, human blood normally contains 2–3 ppm.21 Also, up to 100 ppm formaldehyde is reported to occur naturally in some types of foods.22 Formaldehyde does not accumulate in the body because it is metabolized quickly to sodium formate and is rapidly excreted. Similarly, it does not accumulate in the environment. When present in air, formaldehyde is broken down within a few hours by sunlight; in soil or water, bacteria initiate the breakdown.23
However, because carcinogenicity is associated with the inhalation of free formaldehyde gas,19, 20–23 the assessment of formaldehyde concentration in products is critically important to evaluate the potential health effects of FD preservatives. The most commonly used analytical methods for qualitative and quantitative detection of formaldehyde are high performance liquid chromatography (HPLC) separation followed by ultraviolet/visible light (UV/Vis) detection, which are not specific for non-hydrated formaldehyde. They can; however, accurately describe the presence and quantity of formaldehyde equivalents (both non-hydrated formaldehyde and methylene glycol).24, 25 Accordingly, published values for “formaldehyde” levels should be taken to mean formaldehyde equivalents. Nuclear magnetic resonance (NMR) spectrometry techniques have also been used to measure formaldehyde,26 although the experimental conditions employed (e.g., temperature, pH, etc.) may be different in formulations and under real-life conditions of consumer use.
In relation, the CIR has recently published a report on the safety of formaldehyde and methylene glycol.27 Concentrations of formaldehyde in personal care products containing FD preservatives also have been reported,28 and in another recent study,29 levels of formaldehyde in air were measured after subjects used a variety of personal care products such as shampoo, conditioner, deodorant or moisturizer. Only very low levels, i.e., 1–11 micrograms/m3 of air, of formaldehyde were detected, and the authors concluded that inhalation of formaldehyde from the use of personal care products containing FD preservatives poses no risk to human health.29
Functional Use of FD Preservatives
FD preservatives are effective against both Gram-negative and Gram-positive bacteria. They are especially efficacious against Pseudomonads, organisms of concern in the personal care and cosmetic industry. The antifungal activity of FD preservatives is variable, depending on the compound and the formulation, but by combining FD preservatives with antifungal materials such as parabens, a broader spectrum of activity can be achieved. The benefit of using combinations of preservatives is improved efficacy at lower total concentrations of preservatives.30 Another benefit of FD preservatives is their ability to remain efficacious and stable over time.
Each type of preservative has benefits and limitations.26 The choice of preservation systems for personal care products is influenced by a variety of factors including product type, packaging materials, safety and efficacy for controlling microbial growth, compatibility with other formulation ingredients and cost. Readers should note that no single type is suitable for and effective in all types of personal care products. Individual FD preservatives may not be suitable for a particular formulation due to solubility, color, pH or other factors. Nevertheless, FD preservatives are important components of the repertoire of preservatives available for personal care products. Parameters such as the activity of FD preservatives over a wide pH range (pH 4–10), their compatibility with a variety of surfactants and other ingredients, their water solubility and their ability to be incorporated into formulations at ambient temperatures constitute significant factors in their favor.
The Question of Alternatives
To provide an array of choices for consumers, some formulators may seek alternative preservatives. However, the assessment and substitution of alternative preservatives in products is a technically complex process. It is highly unlikely that an alternative could be a technically simple and cost-effective drop-in replacement for FD preservatives. For example, additional safety tests, microbial challenge tests, and stability and consumer use tests would be needed for any potential replacement preservative. Additional concerns relate to aesthetic effects that alternatives could impart in formulations, including changes in color, viscosity, and fragrance. Such changes could also alter product compatibility with packaging, processing and storage.
Conclusions
In summary, FD preservatives are safe and effective as used in personal care products and cosmetics. FD preservatives have protected consumers of personal care products and cosmetics for many decades, and their safety has been extensively documented.
Acknowledgments: The author would like to thank the Personal Care Products Council Microbiology Committee for its assistance in the preparation of this article.
References
Send e-mail to: [email protected].
1. U.S. Food and Drug Administration, Guide to inspections of cosmetic product manufacturers, available at www.fda.gov/ICECI/Inspections/InspectionGuides/ucm074952.htm (Accessed Apr 22, 2013)
2. ISO, Guidelines for the risk assessment and identification of microbiologically low-risk products, ISO 29621, available at www.iso.org (2010)
3. www.youtube.com/watch?v=UPjoOQDyQMo (Accessed Apr 30, 2013)
4. PA Geis, Preservation of cosmetics and household products: Rationale and application, Dev Ind Microbiol 29 305-315 (1988)
5. MD Lundov, L Moesby, C Zachariae and JD Johansen, Contamination versus preservation of cosmetics: A review on legislation, usage, infections and contact allergy, Contact Derm 60 70-78 (2009)
6. D Steinberg, Frequency of preservative use, Cosm & Toil 125 46-51 (2010)
7. U.S. Food and Drug Administration, Voluntary cosmetic registration program (VCRP), available at: www.fda.gov/Cosmetics/GuidanceComplianceRegulatoryInformation/ VluntaryCosmeticsRegistrationProgramVCRP/default.htm (Accessed Apr 22, 2013)
8. V Turoski, ed, Formaldehyde analytical chemistry and toxicology, American Chemical Society, Washington, DC (1985) p 103
9. JF Walker, Formaldehyde, 3rd edn, Table 14 (1964) pp 59-61
10. W Paulus, ed, Directory of microbicides for the protection of materials–A handbook, Springer Verlag, NY (2005) p 276
11. www.cir-safety.org
12. SCCS opinion concerning the determination of certain formaldehyde releasers in cosmetic products (2002), available at http://ec.europa.eu/food/fs/sc/sccp/out188_en.pdf, adopted by the SCCNFP during the 22nd plenary meeting (Dec 17, 2002)
13. Cosmetic ingredient review, Final report on the safety assessment of DMDM hydantoin, Int J Toxicol 7 245-77 (1988)
14. Cosmetic ingredient review, Imidazolidinyl urea, J Environ Pathol Tox 4(4) 133-146 (1980)
15. Cosmetic ingredient review, Imidazolidinyl urea, Int J Toxicol 22(S1) 1-35 (2003)
16. Cosmetic ingredient review, Final report on the safety assessment of diazolidinyl urea, Int J Toxicol 9 229-245 (1990)
17. Cosmetic ingredient review, Final amended report on the safety assessment of diazolidinyl urea, International J Toxicol 27(Suppl. 1) 77-142 (2008)
18. LC Becker et al, Final report of the amended safety assessment of quaternium-15 as used in cosmetics, Int J Toxicol 19 (Suppl 2) pp 98S-114S (2010)
19. SCCS opinion on quaternium-15, https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_077.pdf (Accessed Apr 22, 2013) pp 1-50
20. H Heck and M Cassanova, The implausibility of leukemia induction by formaldehyde: A critical review of the biological evidence on distant-site toxicity, Regul Toxicol Pharm 40(2) 92-106 (2004)
21. V Turoski, ed, Formaldehyde Analytical Chemistry and Toxicology, American Chemical Society, Washington, DC (1985) pp 318-2018
22. VJ Feron, HP Til, F de Vrijer, RAWoutersen, FR Cassee and PJ van Bladeren, Aldehydes: Occurrence, carcinogenic potential, mechanism of action and risk assessment, Mutat Res-/Genet Tox 259 363–385 (1991)
23. World Health Organization, Formaldehyde, Environ Health Crit, no. 89 (1989)
24. K Kijima et al., A study on release of formaldehyde from its donor type preservatives, Anal Sci 7 913-916 (1991)
25. Oregon OSHA and CROET at Oregon Health and Sciences University, “Keratin-based” hair smoothing products and the presence of formaldehyde, available at www.orosha.org/pdf/Final_Hair_Smoothing_Report.pdf (2010) pp 1-32
26. D Emeis, W Anker and KP Witten, Quantitative 13C NMR spectroscopic studies on the equilibrium of formaldehyde with its releasing cosmetic preservatives, Anal Chem 79(5) 2096-2100 (2007)
27. Cosmetic ingredient report, Final amended report: Formaldehyde and methylene glycol, Cosmetic ingredient review, Washington, DC (2011) pp 1-41
28. D Takahiro, K Kajimura and S Taguchi, Survey of formaldehyde (FA) concentration in cosmetics containing FA-donor preservatives, J Health Sci 56 116-122 (2010)
29. MA Lefebvre, WJ Meuling, R Engel, MC Coroama, G Renner, W Pape and GJ Nohynek, Consumer inhalation exposure to formaldehyde from the use of personal care products/cosmetics, Regul Toxicol Pharmacol 63 171-176 (2012)
30. DC Steinberg, Preservatives for Cosmetics, 3rd edn, Allured Business Media, Carol Stream, IL USA (2012)