Age-related changes in skin appearance including dryness, wrinkling and laxity, are influenced by the behavior of keratinocytes and fibroblasts. As individuals age, skin cells replace themselves more slowly and the lower layer of the skin, the dermis, decreases in thickness. The network of elastin and collagen fibers that allows human skin to stretch and retract begins to unravel.1-3
On the face, thinner and less elastic skin tends to sag and fold. Permanent crease lines and crow’s-feet form on the brow and around the eyes. Tiny wrinkles form around the lips. The tissues of the jaw and neck droop into jowls and double chins. Skin aging is affected by genetic factors such as skin color, environmental factors such as prolonged exposure to the sun,4,5 and the consequences of a lifestyle involving smoking, rapid weight loss and stress.6,7 The aging process cannot be stopped or reversed but good daily skin care can slow the process.
This article describes a placebo-controlled study conducted to determine if the combination of dimethylamino-ethanol, retinol and mineral salts has a positive effect on one aspect of skin aging, the appearance of wrinkles in facial skin.
Three Cosmetic Ingredients
Different groups of molecules are now recognized and used to slow skin aging. Retinoid and α-hydroxy acid (AHA) families and polypeptides are the most frequently used, alone or in combination.
Retinol: Retinol is one of the most effective molecules now on the market for counteracting the effect of cutaneous aging.8,9 Easily absorbed by the epidermis,10 it increases cellular renewal in the basal layer of the epidermis, normalizes cellular differentiation and regulates the keratinization process.11–13 Retinol also has an effect on the dermis. Studies conducted on mice14 and humans15,16 have shown that retinol increases the quantity of collagen in the dermis. It also has been shown to increase skin elasticity and to reduce the depth of wrinkles on women.17
DMAE: Prescribed for many years as a treatment for cognitive disorders due to its remarkable brain-enhancement effects,18–21 dimethylaminoethanol (DMAE) has recently become a popular skin-firming cosmetic ingredient (see DMAE’s Mechanism of Action). Clinical trials have demonstrated the safety and efficacy of DMAE in antiaging formulations.22,23
Mineral salts: The use and development of mineral salts in dermatology are based on the discovery of several interaction sites in the skin. Zinc, copper and magnesium appear to play a role in skin aging.
Zinc is one of the most abundant trace elements in the human body.24 Zinc is the cofactor of more than 90 different enzymes. It protects them from free radicals and UV radiation attacks and regulates keratinization and fibroblast proliferation. Zinc is also involved in melanogenesis and in metabolism of fatty acids, and vitamins A and E.25 Topically applied zinc speeds wound healing, regulates sebum secretion, and exhibits antiseptic and antibacterial activity.
During aging, the natural level of zinc in the skin diminishes.26 Low concentrations of zinc in the epidermis have been observed in older people and may be the result of aging-reduced activity of epidermal enzymes. Conversely, zinc deficiency can retard the activity of other enzymes, leading to intensification of the aging processes.24
Copper also plays a role in skin via keratin synthesis and has a scavenging effect through the activation of the Cu-Zn superoxydismutase. It induces collagen production by fibroblasts thanks to the lysyl oxidase activation. In melanocytes, copper stimulates tyrosinase, therefore promoting
melanin precursor synthesis and photoprotection.25
Magnesium is a mineral of primary importance in the body because it aids in the activation of adenosine triphosphate (ATP), the main energy source for cell functioning. Magnesium also activates several enzyme systems and is important for the synthesis of RNA and DNA. It is important for the synthesis of several amino acids.
DRMC: In the present study, authors investigate the in vivo effects of a cosmetic product containing DMAE, retinol and mineral salts on wrinkles. The mineral salts were a mixture of magnesium aspartate, zinc gluconate and copper acetate. These ingredients have been formulated into a proprietary combination that will be called DRMC in this article. The components of DRMC were chosen because, as already stated, the association of DMAE to mineral salts11 or to retinol12 increases the antiaging efficacy of the final product.
Materials and Methods
A double-blind, randomized, placebo-controlled study was conducted to test the efficacy of an antiaging product containing DRMC.
Subjects: In the present study, 124 female subjects were divided into two groups: one treated with the DRMC-containing antiaging product (n = 62) and one treated with a placebo (n = 62). Each subject was Caucasian, between the ages of 45 and 66, and with wrinkles and fine lines on her face.
The participants were accepted into the study only after receiving a medical examination and giving their free and informed consent. Women with serious illness or under medical treatment that would likely interfere with the efficacy evaluation were excluded. Also excluded were women who had used a cosmetic containing AHA for two weeks preceding the start of the study or who had followed a treatment containing retinol, retinoic acid or vitamin A for the two months preceding the start of the study. A final exclusion was for women who had recently suffered from sunstroke or who had followed heliotherapy during the month preceding the study.
Treatments: Both the test cream containing the DRMC and the placebo cream were white o/w creams, differing primarily by the presence of DRMC as the active ingredient. The placebo had a formula very similar to that of the test cream, but it was not exactly the vehicle of the test cream. Confidentiality prevents disclosure of the complete formulations of the two creams, but it can be said that all the ingredients, except DRMC, are well-known ingredients without any cutaneous rejuvenation efficacy. The concentrations of DRMC were chosen so they provided an efficacy in vivo but remained in the range acceptable for a cosmetic product. The two creams were randomly assigned to the subjects.
Schedule: At baseline (T0), a trained assessor performed a clinical assessment of the wrinkles present on the complete face. Photographs of the face were taken. Skin replicas were created outlining the right or left eye crow’s-feet wrinkles and upper part of the cheek, the forehead, and the area between the eyebrows. The choice of right or left side was determined randomly.
Each subject completed a controlled application of the cream under normal conditions of use and in the presence of laboratory staff. A sufficient amount of the cream was spread with gentle taps from the external corner of the eye to the internal corner, favoring particularly the outline of the eye and the cheekbone, avoiding application too close to the eyelashes. The cream was then applied on the rest of the face, with special attention to the forehead, between the eyebrows, and the area under the chin. Three hours after application, clinical evaluations were performed again and another set of skin replicas was obtained. The purpose of repeating the clinical evaluation and the skin replicas after a 3-hr interval was to take into account the possible immediate short-term firming/tightening effect of topically applied DMAE,27 i.e., the ability of DMAE to induce contraction or shrinkage of keratinocytes both in vitro and in vivo.
After the initial applications, the cream was applied once daily in the morning, seven days a week for 12 consecutive weeks, by the subject at home on cleansed skin and under normal conditions of use.
After 4, 8 and 12 weeks (T4, T8, T12), each subject returned to the laboratory for a controlled application of the cream. Three hours later, clinical assessment, photographs and skin replicas were taken again on the zones previously defined at T0. This comparison was used to take into account both the short- and long-term firming effects of the DRMC.
Assessing parameters and analyzing data: Facial wrinkles were evaluated by a trained assessor on a 12-cm visual analogical scale. For each subject, seven areas were scored on the face: the forehead, crow’s-feet, wrinkles between eyebrows, nasal troughs, cheeks, the periauricular area and the area under the chin.
Replicas of the wrinkles present on the forehead, at crow’s-feet and between the eyebrows were obtained with a silicone rubberamaterial applied each visit at the same location identified by the particular subject’s beauty marks or a mask in translucent plastic with anatomic marks specific to the subject.
Skin imprints were analyzed using a skin analyzerbwith softwarec, enabling the researchers to obtain a set of standardized parameters characterizing the cutaneous relief including the total number of wrinkles, total wrinkled surface and total length of the wrinkled surface.
Statistical analysis: The arithmetic mean values and standard errors on the mean of each parameter were determined at each time point of the study and for each treatment group for the skin relief parameters. The arithmetic mean values and the standard deviations for the scores of individual values collected from each subject were also calculated. For both the DRMC group and the placebo group, an intragroup comparison was performed between baseline values and the values obtained 3 hr after the application of the cream at T0, T4, T8 and T12 weeks. Also performed at those time points were intergroup comparisons of the DRMC group versus the placebo group.
Results
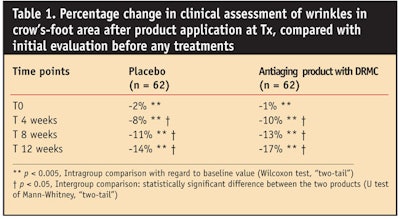
Clinical assessment: For both the active- and placebo-treated groups, a progressive and statistically significant reduction in the appearance of facial wrinkles was observed on all areas of the face considered (P < 0.05; Wilcoxon test) (data not shown). At the end of the study and for DRMC-treated group, the grading of wrinkles on the forehead and between the eyebrows was reduced respectively by 19% and 17%. On cheeks and nasal troughs, the wrinkle grades decreased by 22% and 17%, respectively. After four weeks of using the DRMC cream, a statistically significant difference in the appearance of crow’s-feet wrinkles was demonstrated in comparison to the placebo treatment (see Table 1).
The effects of the DRMC treatment lasted until the end of the study: reductions in wrinkle grades were recorded at 17% in the DRMC-treated group after 12 weeks, versus 14% in the placebo-treated group (P < 0.05; U test of Mann-Whitney). Photographs of one subject before and after the 12 weeks of DRMC treatment are shown in Figure 1.
Cutaneous relief analysis on skin replica: In both groups, depending on the localization, some replicas were not assessable because of problems removing them from the skin. Therefore, the results are given on fewer than 62 replicas.
- Crow’s-foot wrinkles. No significant variation in crow’s-feet wrinkle parameters was measured (see Table 2) in the placebo-treated group, except after 12 weeks of treatment. At that time, the total length of the wrinkled surface was reduced by 6%. On the other hand, compared to initial evaluation, DRMC treatment induced a significant reduction in the three parameters analyzed starting from the fourth week of use. The maximum effect was obtained after 8 weeks of application: a 19% reduction in the total number of wrinkles was observed, as well as 16% decrease in the total wrinkled surface. The total length of the wrinkled surface diminished by 25%. These effects remained stable after 12 weeks of treatment. The overall efficacy showed statistically significant improvement obtained faster with the DRMC treatment than with the placebo treatment.
- Forehead wrinkles. A statistically significant effect was observed with both the DRMC cream and the placebo cream (data not shown). Twelve weeks after the beginning of the study, treatment with either cream led to a comparable and significant reduction in forehead wrinkles.
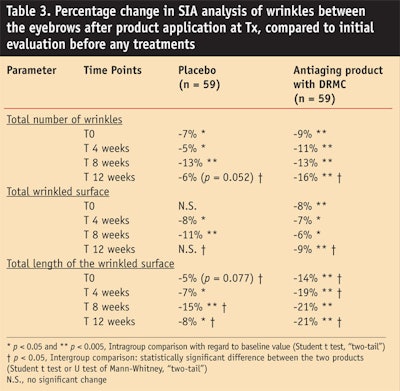
- Wrinkles between the eyebrows. Compared to the placebo treatment, the DRMC treatment showed a statistically significant effect (see Table 3) on the total length of the wrinkled surface as soon as after the first application, but also after 4 and 12 weeks of use. The wrinkles between the eyebrows were reduced 21% with the DRMC cream versus 8% with the placebo at T12 (P < 0.05 with the Student t test). At the end of the treatment, the total number of wrinkles as well as the total wrinkled surface between the eyebrows was significantly less in the DRMC-treated group than in the placebo-treated group.
Even with only a placebo treatment, some improvement was noticed, as in the case of forehead wrinkles. The placebo did not contain moisturizing ingredients, for example, but there is always a moisturizing effect when applying a cosmetic product onto the skin. A real placebo with no physiological effect is nearly impossible to design.
Discussion
The aim of the present double-blind, randomized, placebo-controlled study was to test the efficacy of a combination of retinol, DMAE and mineral salts on wrinkle severity. The 12-week study demonstrated the efficacy of this specific combination.
An obvious improvement of the appearance of facial wrinkles was correlated with a significant reduction of the wrinkles from the crow’s-foot area and between the eyebrows. This improvement was not simply an effect of moisturization, although both creams may have had a moisturizing effect. However, in the DRMC group, a significant improvement versus placebo was observed after four weeks of use on crow’s-feet area (Table 1). This significant difference between the two groups lasted until the end of the study, proving the results were not simply a moisturizing effect but that real antiwrinkle activity had occurred in that area. This activity was confirmed by a crow’s-foot wrinkle analysis with SIA (Table 2). A significant improvement was observed only in the DRMC-treated group and the difference between the products was statistically significant.
The fact that DRMC’s antiaging effect occurred more quickly around the eyes than on the forehead might at first seem inconsistent. In the first case a significant difference was observed between the DRMC cream and the placebo, whereas in the second case no difference was observed. This could be explained by the fact that the forehead wrinkles are deeper and more marked than in the crow’s-feet area. The antiwrinkle effect would be observed first in the crow’s-feet area and later on the forehead.
DMAE has well-documented positive effects for skin firming, skin permeation, antiaging and wrinkle reduction (as discussed in DMAE’s Mechanism of Action). The combination of DMAE with mineral salts (mixture of magnesium aspartate, zinc gluconate and copper acetate) was previously demonstrated in vitro to synergistically stimulate fibroblasts in the synthesis of collagen and glycosaminoglycan and to stimulate cell proliferation.28
On the other hand, a study performed on human skin explants has shown that retinol combined with DMAE significantly stimulates collagen synthesis, raises the number of dividing cells in the basal skin layer and increases epidermis thickness.29
The present study confirmed the in vivo efficacy on human skin of this particular combination of cosmetic ingredients—DMAE, retinol and mineral salts—as a new skin antiaging treatment having both short-term and long-term firming properties.
References
1.NA Fenske and CW Lober, Structural and functional changes of normal aging skin, J Am Acad Dermatol 15 571–585 (1986)
2.C Escoffier et al, Age related mechanical properties of human skin, J Invest Dermatol 93 353–357 (1989)
3.Y Takema, Y Yorimoto, M Kawai and G Imokawa, Age related changes in the elastic properties and thickness of human facial skin, Br J Dermatol 131 641–648 (1994)
4.EF Bernstein et al, Long term sun exposure alters the collagen of the papillary dermis. Comparison of sun protected and photo-damaged skin by northern analysis, immunohistochemical staining and confocal laser scanning microscopy, J Am Acad Dermatol 34 209–218 (1996)
5.GJ Fischer, The pathophysiology of photoaging of the skin, Cutis 75 5–9 (2005)
6.MF Demierre, D Brooks, HK Koh and AC Geller, Public knowledge, awareness and perceptions of the combination between skin aging and smoking, J Am Acad Dermatol 41 27–30 (1999)
7.JS Koh, H Kang, SW Choi and HO Kim, Cigarette smoking associated with premature facial wrinkling: image analysis of facial skin relicas, Int J Dermatol 41 21–27 (2002)
8.CEM Griffiths, The role of retinoids in the prevention and repair of aged and photoaged skin, Clin Exp Dermatol 26 613–618 (2001)
9.REB Watson et al, Retinoic acid receptor alpha expression and cutaneous ageing, Mech Ageing Dev 125 465–473 (2004)
10.C Caldera, L Kersaudy, J Badoual and G Olive, The cutaneous absorption of vitamin A, Dev Pharmacol Ther 7 213–217 (1984)
11.S Kang et al, Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoid acid but without measureable retinoic acid levels or irritation, J Invest Dermatol 105 549–556 (1995)
12.R Eichner et al, Effects of long-term retinoic acid treatment on epidermal differentiation in vivo: specific modifications in the programme of terminal differentiation, Br J Dermatol 135 687–695 (1996)
13.BA Gilchrest, Treatment of photodamage with topical tretinoin: an overview, J Am Dermatol 36 S27–S36 (1997)
14.E Schwartz, FA Crueshank, JA Mezik and AM Kligman, Topical all-trans retinoic acid stimulates collagen synthesis in vivo, J Invest Dermatol 96 975–978 (1991)
15.CE Griffiths et al, Two concentrations of topical tretinoin (retinoic acid) cause similar improvement of photoaging but different degrees of irritation. A double-blind, vehicle-controlled comparison of 0.1% and 0.025% tretinoin creams, Arch Dermatol 131 1037–1044 (1995)
16.J Varani et al, Vitamin A antagonizes decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin, J Invest Dermatol 114 480–486 (2000)
17.C Bertin et al, Retinol + Melibiose: An innovative anti-aging combination, 20th IFSCC Congress (1998)
18.RE Osvaldo, 2-Dimethylaminoethanol (deanol): a brief review of its clinical efficacy and postulated mechanism of action, Curr Ther Res Clin Exp 6 1238–1242 (1974)
19.JA Lewis and R Young, Deanol and methylphenidate in minimal brain dysfunction, Clin Pharmacol Ther 17 534–540 (1975)
20.CL Saccar, Drug therapy in the treatment of minimal brain dysfunction, Am J Hosp Pharm 35 544–552 (1978)
21.JF Flood, GE Smith and A Cherkin, Memory retention: potentiation of cholinergic drug combinations in mice, Neurobiol Aging 4 37–43 (1983)
22.R Grossman, The role of dimethylaminoethanol in cosmetic dermatology, Am J Clin Dermatol 6 39–47 (2005)
23.CA Cole and C Bertin, Dimethylaminoethanol: a new skin-care ingredient for aging skin, In Textbook of Cosmetic Dermatology, 3rd edn, R Baran and HI Maibach, eds, Boca Raton: CRC Press (2004)
24.A Frydrych, J Arct and K Kasiua, Zinc: a critical important element in cosmetology, J Appl Cosmetol 22 1–13 (2004)
25.B Dreno, Oligoéléments et peau, Cosmétologie 8 44–47 (1995)
26.G Brooks, The importance of zinc in the ageing process and the cosmetics effects of a novel zinc yeast derivative, Proceed IFSCC Conference “Biocosmetics–Skin aging,” vol II (1993) p 89
27.US Pat Applic 20060193777, Method of screening compounds for potential efficacy for the treatment of signs of aging, MD Southall et al (Aug 31, 2006) www.freepatentsonline.com/20060193777.html
28.C Bertin et al, Anti-aging efficacy of the combination of dimethylaminoethanol (DMAE) and mineral salts, Proceedings of the 23rd IFSCC Congress, Orlando, 2004, IFSCC (2004)
29.A Robert, J Serrano, T Oddos and G Ries, Use of a skin explant model for evaluating a new combination anti-aging technologies: retinol and dimethylaminoethanol (DMAE), Proceedings of the 23rd IFSCC Congress, Orlando, 2004, IFSCC (2004)
30.I Nagy and K Nagy, On the role of cross-linking of cellular proteins in aging, Mech Ageing Dev 14 245–251 (1980)
31.BB Michniak, Amines and amides as penetration enhancers, In Percutaneous Penetration Enhancers, E Smith and H Maibach, eds, Boca Raton: CRC Press (1995) p 80
32.SA Grando, Biological functions of keratinocyte cholinergic receptors, J Invest Dermatol Proc 2 41–48 (1997)
33.I Uhoda et al, Split face study on the cutaneous tensile effect of 2-dimethylaminoethanol (deanol) gel, Skin Res Technol 8 164–167 (2002)